Abstract
Currently, there are no treatment options available for the deadly contagious disease, coronavirus disease 2019 (COVID-19). Drug repurposing is a process of identifying new uses for approved or investigational drugs and it is considered as a very effective strategy for drug discovery as it involves less time and cost to find a therapeutic agent in comparison to the de novo drug discovery process. The present review will focus on the repurposing efficacy of the currently used drugs against COVID-19 and their mechanisms of action, pharmacokinetics, dosing, safety, and their future perspective. Relevant articles with experimental studies conducted in-silico, in-vitro, in-vivo, clinical trials in humans, case reports, and news archives were selected for the review. Number of drugs such as remdesivir, favipiravir, ribavirin, lopinavir, ritonavir, darunavir, arbidol, chloroquine, hydroxychloroquine, tocilizumab and interferons have shown inhibitory effects against the SARS-CoV2 in-vitro as well as in clinical conditions. These drugs either act through virus-related targets such as RNA genome, polypeptide packing and uptake pathways or target host-related pathways involving angiotensin-converting enzyme-2 (ACE2) receptors and inflammatory pathways. Using the basic knowledge of viral pathogenesis and pharmacodynamics of drugs as well as using computational tools, many drugs are currently in pipeline to be repurposed. In the current scenario, repositioning of the drugs could be considered the new avenue for the treatment of COVID-19.
Similar content being viewed by others
Introduction
Drug repurposing is the process to identify the new indications for existing drugs and considered as an efficient and economical approach [1]. It is also known as repositioning, re-profiling, re-tasking and rescue of drugs [2]. It has been considered that 75% of known drugs could be repositioned for various diseases [1]. Outbreaks of novel emerging infections such as coronavirus disease 2019 (COVID-19) have unique challenges in front of the health professionals to select appropriate therapeutics/pharmacological treatments in the clinical setup with very little time available for the new drug discovery [3]. Further, development of a vaccine for any disease including COVID-19 takes time and even if the process is put on accelerated mode it would take 18–20 months to introduce it as ready-to-use product. Currently, no specific treatment is available against the new virus severe acute respiratory syndrome coronavirus 2 (SARS-CoV2). Hence, the search for effective therapeutic agents to tackle COVID-19 is vital and urgent. The discovery and licensed use of a drug come with a long-gestation period. The cost of the new drug development process amounts to more than a billion dollars extending for a period of 10–15 years [4] with the success rate of only 2.01% [5]. This creates a lag in the productivity of pharmaceutical research to develop a new drug which results in a persistent gap between therapeutic needs and available treatments [6]. Considering the time and cost required for coming up with new therapies, probing the existing antiviral and other drugs against SARS-CoV2 is cost-effective. In recent times, repurposing of available drugs for the management of several disease conditions is increasingly becoming a popular strategy as it uses de-risked compounds with known preclinical, pharmacokinetic, pharmacodynamic profiles which can directly enter phase III or IV clinical trial making the drug development process potentially a low-cost and relatively rapid [7]. Therefore, reassessing the efficacy of licensed and experimental drugs has become go to choice of World Health Organization (WHO) and other health agencies to treat emerging health problems.
Drug repurposing follows mainly two concepts. One is that a single drug interacts with multiple targets, which paves the way for searching new target sites of action for the known compound [8, 9]. The other concept is that targets associated with a disease are often relevant to a number of biological processes of pathogenesis [6, 10] which paves the way for designation of a new indication for the known target. Notionally, a drug that acts on these common elements can, in principle, be useful for several disorders.
Different repurposing approaches
Broadly, there are three kinds of approaches which are widely used in drug repositioning: computational approaches, biological experimental approaches, and mixed approaches. Data such as gene expression, drug-target interactions, protein networks, electronic health records, clinical trial reports, and drug adverse event reports has become accessible in standardized forms. The repository of knowledge and omics data available in pharmaceutical research leads to the rise of some computational methods which are novel and exciting in the field of drug repositioning. These computational methods are capable to make a high-level integration of all the knowledge and data and help in understanding the new signaling pathways and generate novel insights into drug mechanisms, side effects, and interactions which further speed up drug discovery. A recent study presented integrative network-based systems pharmacology, methodology which quantifies the interplay between the coronavirus–human cell interactome and drug targets in the human protein–protein interaction network which help in rapid identification of repurposable drugs against SARS-CoV2. A study using this kind of approach was able to identify 30 potential repurposable drugs against COVID-19 [11, 12].
The SARS-CoV2 mainly spreads through the respiratory tract [13] and affects alveolar cells. Unlike, Middle East respiratory syndrome coronavirus (MERS-CoV) which employs dipeptidyl peptidase 4 (DPP4) [14], SARS-CoV and SARS-CoV2 adopt angiotensin-converting enzyme 2 (ACE2) as receptor for entering the cell. Attachment, fusion and entry of the virus are aided by spike protein which makes it an interesting target for the development of antibodies, entry inhibitors and vaccines [15]. Anti-viral agents including ritonavir, lopinavir, ribavirin, interferons have been used against SARS-CoV and MERS-CoV clinically, however, clinical data is still limited [16]. Currently, with the lack of effective agents against SARS-CoV2 as well as public-health emergency, WHO has identified some therapies which doctors and researchers believe are the most promising, such as a combination of two HIV drugs (lopinavir and ritonavir), anti-malarial drugs (chloroquine and hydroxychloroquine), and an experimental antiviral compound remdesivir. Further, many available drugs with the intention of repurposing against COVID-19 have been subjected to clinical trials [17] (Table 1). However, the search for other agents cannot pause while waiting for the results because the demand for new effective agents is huge.
COVID-19 pandemic
Human population of the twenty-first century is suffering a large-scale epidemic of highly pathogenic coronaviruses such as severe acute respiratory syndrome coronavirus (SARS-CoV) in 2002–03 [18], Middle East respiratory syndrome coronavirus (MERS-CoV) in 2012 [19] and severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) in 2019–20. Coronaviruses are pleomorphic, enveloped, positive sense, 27–32 kb large RNA viruses with typical crown-shape glycoprotein spikes (peplomers) that cause respiratory and enteric diseases in humans and other animals [20]. Very high recombination rates lead to constant transcription errors and RNA-dependent RNA polymerase (RDRP) jumps in coronaviruses [21] give them a chance to develop into diverse zoonotic pathogens such as SARS-CoV2. SARS-CoV2 is a positive-sense single-stranded RNA virus classified as a strain of the species SARS-CoV, (genus beta-coronavirus; subgenus sarbecoronavirus; subfamily orthocoronavirinae; family coronaviridae) which earlier in 2002–03 caused an outbreak of SARS in Guangdong, China [22]. Overall genome sequence of SARS-CoV2 showed 79.5% similarity with SARS-CoV and interestingly, 96.2% similarity with bat coronavirus RaTG13, suggesting its origin from a bat virus [23, 24]. Though not certain, yet it is suspected that the introduction of this new coronavirus to humans might have been facilitated by an intermediate host, the pangolins [25].
As per WHO report, a case of pneumonia of unknown causative pathology from Wuhan city of China was first time reported to the WHO Country Office on 31 December 2019. The outbreak of the coronavirus-associated acute respiratory disease throughout the world was named as COVID-19 and it was classified as 2019-nCoV (SARS-CoV2) [26]. The classification and naming of the virus were done by the Coronaviridae Study Group of the International Committee on Taxonomy of Viruses. Novel coronavirus genome sequencing by China was publicly available on the Global Initiative on Sharing All Influenza Data (GISAID) platform on 12 January 2020. First case of novel coronavirus outside of China was reported in Thailand on 13th January 2020 (WHO). The novel coronavirus outbreak was declared as Public Health Emergency of International Concern on 30 January 2020 (WHO). This disease has been given name as COVID-19 by the WHO on 11th February 2020. As on 11th March 2020, COVID-19 was recognized as a pandemic by the WHO and there have been around 18,354,342 cases and 696,147 deaths reported due to COVID-19 upto 5th August 2020. Till date as per WHO, there is no treatment available for the control of COVID-19. In the current scenario of lacking efficient and specific treatments for SARS-CoV2 infections and the urgent need to restrain the pandemic, drug re-tasking appears to be the most suitable tool to find out the best therapeutic option for COVID-19 [27].
The mechanisms of infection by the SARS-CoV2 are not clear yet, however, it is genetically similar to SARS-CoV and other coronaviruses [28]. Hence the common mode of pathogenicity is being explored for targets of treatment. The therapeutic targets depending on the common mode of pathogenicity can be classified as virus-related targets and host-related targets.
Repurposed drugs that act through virus-related targets such as RNA genome
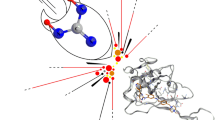
Replication of SARS-CoV2 depends directly on the key enzyme RDRP. It is interesting to note that potential drug targets protease and polymerase for SARS-CoV2 and SARS-CoV are highly conserved with 96% and 97% overall identity. Therefore, blockers developed against the SARS may act as good therapeutic candidates to bind protease or polymerase sites against SARS-CoV2 (GISAID, 2020) (Fig. 1).
Therapeutic targets of the currently considered drugs for repurposing against COVID-19. 1. Coronaviruses suppress the antiviral immunity, hence maintaining an antiviral state with interferons. 2. Virus enters the cell by fusion of the viral spike proteins with cellular ACE2 receptor, followed by ACE2 downregulation. Angiotensin receptor blockers (ARBs), Angiotensin-converting enzyme inhibitors (ACEIs) and statins increase ACE2 expression, hence may have efficacy in this condition. 3. Fusion is followed by endocytosis of the virus, where low endosomal pH helps in lysis of the viral structural proteins. Disruption of this acidic environment by diprotic bases like chloroquine and hydroxychloroquine may produce an antiviral effect. 4, 5. Release of nucleic acid (NA) into the cytoplasm and translation of the viral proteins using host ribosomes, 6. Proteolysis by viral main protease enzyme makes functional proteins e.g. RNA dependent RNA polymerase (RDRP). Thus, inhibition of the main protease enzyme by inhibitors like lopinavir, ritonavir and darunavir, may have efficacy against the virus. 7. RDRP is essential for replication and transcription of the virus. RDRP inhibitors, remdesivir, favipiravir, ribavirin and arbidol may be effective against coronaviruses. 7, 8. Subsequent translation and proteolysis into structural proteins followed by packaging makes intact virions, which get exocytosed (9) from the cell
Remdesivir
On the basis of current knowledge about the use of remdesivir in SARS-CoV2 infection, remdesivir is considered as the potential drug candidate for repurposing against the COVID-19. Remdesivir (GS-5734) is a nucleoside analog originally developed against Ebola viruses by Gilead Sciences Inc, a USA based Biopharmaceutical Company (Table 2). Though the drug failed to show effect against Ebola, the preliminary results from in-vitro and in-vivo preclinical studies as well as case reports indicate its efficacy against SARS-CoV2. Recently, in Vero E6 cells, remdesivir has been shown to block the viral infection at low concentration (EC50 = 0.77 μM) with high selectivity index (SI > 129.87). The EC90 value (1.76 µM) was also low enough to be achieved in non-human primate models [29]. It also efficiently inhibited SARS-CoV2 infection in human liver cancer Huh-7 cells [23]. In Rhesus macaques, the drug showed therapeutic and prophylactic efficacy against both SARS and MERS coronaviruses, indicating its potential against diverse coronaviruses including SARS-CoV2 [30]. Similarly, in a Ces1c-knockout mouse model of SARS-CoV infection, remdesivir in both prophylactic and therapeutic use significantly reduced lung viral titers, though the survival and lung pathology were not improved [31]. Treatment with intravenous remdesivir on day 7 of hospital admission, clearly improved the clinical condition from the next day in a 35-year-old COVID-19 patient in Washington, USA [32]. In Italy, remdesivir has been used (200 mg every 12 h as a loading dose followed 100 mg every 12 h for 10 days thorough intravenous route) with other supportive therapy in the clinical management of COVID-19 patients presented with a range of modified early warning score (MEWS) from less than 3 to more than 4 [33]. In a recent report, clinical improvement in 36 of 53 patients (68%) of severe COVID-19 patients has been observed with the compassionate-use of remdesivir in a cohort study [34]. According to another study remdesivir was superior over the placebo in shortening the time to recovery in adult patients hospitalized with COVID-19. In the trial that was conducted at 60 sites and 13 subsites in different parts of the world, mortality rate was 7.1% with remdesivir and 11.9% with placebo by 14 days in 1059 patients in which 538 were in remdesivir group and 521 were in the placebo group [35]. Another trial, however, suggests differently, that remdesivir has no such statistically significant clinical benefits, although, patients who were administered with remdesivir showed a faster clinical improvement in numerical terms, with symptom duration of 10 days or less in a study of 237 patients in which 158 patients received remdesivir and 79 (one withdrew) were on placebo in a randomised, double-blind, placebo-controlled, multicentre trial at ten hospitals in Hubei, China [36]. Another clinical trial done with remdesivir in severe COVID-19 patients without mechanical ventilation support indicated no significant difference between 5-day and 10-day course of remdesivir therapy, however, by day 14, a clinical improvement was noticed in 64% of patients in the 5-day group and in 54% in the 10-day group at 2 points or more on the ordinal scale out of 7-point ordinal scale [37].
Being a nucleotide (adenosine) analog, remdesivir gets incorporated into the replicating genome of the virus after being converted into its triphosphate form. The triphosphate forms compete with adenosine triphosphate (ATP) to act as a substrate of RDRP and have been found to cause significantly efficient incorporation as compared to ATP. Remdesivir adds three more nucleotides before terminating the growing RNA chain [38]. The extra three nucleotides may protect the inhibitor from removal by the viral 3′–5′ exonuclease activity [39], contributing to the lack of acquiring resistance.
The pharmacokinetics, metabolism and distribution of remdesivir have been studied in non-human primates, previously. In rhesus monkeys, 10 mg/kg dose yields a half life (t1/2) of 0.39 h with fast systemic elimination. The active metabolite of remdesivir, 1′-cyano-substituted adenine C-nucleoside ribose analogue (Nuc) then appears which produces antiviral activity [40]. However, the drug is parenterally administered through intravenous route and is not expected to be given to mildly infected patients, though potent action is expected in mild infection. It is under clinical trial in China and USA against COVID-19 by 15th of April, 2020 (NCT04252664 and NCT04257656). However, seven clinical trials are underway on remdesivir to evaluate its efficacy on SARS-CoV2 infection (https://www.gilead.com/purpose/advancing-global-health/covid-19/remdesivir-clinical-trials). The results of NCT04257656 clinical trial showed that intravenous remdesivir was adequately tolerated by COVID-19 patients [36]. In the third trial (NCT04292899) with the severely infected COVID-19 patients, with the use of remdesivir, common adverse effects were observed such as nausea, acute respiratory failure, increased alanine aminotransferase (ALT) and constipation [37]. In all these studies ramdesivir was administered intravenously as 200 mg SID on day 1 and followed by 100 mg SID for the subsequent period.
Favipiravir
As per the information obtained from the Chinese news channel by Dong and Co-workers, this drug got an approval against the SARS-CoV2 infected patients in China on 15th February, 2020 since it had drastically attenuated the illness of the SARS-CoV2 infected patients [41]. Favipiravir (6-fluoro-3-hydroxy-2-pyrazinecarboxamide, T-705 also marketed as AVIGAN) was developed by a Japanese firm, Toyama Chemical Co., Ltd. and has been approved in Japan for Influenza since 2014 (Table 2). The drug effectively inhibits the enzyme RDRP. In Vero E6 cells this drug showed an EC50 of 61.88 µM and selectivity index of more than 6.46 against SARS-CoV2 [29]. Though this concentration against SARS-CoV2 is very high, the drug may be tested in an animal model based on its performance against Ebola virus. Remarkably this drug had shown 100% efficacy against Ebola in a mice model although it had a very high EC50 in Vero cells [42]. Randomized trials are being conducted for this drug in combination with other drugs against coronaviruses (Favipiravir + interferon-α, ChiCTR2000029600, favipiravir + baloxavir, ChiCTR2000029544).
Favipiravir is a nucleoside precursor which inhibits the broad range of influenza virus strains, however, it shows antiviral activity through its NTP form (converted into an active phosphoribosylated form, T-705 RTP) via direct inhibition of the RDRP activity of influenza A virus polymerase and it has also shown lethal mutations within the viral genome [43]. Nevertheless, the exact mode of action, precise molecular mechanism and its interaction between the nucleotide and the viral polymerase could be investigated in the SARS-CoV2 infection.
Favipiravir exerts its antiviral action in a dose-dependent manner [44]. Its oral bioavailability is close to 100% and has a short half life of 2–5.5 h [45, 46]. In Humans, the plasma protein binding of favipiravir is 54%. Favipiravir undergoes metabolism in the liver mainly by aldehyde oxidase (AO) and partially by xanthine oxidase (XO) and the inactive oxidative metabolite (T-705 M1) is excreted by kidneys. A study reported that in Pichindé arenavirus infection the kinetics of absorption, elimination and time to maximum drug concentration is altered. The T-705 M1 levels were higher in the infected animals [47]. Further, favipiravir concentration may be increased by drugs those undergo metabolism through AO such as raloxifene, tamoxifen, estradiol, cimetidine, felodipine, amlodipine, verapamil, propafenone, amitriptyline, zaleplon, citalopram, sulindac and famciclovir. Concomitant administration of acetaminophen and favipiravir showed increased area under curve (AUC) of former drug possibly due to inhibition of the sulfate transferase by the latter drug [46].
The drug did not show any toxicity at the oral dose of 500 mg/kg/day for 10 days in guinea pigs [48]. This can be converted to 108 mg/kg of human equivalent dose [49] which is far greater than the dose prescribed for COVID-19 on first day i.e. 53 mg/kg/day (1600 mg BID on the first day) (https://www.clinicaltrialsarena.com/comment/influenza-favipiravir-covid-19/). However, use of this drug may be approved with the availability of more number of clinical results.
Ribavirin
Ribavirin, a broad-spectrum antiviral drug, is a guanosine analog approved for treating hepatitis C virus in combination, and respiratory syncytial virus as monotherapy. Effect of this drug has been assessed in patients with SARS [50] and MERS [51] (Table 2). Against SARS-CoV2, it has very high EC50 of 109.50 μM and selectivity index more than 3.65 in Vero cells. Ribavirin in its monophosphate form inhibits host inosine monophosphate dehydrogenase (IMPDH) enzyme that controls intracellular guanosine triphosphate (GTP) pools [52]. Exhaustion of intracellular GTP pool indirectly inhibits viral RDRP enzyme. It also interferes with mRNA capping. Ribavirin at a dose rate of 500 mg 2–3 times/day in combination with other drugs such as lopinavir/ritonavir or interferon (IFN)-α through intravenous route for not more than 10 days made the SARS-CoV2 infected patients more resistant to respiratory distress syndrome as well as death [41].
The oral bioavailability of ribavirin is 52%, which is due to modest first-pass metabolism in liver [53]. Estimated half-life is 3.7 h [54]. Ribavirin-induced hemolytic anemia is a most commonly reported adverse effect and frequently it requires dose reduction [55]. Further, close monitoring of renal impairment in terms of creatinine clearance during therapy is required. Old age, decreased renal function, low body weight and female gender are other risk factors to be considered during ribavirin therapy [56, 57].
Animal studies have shown teratogenic potential of ribavirin. Therefore, exposure during pregnancy should be avoided. Other less common yet pertinent adverse effects are bronchospasm and pulmonary edema on inhalation [43, 57]. Neutropenia, thrombocytopenia, skin rashes, anorexia and depression are some minor adverse effects of ribavirin that need to be monitored in the sensitive patients [58]. In a recent clinical trial with combinations of ribavirin + interferon-alpha, lopinavir/ritonavir + interferon-alpha and ribavirin + lopinavir/ritonavir + interferon-alpha in patients with mild to moderate COVID-19 showed that ribavirin plus lopinavir/ritonavir combination showed a significant increase in gastrointestinal adverse effects [59].
Repurposed drugs acting through polypeptide packing
The viral RNAs get translated into polypeptide chains which get cleaved into functional proteins before packing into virions. The viral main proteases are responsible for the cleavage of these polypeptide chains [60]. SARS-CoV2 protease has 96% overall similarity with SARS-CoV [61]. Protease inhibitors used in HIV-1 therapy are shown to be effective against SARS-CoV [62]. In-silico and in-vitro approaches have been used to validate the inhibition of SARS-CoV2 protease by HIV-1 protease inhibitors [63, 64]. Numerous protease inhibitors are approved by FDA for use in HIV therapy. The agents available from this class include saquinavir, amprenavir, indinavir, nelfinavir, ritonavir, and lopinavir. Few of them are being considered against SARS-CoV2 (Fig. 1).
Lopinavir-ritonavir combination
This drug combination is available under brand name Kaletra and was developed by Abbott Laboratories, USA. This drug was approved by FDA in the year 2000 as an anti-retroviral for the treatment of HIV patients. Lopinavir is rapidly degraded in the human body by the host proteases, hence is given with ritonavir (another protease inhibitor) at a lower dose, which helps lopinavir remain active for a longer time by inhibiting the metabolizing enzyme cytochrome P450 [65] (Table 2).
Coronavirus main proteases are cysteine proteases whereas HIV main proteases are aspartic proteases. Nonspecific protease inhibition by protease inhibitors used in HIV therapy has been found effective against SARS-CoV. Lopinavir has comparable binding energies against the SARS-CoV2 and HIV-1 proteases [66]. In SARS-CoV2 patients treated with lopinavir-ritonavir, a significant virus clearance has been achieved [67, 68]. A 47-year old patient, who failed to respond to methylprednisolone and interferon therapy, got quick improvement with additional lopinavir and ritonavir tablets therapy [69]. In 36 paediatric patients between the age of 0–16 years with confirmed COVID-19 from three hospitals in Zhejiang, China, use of lopinavir-ritonavir (syrup twice a day) along with interferon α to 14 patients (6 patients needed oxygen inhalation but not sure whether it included lopinavir-ritonavir group) showed a mean of hospital stay time of 14 days and all were cured [70]. Based on these initial successful reports, SARS-CoV2 main protease has been docked for 1. 3 billion protease inhibitor compounds [71]. Also, lopinavir-ritonavir has been included in SOLIDARITY trial (NCT04330690).
However, in a recent clinical trial with 199 severely ill patients with confirmed SARS-CoV2 infections, lopinavir-ritonavir treatment did not improve the time to clinical improvement and mortality rate beyond standard care and further, some patients also showed adverse drug effects such as gastrointestinal disturbances [72]. In a study on four COVID-19 patients treated with Western (lopinavir 400 mg/ritonavir in a ratio of 400 mg to 100 mg for q12 h through oral route), arbidol (0.2 g, three times in a day through oral route), and Chinese traditional medicine Shufeng Jiedu capsule (SFJDC, 2.08 g, three times in a day through oral route) combination for 6–15 days, it has been observed that out of the two mild and two severe SARS-CoV2-infected pneumonic patients, three patients showed significant improvement in pneumonia associated symptoms and the remaining patient with severe pneumonia has also shown signs of improvement till the date of reporting [73].
Most HIV protease inhibitors show poor bioavailability. They are extensively metabolized by microsomal CYP3A4 enzymes. Other agents that induce or inhibit these metabolizing enzymes influence their effectiveness. Side effects associated with the use of protease inhibitors include diarrhea, vomiting, diabetes, hypertriglyceridemia, and hypercholesterolemia. It also could cause severe hepatic damage [74]. Therefore, a lot of clinical and experimental study regarding the use of lopinavir-ritonavir is required to reach a conclusive statement.
The most common adverse effect of ritonavir/lopinavir is diarrhea and gastrointestinal disturbance. Elevated liver enzymes, dyslipidemia, asthenia, headache and skin rashes are some minor side effects [75,76,77]. Ritonavir/lopinavir co-administration causes mild hepatotoxicity (elevated alanine aminotransferase, ALT) [78]. However, sole use of ritonavir at a higher dose (600 mg/BID) may increase the risk of severe hepatotoxicity [79]. A rarely observed adverse effect with ritonavir is retinal pigment epitheliopathy. The risk factors for the retinopathy are high dose and hepatic impairment [80].
Darunavir
Darunavir is a second-generation non-peptide protease inhibitor effective against HIV-1. It has a distinct chemical structure that enhances binding affinity and reduces dissociation rate, making it more potent than the other protease inhibitors [81] (Table 2). Using computational drug design methods, darunavir was identified as one of the promising hits for inhibition of chymotrypsin-like protease of SARS-CoV2 [82]. Results of a structural analysis showed no binding of darunavir to SARS-CoV2 protease (https://www.eacsociety.org/home/covid-19-and-hiv.html). Anyhow, recently in Shanghai, in-silico and an enzyme activity test based drug screening revealed 30 agents with potential antiviral activity against SARS-CoV2 including darunavir (https://www.simm.ac.cn/xwzx/kydt/202001/t20200125_5494417.html). Interestingly, therapeutic doses of darunavir are reported to be too low to cause cytotoxic effects, affording a wide margin of safety [81]. In an in-vitro study, darunavir at 300 micromolar concentration was found to inhibit SARS-CoV2 virus replication by 280 times in comparison to the untreated group (https://www.sd.chinanews.com/2/2020/0205/70145.html). Further, darunavir has been used (600 mg tablet every 12 h) along with other anti-viral drugs and supportive therapy in the clinical management of COVID-19 patients presented with a range of MEWS from less than 3 to more than 4 in Italy [33].
Darunavir is rapidly absorbed after oral administration and has a terminal elimination half-life of 15 h. Approximately 95% of the drug is plasma protein bound and metabolized exclusively by CYP3A4. Therefore, co-administration of small doses of ritonavir (CYP3A4 inhibitor) increases the bioavailability of darunavir. Combination therapy with other CYP3A4 inhibitors (e.g. statins) with darunavir/ritonavir requires caution or is even contraindicated [83, 84].
A recent study correlated the daurnavir use with increased risk of myocardial infarction in HIV patients and concludes that cardiovascular disease (CVD) risk increases with darunavir exposure. Therefore, it may be used cautiously in patients with underlying cardiac diseases [85]. Before, regular use of this drug, more pharmacological profile may further be investigated during the use in COVID-19 patients.
Repurposed drugs acting through host targets such as antiviral immunity
The pattern recognition receptors in immune cells recognize the viral pathogen-associated molecular pattern (PAMPs) to stimulate antiviral interferon responses in the host [86]. The secreted interferons activate hundreds of interferon-stimulated genes which encode proteins with profound effects against the virus (Fig. 1).
Interferons (pegylated IFNα-2a and pegylated IFNα-2b)
The interferons (IFNs) are antiviral molecules classified into type I (IFNα, IFNβ, IFNω, and IFNτ) and type II (IFNγ). IFNα plays a critical role in innate immunity against viral infection which prompts its use in the treatment of many clinical viral infections. The recombinant IFNα is interferon alfacon-1. The pegylated types are pegylated IFNα-2a and pegylated IFNα-2b [87]. Pegylated interferon alfa-2b acts to target B cells through host interferon receptor, IFNAR1 signalling and enhances immune response against viral infections [88]. Recombinant human IFNα-2b has been shown to possess wide antiviral spectrum, low toxicity and high therapeutic index in vitro. Quantitative reverse transcriptase-polymerase chain reaction (RT-PCR) results revealed anti-viral effect of recombinant human IFNα-2b on respiratory viruses such as influenza B virus, parainfluenza virus, respiratory syncytial virus and coronavirus which was stronger in comparison to the effect of ribavirin [89]. In another study, IFNα-2b administered by nasal spray reduced positive rates of immunoglobulin M (IgM) antibody against all four respiratory viruses (parainfluenza virus, influenza B, adenovirus and respiratory syncytial virus) possibly suggesting the lower virus titre due to IFNα-2b [90]. In the rhesus macaque, IFNα-2b with ribavirin showed very good effect against beta-coronavirus emerged in Saudi [91], however, was unsatisfactory in humans [92]. In China, intranasal IFNα (5 × 106 U) twice a day in combination with ribavirin is one of the guidelines for the treatment of COVID-19 patients [41, 93]. It was further showed that the infection rate of SARS-CoV2 was decreased by IFNα-2b sprays [94]. IFNα-2b has demonstrated potent anti-viral activities against respiratory viruses and may serve for the prevention and treatment of SARS-CoV2 as well. Nevertheless, thorough evaluation in appropriately designed randomized trials is recommended before forging on.
PEG-IFNα-2b is a linear 12 kDa molecule and susceptible to hydrolysis. It is absorbed quickly and distributed widely. After administration PEG-IFNα-2b gets hydrolysed and the free IFNα-2b circulates in the body. Most of the free IFNα-2b excreted solely through kidneys. Therefore, it requires dose adjustment in patients with renal impairment. Further, the clearance of PEG-IFNα-2b decreases after repeated dosing. Therefore, it is administered according to body weight. Conversely, the PEG-IFNα -2a is a 40 kDA branched chain molecule which is absorbed more slowly and has poor tissue distribution. PEG-IFNα -2a has long half-life. Therefore, it is used at a fixed dose of 180 μg per week for the treatment of hepatitis C virus infection. PEG-IFNα-2a is metabolized by both the kidneys and liver, therefore this drug does not require a major dose modification [95].
Number of adverse events was recorded with PEG-IFNα therapy. Dizziness, headache, depression, fatigue, insomnia, alopecia, myalgia, arthralgia, pyrexia, anorexia are the most common constitutional symptoms observed with PEG-IFNα-2a and PEG-IFNα-2b therapy in various other diseases. Hematologic adverse events such as leucopoenia, thrombocytopenia and myelosuppression, thyroid disease, lung disease and retinopathy are the other adverse events recorded in the patients receiving PEG-IFNα-2a/PEG-IFNα-2b [96,97,98]. Dose limiting, mild injection site reactions/inflammations were observed commonly in PEG-IFNα -2b therapy [96]. Most of the adverse events are well tolerated and become less severe during the progression of therapy. The neuropsychiatric adverse effects were observed in patients undergoing interferon-α therapy and may require timely medical interventions [99]. In the case of severe events, discontinuation and dose reduction will help to overcome these adverse events [96].
Lopinavir-ritonavir and interferon-β combination
Lopinavir-ritonavir along with interferon beta is undergoing clinical trial (MIRACLE) against MERS in Saudi Arabia wherein a total of 76 patients have enrolled till Jan 2020 [100]. Interferons are the antiviral weapons of the body and coronaviruses are known to reduce the host antiviral immunity by suppressing the production of interferons. So, replacement therapy with interferons or interferon inducers has the potential to reduce the viral load in the body. They augment the host response to the viral infection. Lopinavir-Ritonavir-Interferon-β1b treatment improved the clinical findings and lowered the lung viral load in MERS-affected common marmosets [101]. Results of a recent clinical trial (NCT04276688) on COVID-19 patients showed the safety and efficacy of this combination. Adverse events like self-limited nausea and diarrhoea were recorded in this study. However, there was no significant difference in the adverse effects between the combination group and ritonovir/lopinavir alone (control group) [102].
Repurposed drugs targeting the virus uptake pathways
The coronaviruses enter the cell by two ways, (1) endocytosis, virus is taken up into the cell along with the endosomes, (2) fusion of the viral spike protein with the cell surface receptor ACE2 [103], the latter being the predominant pathway of virus entry [104]. Blockade of entry pathways may be effective targets for treatment (Fig. 1).
Chloroquine and hydroxychloroquine
Chloroquine, primarily known for its anti-plasmodium actions, has antiviral activity as well. This drug originally derived from Cinchona plant is now largely a synthetic drug (4-amino quinoline) discovered by Bayer Laboratories. Chloroquine and its analogs are potent inhibitors of most coronaviruses [105] (Table 2).
Chloroquine and hydroxychloroquine are weak diprotic bases. These drugs take a similar pathway like the virus and concentrate in the endosomes increasing the pH of the endosomal fluid. The acidic pH of the endosomes is necessary for the optimal activity of viral enzymes responsible for proteolysis and post-translational modification of nascent proteins. Disruption of acidic pH thus blocks the replication and lifecycle of the virus [106]. In addition, the drug is known to interfere with the glycosylation of the host receptor for the virus, ACE2. Faulty terminal glycosylation may affect the binding and subsequent entry of the virus into host cells [107].
Cell culture studies in African green monkey Vero E6 cells indicate significantly higher potency for chloroquine compared to hydroxychloroquine [108]. Chloroquine has EC90 value of 6.90 μM against the SARS-CoV2 in Vero E6 cells, which can be clinically achievable after administration of 500 mg as shown in rheumatoid arthritis patients [109]. A safe dosage of 6–6.5 mg/kg per day of hydroxychloroquine could yield serum levels of 1.4–1.5 μM in humans [110]. It is speculated that with a safe dosage, hydroxychloroquine could achieve concentration in the above tissues to inhibit SARS-CoV2 infection. Hydroxychloroquine phosphate (400 mg tablet every 12 h as a loading dose followed 200 mg tablet every 12 h for 10 days) or chloroquine phosphate (250 mg of two tablet every 12 h for 10 days) along with other anti-viral drugs and supportive therapy have been used in the clinical management of COVID-19 patients presented with a range of MEWS from less than 3 to more than 4 in Italy [33]. Chloroquine and hydroxychloroquine have been considered for SOLIDARITY trial (NCT04330690). Hydroxychloroquine has shown side effects such as prolonged QT interval and heart failure, though controversy exists. Recently, WHO stopped hydroxychloroquine arm of the Solidarity Trial to find an effective COVID-19 treatment. This recommendation was developed on the basis of data obtained from Solidarity trial (including the French Discovery trial data and UK's Recovery trial data). These trials showed that hydroxychloroquine did not result in the reduction of mortality of COVID-19 patients who were hospitalized when compared with standard care (briefed on 17 June 2020 by WHO).
Use of chloroquine for SARS-CoV2 asks for a high dose but an overdose of chloroquine has been reported to cause poisoning and death [111]. In comparison, hydroxychloroquine is safer with 40% less toxicity in animals [112]. Oral absorption of chloroquine and hydroxychloroquine in humans is efficient. Both the drugs distribute similarly in different tissues with high concentrations in the liver, kidney, lungs and spleen [113].
Chloroquine is two to three times as toxic in animals as hydroxychloroquine [112]. The acute toxicity of chloroquine causes death due to cardiac and respiratory arrest [111]. Adverse effects of chloroquine/hydroxychloroquine therapy at the therapeutic doses include retinopathy, myopathy, electrocardiographic changes, bleaching of hair, pruritus, headaches, dizziness and gastrointestinal upset [114,115,116]. Hydroxychloroquine retinal toxicity in the patients is far more common in patients taking this drug for greater than 5 years with the overall prevalence of 7.5% [117]. A study showed even with the recommended therapeutic dose of hydroxychloroquine produced bilateral maculopathy and it was attributed to the differential susceptibility of the retinal epithelium to hydroxychloroquine [118].
Hydroxychloroquine and chloroquine are orally well absorbed and show 70–80% oral bioavailability. Chloroquine is 60% bound to plasma proteins and distributed extensively. Following administration, chloroquine is rapidly dealkylated to pharmacologically active desethylchloroquine and bisdesethylchloroquine. Metabolism of hydroxychloroquine is similar to chloroquine except for a third metabolite desethylhydroxychloroquine which is also produced during metabolism. Both hydroxychloroquine and chloroquine have prolonged half-lives, between 40 and 50 days, and low blood clearance by kidneys and liver. For both chloroquine and hydroxychloroquine, approximately 40–60% is excreted as an unchanged or metabolized drug through the kidneys, 8–25% is excreted in an unchanged or changed form in the feces, 5% is sloughed off through the skin, and 25–45% is stored long term in lean body tissues [119,120,121,122].
Hydroxychloroquine should be cautiously used in patients with known hepatic or renal dysfunction. The co-administered drugs with hydroxychloroquine excreted via liver/kidney may interact with hydroxychloroquine and modulates its pharmacokinetics and toxicity. Hydroxychloroquine has been reported to cause severe hypoglycaemia when co-administered with oral hypoglycaemic drugs. There have been few reports of mild to severe hepatic failure in patients with hydroxychloroquine treatment [117]. Racial difference in toxicity has also been reported wherein the incidence of pericentral maculopathy was common in Asian (50%) than Caucasian (2%) patients [123]. Further, the use of chloroquine and hydroxychloroquine may be warranted in prophylaxis strategy as well as in COVID-19 patients with more number of clinical trials.
Arbidol
Arbidol, an indole-derivative, also known as umifenovir is a potent broad-spectrum antiviral agent. This drug has shown activity against a wide range of enveloped and non-enveloped viruses [124] (Table 2). It is effective against numerous pathogenic respiratory viruses and relatively very safe for use [125, 126]. Arbidol is an approved therapeutic agent against influenza in Russia and China [127]. Arbidol and arbidol mesylate were reported to act directly on viral replication of SARS-CoV at an early stage in vitro [128, 129]. The anti-viral mechanism of arbidol against influenza A and B involves viral fusion inhibition by hindering the hemagglutinin fusion machinery with the membrane, thus blocks virus entry into the cell [127]. Arbidol is one of the drugs in clinical trial phase 4 for pharmaceutical interventions of COVID-19 and treatment of COVID-19 patients with arbidol leads to a reduction in the mortality rate and increase in the recovery rate [130, 131]. Arbidol treatment coupled with lopinavir/ritonavir reckoned to retard the development of pulmonary lesions concurrently reducing the respiratory and gastrointestinal COVID-19 viral load thus lowering the transmission [132]. Encouragingly, seasonal prophylaxis with arbidol reduced influenza morbidity in patients with asthma and COPD during epidemic [133]. Four cases with mild to severe SARS-CoV2 pulmonary symptoms were subjected to antiviral therapy consisting of arbidol, lopinavir/ritonavir, and Shufeng Jiedu (traditional Chinese medicine) along with supportive care. Following medication, three patients showed a notable reduction in pneumonia-related manifestations and two of them were discharged after testing negative for SARS-CoV2 [73]. In COVID-19 patients, reduction in fever and cough; improvement in chest computed tomography (CT) and clinical status among the arbidol, lopinavir/ritonavir and control group remained statistically indifferent. However, arbidol was better tolerated by patients whereas test subjects administered with lopinavir/ritonavir experienced adverse events during the follow-up period [134].
The half-life of arbidol is between 17 and 21 h and 40% of the total drug excreted unchanged in feces. Arbdiol conjugation with glucuronide and sulfonation is evident. The major Phase I enzyme involved in its metabolism was CYP3A4, therefore, a possible interaction with CYP3A4 inducers/inhibitors may happen [135].
The drug has oral lethal dose (LD50s) of 340–400 mg/kg in mice, and > 3000 mg/kg in rats and guinea pigs. Intravenous LD50s are 109 mg/kg in mice and 140 mg/kg in rats. In long term animal studies, it showed no adverse effects with 10–50 times of human dose and no developmental toxicities were observed [127].
Angiotensin receptor blockers
It has been noticed that SARS-CoV2 binds the ACE2 receptors similar to the SARS coronavirus [23]. COVID-19 is associated with the acute respiratory distress syndrome (ARDS) and higher activity of ACE2 leads to attenuation in ARDS [136, 137]. ACE2 expression is downregulated in COVID-19 patients [138]. It is a general observation throughout the world that aged persons affected with COVID-19 suffer from high mortality. Association between COVID-19 infection and the process of chronological aging has been understood with the presence of two host receptors CD26 and ACE2 which are associated with senescence. The major pathway of virus entry into the host in case of SARS-CoV and SARS-CoV2 is through the target cell ACE2. Spike proteins of the virus attach to the cell surface ACE2 expressed in epithelial cells of the oral mucosa, lungs, intestine, blood vessels and kidney.
Angiotensin receptor blockers (ARBs) which inhibit the action of ACE, an isoform of ACE2, are known to increase the expression of ACE2 [139] (Fig. 1; Table 2). The patients with diabetes and cardiovascular diseases regularly take ACE inhibitors and ARBs. Hence, they remain under continuous risk of SARS-CoV2 infection. However, experimental studies with ARDS indicate a higher level of ACE2 reduces the severity of the disease [137]. Increased ACE2/ACE ratio may improve the host response to viral infection by correcting the endothelial dysfunction common to most viral infections [140]. Experimental and clinical studies are needed urgently on this aspect for repurposing of commonly used ARBs or ACE inhibitors (ACEIs). ARBs are well-tolerated drugs with minimal side effects in the population. However, a lot of patients from South America, Central America and Spain, had stopped or intended to interrupt their treatments with anti-hypertensive drugs like ACEIs such as enalapril or ARBs such as losartan fearing the risk of SARS-CoV2 infection [141]. However, another school of thought provided alternative suggestions about the use of renin–angiotensin–aldosterone system (RAAS) inhibitors. They suggested that though ACE2 expression may be enhanced with the use of RAAS inhibitors, sudden withdrawal of these drugs may be responsible for higher risk in patients of COVID-19 already affected with cardiovascular illnesses. Therefore, the use of RAAS inhibitors should continue in patients with COVID-19 under continuous observation [138, 142]. Out of 42 COVID-19 patients taking antihypertensive drugs, 17 patients who were treated with ACEIs/ARBs showed less mortality in comparison to patients taking drugs other than ACEIs/ARBs in Shenzhen Third People’s Hospital, China [143]. ACEIs and ARBs may improve the clinical condition of the COVID-19 patients in a clinical setting by maintaining a low level of IL-6 level in peripheral blood and through increment in a cluster of differentiation CD3 and CD8 T cell counts in peripheral blood and decreased the peak viral load [143]. Contrary to the previous report, another retrospective study from the Union Hospital of Wuhan, China done on 112 patients suggested that ACEI/ARBs medication has no difference in the critical group and general group and further, did not show any effect on the morbidity and mortality of COVID-19 patients associated with cardiovascular diseases [144]. It is reported that ACE inhibits the formation of Ang II in the presence of ACEIs; therefore, these prevent the negative effects induced due to AT1R as well as positive effects derived from the Ang II binding with AT2R and further, its transformation into Ang-(1–7) [145]. Therefore, it could be assumed that the use of ACEIs leads to the protective effects in the lung in COVID-19 patients.
Angiotensin receptor blockers are the most commonly used drugs for hypertension and related cardiac problems. In general, ACEIs and ARBs have few interactions with ritonavir [146, 147]. Generally, ARBs are well tolerated and the frequency of adverse reactions is less than 2%. There are no specific considerations for use in patients with hepatic and renal impairment [148]. ARBs are structurally related to losartan (except eprosartan) but the individual members exhibit diverse pharmacokinetic properties. Their bioavailabilities vary from 13–60% and half-lives are between 2 and 24 h. They are highly plasma protein bound (90–100%) and mostly metabolized and eliminated by the liver and, therefore, can be safely used in mild to moderate renal failure without any dose adjustment. However, losartan should be started with a lower dose in hepatic impairment and telmisartan should be avoided in case of renal impairment and congestive heart failure/digoxin therapy [149, 150].
Statins
Statins, the lipid-lowering drugs, have shown pleiotropic activity through anti-inflammatory and immunomodulatory properties to prevent acute lung injury in different experimental and clinical conditions; therefore, it may be used as re-tasking drug for the COVID-19 patients [151] (Table 2). Statins are commonly taken for a lifetime to prevent coronary artery diseases. Studies have reported that these drugs increase the ACE2 expression [152]. ACE2 cleaves Ang II, a peptide which promotes endothelial dysfunction and cardiovascular diseases, and produces Ang-(1–7) which counters the effects of Ang II [153]. Stimulation of ACE2/angiotensin-(1–7)/Mas and angiopoietin/Tie-2 signaling axes help restoring viral infection-induced endothelial dysfunction and maintain the homeostasis of the patients [154]. ACE2 activity has shown to be up-regulated with the use of ARBs and atorvastatin [152, 155] (Fig. 1). Statins being immunomodulatory have been hypothesized to work against MERS coronaviruses [156]. However, in animal models of SARS and MERS infections, reduced expression of the adapters, TIR-domain-containing adapter-inducing interferon-β (TRIF) or Myeloid differentiation primary response protein (MYD) 88 causes severe respiratory disease leading to death. Hence, the use of statins, which can further suppress MYD88 signaling, may exacerbate the disease condition [157].
Fungus derived statins (lovastatin, pravastatin and simvastatin) have elimination half-lives of 1–3 h and other synthetic compounds have elimination half-lives ranging from 1 h for fluvastatin to 19 h for rosuvastatin. Bioavailability of statins varies from 5–80% and 6–30% of the absorbed statins. Majority of satins are metabolized by the liver and the biliary excretion is the predominant route of excretion. Pravastaitn and rosuvastatin are excreted mostly unchanged by liver and kidneys. All statins are extensively bound to plasma proteins (> 90%) with the exception to pravastatin which is only 50% bound to plasma proteins [158].
Myopathy and rhabdomyolysis are the most frequent adverse effect of statins. Other common adverse effects include hepatotoxicity, peripheral neuropathy, cardiac toxicity, cognitive dysfunction, cataracts, diabetes mellitus and autoimmune necrotizing myopathy [159, 160]. Most of the statins are CYP3A4 substrates and the inhibitors of this enzyme may increase blood levels of statins and therefore associated with adverse effects [161]. Statins have potential interactions with the protease inhibitor drugs and they are contraindicated in combination therapy [162]. USFDA warns using atorvastatin, lovastain, rosuvsatin, pravstatin, simvastatin with antiviral protease inhibitor drugs (For list please see https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-interactions-between-certain-hiv-or-hepatitis-c-drugs-and-cholesterol#dose). In this case dose reduction and therapeutic drug monitoring will reduce the occurrence of adverse effects.
Drugs acting on host proinflammatory cytokines
Tocilizumab
Tocilizumab is a drug used against rheumatoid arthritis as well as cytokine release syndrome/systemic inflammatory response syndrome. This drug was introduced in Japan in the year 2008 and subsequently in the European Union (trade name RoActemra) in the year 2009. Later in the year 2010, it was used in the USA (Actemra) and this drug is an anti-human monoclonal antibody of the immunoglobulin G1k subclass [163]. This drug does not act on the virus but it acts to reduce the cytokine response of the host. Tocilizumab is a recombinant humanized monoclonal antibody which is responsible to bind the human IL-6 receptor and inhibiting its signal transduction pathway [164] (Fig. 1). A 60-year old man of Wuhan, China developed symptoms of chest tightness without fever and cough on 1st February 2020 and confirmed to be the first case of COVID-19 with multiple myeloma. The case was successfully treated with humanized anti–IL-6 receptor antibody, tocilizumab. The patient was administered 8 mg/kg tocilizumab through IV route for one time from day 9 to day 12 upon hospital admission and a decrease in IL-6 level was recorded, however, a rebound phenomenon of IL-6 level was observed, may be due to the recovery of the normal T cells and the patient was cured on day 19. The patient, however, had also received other therapies like 200 mg dose of umifenovir (arbidol) tablets orally for three times daily as antiviral treatment and 400 mg of moxifloxacin IV daily for three days [165]. However, in another 56 years old COVID-19 patient of end-stage renal disease (ESRD) in the USA treated with tocilizumab, hydroxychloroquine and broad spectrum antibiotics, the patient remained in critical condition throughout the study period [166]. A 57 years old woman from Switzerland suffering from systemic sclerosis (SSc) was on tocilizumab treatment for arthritis and SSc-associated interstitial lung disease (SSc-ILD) @ 8 mg/kg body weight every 4 weeks intravenously. On the basis of observation it has been reported that tocilizumab treatment given for chronic autoimmune diseases may prevent the development of severe COVID-19 [167]. A case of 42 years old patient with a respiratory failure linked to COVID-19 from France showed a rapid favourable outcome after two infusions of the tocilizumab at a dose rate of 8 mg/kg through intravenous route along with other therapy like lopinavir-ritonavir [168]. Tocilizumab treatment in 15 of the 20 patients showed improvement in terms of oxygen intake, lung lesion opacity (in 19 patients), lymphocytes in peripheral blood (10 patients), C-reactive protein (16 patients) and further, all patients were discharged on average 15.1 d after giving tocilizumab in a study conducted in China between 5 and 14 February 2020 [169]. A study on 100 COVID-19 patients with severe pneumonia treated with tocilizumab and other supportive therapy from 9–20 March 2020 in Italy, the respiratory condition was improved or stabilized in 77 patients in 10 days. 61 of these 77 patients had shown clearing of diffuse bilateral opacities on chest X-ray and 15 were discharged from the hospital; however, respiratory condition of 23 patients was worse during the treatment and 20 died out of 23 patients [170]. In another clinical study, tocilizumab administration showed a reduction in serum IL-6 level in 10 patients out of 15 patients, however, it could not decrease IL-6 level in four patients. Still, observations suggest that tocilizumab is a good treatment option in patients who have a risk of cytokine storms [171]. However, it is difficult to reach on a conclusion regarding the use of tocilizumab in COVID-19 patients with the limited number of clinical sample size.
The absolute bioavailability of tocilizumab following subcutaneous administration was estimated to be 79.5% [172]. Tocilizumab is eliminated from the body in a concentration-dependent manner. Therefore, its half-life is directly proportional to its serum concentration. At higher serum concentration, it shows linear elimination and has the terminal half-life of 21.5 days (https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/125276s127,125472s040lbl.pdf).
The notable adverse effect of tocilizumab is liver toxicity. Steatosis, steatohepatitis and focal hepatocellular necrosis were seen in this drug-induced hepatotoxicity. Other common adverse reactions observed were skin and soft tissue infections, dyslipidemia, transient neutropenia, headache, nausea and flu-like symptoms. Therefore, a full assessment for liver injury and dose adjustment is required in patients administered with tocilizumab [173, 174]. Tocilizumab also showed good efficacy in long term dosing for rheumatoid arthritis with similar adverse reactions and few malignancies also were reported [175].
Other probable potential re-tasking agents for the treatment of COVID-19
In a recent study, it has been demonstrated that an antiprotozoal agent nitazoxanide has shown antiviral activity against a number of viruses which include human and animal coronaviruses. It is used orally and chemically, this drug is nitrothiazoly-salicylamide, a broad-spectrum anthelmintic and antiviral prodrug which is metabolized to an active compound tizoxanide [176, 177] (Table 2). It had shown inhibitory potential against SARS-CoV2 at a low-concentration with 50% effective concentration of 2.12 μM in Vero E6 cells. It has been observed that antiviral activity of this compound may be due to interference with host-regulated pathways which are involved in viral replication rather than virus-specific pathways [178, 179].
Nafamostat is an anticoagulant in nature and further, its activity was also evaluated in the Vero E6 cells infected with SARS-CoV2. This drug is a synthetic serine protease inhibitor and inhibited the SARS-CoV2 at 50% effective concentration of 22.50 μM in Vero E6 cells. Further, it is speculated that it may act through the inhibition of membrane fusion by reducing the release of cathepsin B. As this agent has previously shown potential against the MERS-CoV through prevention of membrane fusion, it could also be recommended for further in-vivo evaluation against COVID-19 [29] (Fig. 2; Table 2).
Ivermectin is a broad-spectrum anti-parasitic agent and has also shown efficacy against some viral infections. This drug is basically a macrolide endectocide macrocyclic lactone derived from Streptomyces avermitilis with anthelmintic activity (Table 2). Recently, this drug has been investigated against SARS-CoV2 in in-vitro cell culture system in Australia. It reduced the viral RNA of SARS-CoV2 at two hours post infection in Vero-hSLAM cells with the addition of single dose and its reduction intensity was approximately 5000-fold. This study hypothesized that ivermectin may act through inhibition of importin (IMP) α/β1-mediated nuclear import of viral proteins similar to other RNA viruses, however; further investigation is required to understand the precise mechanism of action in SARS-CoV2 [180]. It is also important to conduct the trials in in-vivo animal models and further, the pre-clinical trials in human beings are also required with fast pace if in-vivo models results have potential to mitigate the COVID-19 infection (Fig. 2).
Cepharanthine, a naturally occurring plant alkaloid, is an anti-inflammatory and anti-neoplastic and is approved for leukopenia treatment (Table 2). Cepharanthine showed the most potent inhibition of GX_P2V infection and decreased the viral RNA yield in the pangolin coronavirus GX_P2V workable model, therefore, this therapeutic agent may also be a potential candidate for repurposing against COVID-19 [181]. Further, it has been speculated that this drug may target host cell pathways [181].
A gold-containing triethyl phosphine coated drug, auranofin, is used as therapy for the arthritis and approved from FDA for this treatment (Table 2). Previously, this drug has shown effects against viral, bacterial and parasitic infections. Recently, auranofin has shown inhibitory effect against viral RNA in Huh7 cells infected with SARS-CoV2 as well as a reduction in proinflammatory cytokines in an in-vitro study [182]. Endoplasmic reticulum (ER) stress and unfolded protein response (UPR) activation are responsible for the viral replication and pathogenesis in coronavirus infection [182, 183] and high levels of UPR activation has been recorded in the cells which have higher expression of SARS-COV spike protein [182, 184, 185]. Therefore, it is speculated that auranofin may act through the inhibition of redox enzymes such as thioredoxin reductase and induction of ER stress, however, its confirmatory investigation is required [182, 186].
Janus Kinase inhibitors have anti-inflammatory and antiviral effects and may have a purported advantage over other immunomodulatory strategies in COVID-19 [187]. JAK1/2 inhibitor ruxolitinib, baricitinib, JAK 1/3 inhibitor tofacitinib and pan-JAK inhibitor TD-0903 are presently under different phases of clinical trial and being investigated for COVID-19 treatment (https://www.pharmaceutical-technology.com/comment/incyte-eli-lilly-jak-inhibitor-covid-19/). A phase-I clinical study showed improvement in COVID-19 inflammation score with clinical improvement by ruxolitinib. However, it has toxicity on the liver and hematopoietic system. But in this study with the short term dosage, it did not show any major adverse events [188]. Baricitinib is presently under phase-III clinical trial (https://www.clinicaltrialsarena.com/news/lilly-baricitinib-covid-19-trial/). Initial studies showed the drug baricitinib corrected the immune dysregulation and improved clinical disease outcome with no serious adverse events [189, 190]. All the above drugs are under different phases of clinical trials and none have been approved for the treatment of COVID-19.
Dexamethasone is a corticosteroid and it has been used in some clinical trials. The preliminary results of RECOVERY trial conducted in 2104 patients showed dexamethasone @ 6 mg once daily for up to 10 days showed improvement in recovery with 22.9% mortality in the dexamethasone group compared to the 25.7% mortality in the usual care group [191]. However, few concerns have been raised on RECOVERY trial outcome in choosing the correct steroid and dose. In another study, methylprednisolone reduced the mortality rates in COVID-19 patients with ARDS. Further, the effect of dexamethasone in geriatric patients and effects on viral load are not mentioned in the RECOVERY trial [192]. Theoharides and Conti have also suggested restricting the use of dexamethasone during the initial phase of disease in severely ill patients for shorter period. Using this drug in recovery phase may lead to reduced viral clearance and nosocomial infections [193]. As it suppresses immunity and it may aggravate some latent infections whose occurrence is of no importance to the developed countries. Therefore, region-specific study is required before incorporating into COVID-19 therapy [194].
Molecular docking and computational modelling based approach to identify the therapeutic candidates for repurposing against COVID-19
A chemotherapeutic fluoroquinolone antibiotic, prulifloxacin, with broad-spectrum activity, and some anti-HIV drugs such as tegobuvir, (a novel non-nucleoside inhibitor of human coronavirus RNA replication), nelfinavir (a protease inhibitor which inhibits the cleavage of the polyprotein gag-pol) and bictegravir (HIV-1 integrase inhibitor) have protein binding sites of the proteases which have been shown using the high throughput screening as molecular docking with bioinformatics analysis and would be considered the potential candidates for re-tasking against COVID-19 in future [195] and in-vitro and in-vivo animal models may provide a lead against this disease. According to another recent molecular docking study based on RDRP modelling and multiple sequence alignment (MSA) showed a binding capacity to RDRP against SARS-CoV2 by various antiviral agents such as sofosbuvir (FDA approved against Hepatitis C virus), ribavirin, remdesivir and IDX-184; (under clinical trial against Hepatitis C virus) [196]. Elbasvir, an antiviral drug, (approved for the treatment of hepatitis C) has shown predicted multiple binding sites at RDRP, papain-like proteinase and helicase of SARS-CoV2 using the docking simulations and computational modelling [197] (Fig. 2; Table 2).
In a recent review, it has been stated that azithromycin, quercetin, rapamycin and doxycycline may have senolytic activity and azithromycin and doxycycline are used to inhibit viral replication and IL-6 production, therefore, these therapeutic agents may be potential re-tasking agents against COVID-19 in future with proof of sufficient in-vitro and in-vivo studies [198] (Fig. 2; Table 2).
Peptide (EK1), TDF, 3TC (RNA synthesis inhibitors), ShuFeng JieDu and Lianhuaqingwen capsules (Chinese traditional medicine) are screened using standard bioassays, chemical screening and genome and biophysical understanding of the targeted virus for being the potential drug candidates against SARS-CoV2 infection as described in a recent review. However, the efficacy and safety of these drugs for SARS-CoV2 still need to be further confirmed by clinical experiments [93] (Fig. 2).
Future perspective and conclusion
COVID-19 is a pandemic and has no treatment till date including vaccine and drugs. However, there are number of available drugs with approval of FDA for treatment of other diseases which could be used on the basis of the trial against COVID-19 and considered as the repurposed drugs. These are antivirals, antimalarials, ACEIs, ARBs, statins and monoclonal antibodies. It has been noticed that remdesivir, favipiravir, ribavirin, lopinavir-ritonavir combination, arbidol, tocilizumab have shown benefits in various clinical studies done on the basis of compassionate use to save the life of COVID-19 patients; therefore, in future, these drugs could be the potential drug therapy against this deadly disease. However, the use of chloroquine and hydroxychloroquine has shown the controversial results in the different trials, therefore, WHO also stopped their SOLIDARITY trials recently. In future, chloroquine and hydroxychloroquine require a large number of research studies to reach a conclusion for its use in COVID-19 patients. Further, ACEIs and ARBs could be the potential supportive therapy against this infection. Some drugs are in the early phase of investigation like ivermectin and auranofin to be used against the COVID-19 and these agents could be potential therapeutic agents in future. Molecular docking would be the central technique to identify the probable therapeutic agents against COVID-19 patients and the screened agents, thereby, could be verified for their effectiveness in in-vitro and in-vivo studies.
References
Huang F, Zhang C, Liu Q, Zhao Y, Zhang Y, Qin Y, et al. Identification of amitriptyline HCl, flavin adenine dinucleotide, azacitidine and calcitriol as repurposing drugs for influenza A H5N1 virus-induced lung injury. PLoS Pathog. 2020;16(3):e1008341. https://doi.org/10.1371/journal.ppat.1008341.
Scherman D, Fetro C. Drug repositioning for rare diseases: Knowledge-based success stories. Therapie. 2020;75:161–7. https://doi.org/10.1016/j.therap.2020.02.007.
Dyall J, Coleman CM, Hart BJ, Venkataraman T, Holbrook MR, Kindrachuk J, et al. Repurposing of clinically developed drugs for treatment of Middle East respiratory syndrome coronavirus infection. Antimicrob Agents Chemother. 2014;58:4885–933. https://doi.org/10.1128/AAC.03036-14.
Sertkaya A, Birkenbach A, Berlind A, Eyraud J. Examination of clinical trial costs and barriers for drug development. US Department of Health and Human Services, office of the assistant secretary for planning and evaluation report. 2014;1:1–92.
Yeu Y, Yoon Y, Park S. Protein localization vector propagation: a method for improving the accuracy of drug repositioning. Mol Biosyst. 2015;11:2096–102. https://doi.org/10.1039/c5mb00306g.
Hodos RA, Kidd BA, Shameer K, Readhead BP, Dudley JT. In silico methods for drug repurposing and pharmacology. Wiley Interdiscip Rev Syst Biol Med. 2016;8:186–21010. https://doi.org/10.1002/wsbm.1337.
Pushpakom S, Iorio F, Eyers PA, Escott KJ, Hopper S, Wells A, et al. Drug repurposing: progress, challenges and recommendations. Nat Rev Drug Discov. 2019 Jan;18(1):41–58. https://doi.org/10.1038/nrd.2018.168.
Paolini GV, Shapland RH, van Hoorn WP, Mason JS, Hopkins AL. Global mapping of pharmacological space. Nat Biotechnol. 2006;24:805–15. https://doi.org/10.1038/nbt1228.
Koch U, Hamacher M, Nussbaumer P. Cheminformatics at the interface of medicinal chemistry and proteomics. Biochim Biophys Acta. 2014;1844:156–61. https://doi.org/10.1016/j.bbapap.2013.05.010.
Piro RM. Network medicine: linking disorders. Hum Genet. 2012;131:1811–20. https://doi.org/10.1007/s00439-012-1206-y.
Zhou Y, Hou Y, Shen J, Huang Y, Martin W, Cheng F. Network-based drug repurposing for novel coronavirus 2019-nCoV/SARS-CoV-2. Cell Discov. 2020;6:14. https://doi.org/10.1038/s41421-020-0153-3.
Li X, Yu J, Zhang Z, Ren J, Peluffo AE, Zhang W, et al.Network bioinformatics analysis provides insight into drug repurposing for COVID-2019. Preprints 2020, 2020030286 (doi: 10.20944/preprints202003.0286.v1.
Tai W, He L, Zhang X, Pu J, Voronin D, Jiang S, et al. Characterization of the receptor-binding domain (RBD) of 2019 novel coronavirus: implication for development of RBD protein as a viral attachment inhibitor and vaccine. Cell Mol Immunol. 2020;17:613–20. https://doi.org/10.1038/s41423-020-0400-4.
Wang N, Shi X, Jiang L, Zhang S, Wang D, Tong P, et al. Structure of MERS-CoV spike receptor-binding domain complexed with human receptor DPP4. Cell Res. 2013;23(8):986–93. https://doi.org/10.1038/cr.2013.92.
Du L, He Y, Zhou Y, Liu S, Zheng BJ, Jiang S. The spike protein of SARS-CoV-a target for vaccine and therapeutic development. Nat Rev Microbiol. 2009;7:226–36. https://doi.org/10.1038/nrmicro2090.
Zumla A, Chan JF, Azhar EI, Hui DS, Yuen KY. Coronaviruses-drug discovery and therapeutic options. Nat Rev Drug Discov. 2016;15:327–47. https://doi.org/10.1038/nrd.2015.37.
Harrison C. Coronavirus puts drug repurposing on the fast track. Nat Biotechnol. 2020;38(4):379–81. https://doi.org/10.1038/d41587-020-00003-1.
Woodhead M, Ewig S, Torres A. Severe acute respiratory syndrome (SARS). Eur Respir J. 2003;21:739–40. https://doi.org/10.1183/09031936.03.00035403.
Zaki AM, Boheemena S, Bestebroer TIM, Osterhaus A, Fouchier R. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med. 2012;367:1814–20. https://doi.org/10.1056/NEJMoa1211721.
Woo PC, Huang Y, Lau SK, Yuen KY. Coronavirus genomics and bioinformatics analysis. Viruses. 2010;2:1804–20. https://doi.org/10.3390/v2081803.
Drexler JF, Gloza-Rausch F, Glende J, Corman VM, Muth D, Goettsche M, et al. Genomic characterization of severe acute respiratory syndrome-related coronavirus in European bats and classification of coronaviruses based on partial RNA-dependent RNA polymerase gene sequences. J Virol. 2010;2010(84):11336–49. https://doi.org/10.1128/JVI.00650-10.
LeDuc JW, Barry MA. SARS, the first pandemic of the 21st century. Emerg Infect Dis. 2004;10:e26. https://doi.org/10.3201/eid1011.040797_02.
Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–3. https://doi.org/10.1038/s41586-020-2012-7.
Benvenuto D, Giovanetti M, Ciccozzi A, Spoto S, Angeletti S, Ciccozzi M. The 2019-new coronavirus epidemic: evidence for virus evolution. J Med Virol. 2020;92:455–9. https://doi.org/10.1002/jmv.25688.
Shield C. Coronavirus: From bats to pangolins, how do viruses reach us?. Deutsche Welle. 7 February 2020. Retrieved 13 March 2020.
Gorbalenya AE, Baker SC, Baric RS, de Groot RJ, Drosten C, Gulyaeva AA, et al. The species severe acute respiratory syndrome-related coronavirus: classifying 2019-nCoV and naming it SARS-CoV-2. Nat Microbiol. 2020;5:536–44. https://doi.org/10.1038/s41564-020-0695-z.
Baron SA, Devaux C, Colson P, Raoult D, Rolain JM. Teicoplanin: an alternative drug for the treatment of coronavirus COVID-19? Int J Antimicrob Agents. 2020:105944. doi: 10.1016/j.ijantimicag.2020.105944.
Ahmed SF, Quadeer AA, McKay MR. Preliminary identification of potential vaccine targets for the COVID-19 Coronavirus (SARS-CoV-2) based on SARS-CoV immunological studies. Viruses. 2020;12(3):E254. https://doi.org/10.3390/v12030254.
Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020;30:269–71. https://doi.org/10.1038/s41422-020-0282-0.
de Wit E, Feldmann F, Cronin J, Jordan R, Okumura A, Thomas T, et al. Prophylactic and therapeutic remdesivir (GS-5734) treatment in the rhesus macaque model of MERS-CoV infection. Proc Natl Acad Sci USA. 2020;117(12):6771–6. https://doi.org/10.1073/pnas.1922083117.
Sheahan TP, Sims AC, Graham RL, Menachery VD, Gralinski LE, Case JB, et al. Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses. Sci Transl Med. 2017;9:eaal3653. https://doi.org/10.1126/scitranslmed.aal3653.
Holshue ML, DeBolt C, Lindquist S, Lofy KH, Wiesman J, Bruce H, et al. First case of 2019 novel coronavirus in the United States. N Engl J Med. 2020;382:929–36. https://doi.org/10.1056/NEJMoa2001191.
Nicastri E, Petrosillo N, Bartoli TA, Lepore L, Mondi A, Palmieri F, et al. National Institute for the infectious diseases "L. Spallanzani", IRCCS. Recommendations for COVID-19 clinical management. Infect Dis Rep. 2020;12:8543.
Grein J, Ohmagari N, Shin D, Diaz G, Asperges E, Castagna A, et al. Compassionate use of remdesivir for patients with severe Covid-19. N Engl J Med. 2020;382:2327–36. https://doi.org/10.1056/NEJMoa2007016.
Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, et al. Remdesivir for the treatment of Covid-19-preliminary report. N Engl J Med. 2020;NEJMoa2007764. doi: 10.1056/NEJMoa2007764.
Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020;395:1569–78. https://doi.org/10.1016/S0140-6736(20)31022-9.Erratum.In:Lancet.2020;395:1694.d.
Goldman JD, Lye DCB, Hui DS, Marks KM, Bruno R, Montejano R, et al. Remdesivir for 5 or 10 days in patients with severe Covid-19. N Engl J Med. 2020 May 27. https://doi.org/10.1056/NEJMoa2015301.
Gordon CJ, Tchesnokov EP, Feng JY, Porter DP, Gotte M. The antiviral compound remdesivir potently inhibits RNA-dependent RNA polymerase from Middle East respiratory syndrome coronavirus. J Biol Chem. 2020;295(15):4773–9. https://doi.org/10.1074/jbc.AC120.013056.
Agostini ML, Andres EL, Sims AC, Graham RL, Sheahan TP, Lu X, et al. Coronavirus susceptibility to the antiviral remdesivir (GS-5734) is mediated by the viral polymerase and the proofreading exoribonuclease. mBio. 2018;9(2):e00221–18. doi: 10.1128/mBio.00221–18.
Warren TK, Jordan R, Lo MK, Ray AS, Mackman RL, Soloveva V, et al. Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys. Nature. 2016;531:381–5. https://doi.org/10.1038/nature17180.
Dong L, Hu S, Gao J. Discovering drugs to treat coronavirus disease 2019 (COVID-19). Drug Discov Ther. 2020;14:58–60. https://doi.org/10.5582/ddt.2020.01012.
Oestereich L, Lüdtke A, Wurr S, Rieger T, Muñoz-Fontela C, Günther S. Successful treatment of advanced Ebola virus infection with T-705 (favipiravir) in a small animal model. Antiviral Res. 2014;105:17–211. https://doi.org/10.1016/j.antiviral.2014.02.014.
Jordan PC, Stevens SK, Deval J. Nucleosides for the treatment of respiratory RNA virus infections. Antivir Chem Chemother. 2018;26:2040206618764483. https://doi.org/10.1177/2040206618764483.
Pires de Mello CP, Tao X, Kim TH, Vicchiarelli M, Bulitta JB, Kaushik A, et al. Clinical regimens of favipiravir inhibit zika virus replication in the hollow-fiber infection model. Antimicrob Agents Chemother. 2018;62:e00967–18. doi: 10.1128/AAC.00967-18.
Nguyen TH, Guedj J, Anglaret X, Laouénan C, Madelain V, Taburet AM, et al. Favipiravir pharmacokinetics in Ebola-Infected patients of the JIKI trial reveals concentrations lower than targeted. PLoS Negl Trop Dis. 2017;11:e0005389. https://doi.org/10.1371/journal.pntd.0005389.
Du YX, Chen XP. Favipiravir: pharmacokinetics and concerns about clinical trials for 2019-nCoV infection. Clin Pharmacol Ther. 2020. https://doi.org/10.1002/cpt.1844.
Gowen BB, Sefing EJ, Westover JB, Smee DF, Hagloch J, Furuta Y, et al. Alterations in favipiravir (T-705) pharmacokinetics and biodistribution in a hamster model of viral hemorrhagic fever. Antiviral Res. 2015;121:132–7. https://doi.org/10.1016/j.antiviral.2015.07.003.
Mendenhall M, Russell A, Smee DF, Hall JO, Skirpstunas R, Furuta Y, et al. Effective oral favipiravir (t-705) therapy initiated after the onset of clinical disease in a model of arenavirus hemorrhagic fever. PLoS Negl Trop Dis. 2011;5:e1342. https://doi.org/10.1371/journal.pntd.0001342.
Reagan-Shaw S, Nihal M, Ahmad N. Dose translation from animal to human studies revisited. FASEB J. 2008;22:659–61. https://doi.org/10.1096/fj.07-9574LSF.
Chu CM, Cheng VC, Hung IF, Wong MM, Chan KH, Chan KS, et al. Role of lopinavir/ritonavir in the treatment of SARS: initial virological and clinical findings. Thorax. 2004;59(3):252–6. https://doi.org/10.1136/thorax.2003.012658.
Falzarano D, De Wit E, Rasmussen AL, Feldmann F, Okumura A, Scott DP, et al. Treatment with interferon-α2b and ribavirin improves outcome in MERS-CoV–infected rhesus macaques. Nat Med. 2013;19:1313–7. https://doi.org/10.1038/nm.3362.
Graci JD, Cameron CE. Mechanisms of action of ribavirin against distinct viruses. Rev Med Virol. 2006;16:37–48. https://doi.org/10.1002/rmv.483.
Preston SL, Drusano GL, Glue P, Nash J, Gupta SK, McNamara P. Pharmacokinetics and absolute bioavailability of ribavirin in healthy volunteers as determined by stable-isotope methodology. Antimicrob Agents Chemother. 1999;43:2451–6.
Loustaud-Ratti V, Stanke-Labesque F, Marquet P, Gagnieu MC, Maynard M, Babany G, et al. Optimizing ribavirin dosage: a new challenge to improve treatment efficacy in genotype 1 hepatitis C patients. Gastroenterol Clin Biol. 2009;33:580–3. https://doi.org/10.1016/j.gcb.2009.04.009.
Molina-Cuadrado E, Mateo-Carrrasco H, Collado A, Casado MM. Anaemia predictors in patients with chronic hepatitis C treated with ribavirin and direct-acting antiviral agents. Eur J Hosp Pharm. 2018;25:132–7. https://doi.org/10.1136/ejhpharm-2017-001277.
Russmann S, Grattagliano I, Portincasa P, Palmieri VO, Palasciano G. Ribavirin-induced anemia: mechanisms, risk factors and related targets for future research. Curr Med Chem. 2006;13:3351–7. https://doi.org/10.2174/092986706778773059.
Naik GS, Tyagi MG. A pharmacological profile of ribavirin and monitoring of its plasma concentration in chronic hepatitis C infection. J Clin Exp Hepatol. 2012;2:42–544. https://doi.org/10.1016/S0973-6883(12)60090-5.
Morello J, Rodríguez-Novoa S, Jiménez-Nácher I, Soriano V. Usefulness of monitoring ribavirin plasma concentrations to improve treatment response in patients with chronic hepatitis C. J Antimicrob Chemother. 2008;62:1174–80. https://doi.org/10.1093/jac/dkn421.
Chen YK, Huang YQ, Tang SQ, Xu XL, Zeng YM, He XQ et al. Comparative effectiveness and safety of ribavirin plus interferon-alpha, lopinavir/ritonavir plus interferon-alpha and ribavirin plus lopinavir/ritonavir plus interferon-alpha in patients with mild to moderate novel coronavirus pneumonia: results of a randomized, open-labeled prospective study (4/14/2020). SSRN: https://ssrn.com/abstract=3576905 or https://doi.org/10.2139/ssrn.3576905.
Wu C, Liu Y, Yang Y, Zhang P, Zhong W, Wang Y, et al. Analysis of therapeutic targets for SARS-CoV-2 and discovery of potential drugs by computational methods. Version 2. Acta Pharm Sin B. 2020;10:766–88. https://doi.org/10.1016/j.apsb.2020.02.008.
Chen YW, Yiu CB, Wong KY. Prediction of the SARS-CoV-2 (2019-nCoV) 3C-like protease (3CL pro) structure: virtual screening reveals velpatasvir, ledipasvir, and other drug repurposing candidates. F1000Res. 2020;9:129. doi: 10.12688/f1000research.22457.2.
Savarino A. Expanding the frontiers of existing antiviral drugs: possible effects of HIV-1 protease inhibitors against SARS and avian influenza. J Clin Virol. 2005;34:170–8. https://doi.org/10.1016/j.jcv.2005.03.005.
Ngo ST, Quynh Anh Pham N, Le Thi L, Pham DH, Vu VV. Computational determination of potential inhibitors of SARS-CoV-2 main protease. J Chem Inf Model. 2020. https://doi.org/10.1021/acs.jcim.0c00491.
Martinez MA. Compounds with therapeutic potential against novel respiratory 2019 coronavirus. Antimicrob Agents Chemother. 2020;64:e00399–e420. https://doi.org/10.1128/AAC.00399-20.
Chandwani A, Shuter J. Lopinavir/ritonavir in the treatment of HIV-1 infection: a review. Ther Clin Risk Manag. 2008;4:1023–33. https://doi.org/10.2147/tcrm.s3285.
Ortega JT, Serrano ML, Pujol FH, Rangel HR. Unrevealing sequence and structural features of novel coronavirus using in silico approaches: the main protease as molecular target. EXCLI J. 2020;19:400–9. https://doi.org/10.17179/excli2020-1189.
Lim J, Jeon S, Shin HY, Kim MJ, Seong YM, LeeWJ, et al. Case of the index patient who caused tertiary transmission of COVID-19 infection in Korea: the application of lopinavir/ritonavir for the treatmentof COVID-19 infected pneumonia monitored by quantitative RT-PCR. J Korean Med Sci. 2020;35:e79. doi: 10.3346/jkms.2020.35.e79.
Xu K, Cai H, Shen Y, Ni Q, Chen Y, Hu S, et al. Management of corona virus disease-19 (COVID-19): the Zhejiang experience. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2020 21;49:0.
Han W, Quan B, Guo Y, Zhang J, Lu Y, Feng G, et al. The course of clinical diagnosis and treatment of a case infected with coronavirus disease 2019. J Med Virol. 2020;92:461–3. https://doi.org/10.1002/jmv.25711.
Qiu H, Wu J, Hong L, Luo Y, Song Q, Chen D. Clinical and epidemiological features of 36 children with coronavirus disease 2019 (COVID-19) in Zhejiang, China: an observational cohort study. Lancet Infect Dis. 2020;20:689–96. https://doi.org/10.1016/S1473-3099(20)30198-5.
Ton AT, Gentile F, Hsing M, Ban F, Cherkasov A. Rapid identification of potential inhibitors of SARS-CoV-2 main protease by deep docking of 1.3 billion compounds. Mol Inform. 2020. https://doi.org/10.1002/minf.202000028.
Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, et al. A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020;382(19):1787–99. https://doi.org/10.1056/NEJMoa2001282.
Wang Z, Chen X, Lu Y, Chen F, Zhang W. Clinical characteristics and therapeutic procedure for four cases with 2019 novel coronavirus pneumonia receiving combined Chinese and Western medicine treatment. Biosci Trends. 2020;14:64–8. https://doi.org/10.5582/bst.2020.01030.
Neuman MG, Schneider M, Nanau RM, Parry C. HIV-antiretroviral therapy induced liver, gastrointestinal, and pancreatic injury. Int J Hepatol. 2012;2012:760706. https://doi.org/10.1155/2012/760706.
Cvetkovic RS, Goa KL. Lopinavir/ritonavir: a review of its use in the management of HIV infection. Drugs. 2003;63:769–802. https://doi.org/10.2165/00003495-200363080-00004.
Bongiovanni M, Cicconi P, Landonio S, Meraviglia P, Testa L, Di Biagio A, et al. Predictive factors of lopinavir/ritonavir discontinuation for drug-related toxicity: results from a cohort of 416 multi-experienced HIV-infected individuals. Int J Antimicrob Agents. 2005;26:88–91. https://doi.org/10.1016/j.ijantimicag.2005.03.003.
Robbins BL, Capparelli EV, Chadwick EG, Yogev R, Serchuck L, Worrell C, et al. Pharmacokinetics of high-dose lopinavir-ritonavir with and without saquinavir or nonnucleoside reverse transcriptase inhibitors in human immunodeficiency virus-infected pediatric and adolescent patients previously treated with protease inhibitors. Antimicrob Agents Chemother. 2008;52:3276–83. https://doi.org/10.1128/AAC.00224-08.
Canta F, Marrone R, Bonora S, D'Avolio A, Sciandra M, Sinicco A, et al. Pharmacokinetics and hepatotoxicity of lopinavir/ritonavir in non-cirrhotic HIV and hepatitis C virus (HCV) co-infected patients. J Antimicrob Chemother. 2005;55:280–1. https://doi.org/10.1093/jac/dkh516.
Sulkowski MS, Thomas DL, Chaisson RE, Moore RD. Hepatotoxicity associated with antiretroviral therapy in adults infected with human immunodeficiency virus and the role of hepatitis C or B virus infection. JAMA. 2000;283:74–80. https://doi.org/10.1001/jama.283.1.74.
Tu Y, Poblete RJ, Freilich BD, Zarbin MA, Bhagat N. Retinal toxicity with ritonavir. Int J Ophthalmol. 2016;9:640–2. https://doi.org/10.18240/ijo.2016.04.29.
Pasquau Liaño J, Hidalgo TC. Chemical characteristics, mechanism of action and antiviral activity of darunavir. Enferm Infecc Microbiol Clin. 2008;26:3–9. https://doi.org/10.1016/s0213-005x(08)76547-9.
Khan SA, Zia K, Ashraf S, Uddin R, Ul-Haq Z. Identification of chymotrypsin-like protease inhibitors of SARS-CoV-2 via integrated computational approach. J Biomol Struct Dyn. 2020:1–10. doi: 10.1080/07391102.2020.1751298.
Rittweger M, Arastéh K. Clinical pharmacokinetics of darunavir. Clin Pharmacokinet. 2007;46:739–56. https://doi.org/10.2165/00003088-200746090-00002.
Back D, Sekar V, Hoetelmans RM. Darunavir: pharmacokinetics and drug interactions. Antivir Ther. 2008;13:1–13.
Triant VA, Siedner MJ. Darunavir and cardiovascular risk: evaluating the data to inform clinical care. J Infect Dis. 2020;221:498–500. https://doi.org/10.1093/infdis/jiz482.
Xie S, Chen XX, Qiao S, Li R, Sun Y, Xia S, et al. Identification of the RNA pseudoknot within the 3' end of the porcine reproductive and respiratory syndrome virus genome as a pathogen-associated molecular pattern to activate antiviral signaling via RIG-I and toll-like receptor 3. J Virol. 2018;92:e00097–e118. https://doi.org/10.1128/JVI.00097-18.
Li J, Lehmann C, Chen X, Romerio F, Lu W. Total chemical synthesis of human interferon alpha-2b via native chemical ligation. J Pept Sci. 2015;21:554–60. https://doi.org/10.1002/psc.2760.
Thomas H, Foster G, Platis D. Mechanisms of action of interferon and nucleoside analogues. J Hepatol. 2003;39:S93–S9898. https://doi.org/10.1016/s0168-8278(03)00207-1.
Wang HQ, Ma LL, Jiang JD, Pang R, Chen YJ, Li YH. Recombinant human interferon alpha 2b broad-spectrum anti-respiratory viruses pharmacodynamics study in vitro. Yao Xue Xue Bao. 2014;49(11):1547–53.
Yu DX, Chen Q, Zhang LL, Liu Y, Yu ZA, Li ZF, et al. A field trial of recombinant human interferon alpha-2b for nasal spray to prevent SARS and other respiratory viral infections. Zhonghua Shi Yan He Lin Chuang Bing Du Xue Za Zhi. 2005;19:216–9.
Falzarano D, De Wit E, Martellaro C, Callison J, Munster VJ, Feldmann H. Inhibition of novel β coronavirus replication by a combination of interferon-α2b and ribavirin. Sci Rep. 2013;3:1686. https://doi.org/10.1038/srep01686.
Arabi YM, Shalhoub S, Mandourah Y, Al-Hameed F, Al-Omari A, Al Qasim E, et al. Ribavirin and interferon therapy for critically ill patients with middle east respiratory syndrome: a multicenter observational study. Clin Infect Dis. 2020;70(9):1837–44. https://doi.org/10.1093/cid/ciz544.
Lu H. Drug treatment options for the 2019-new coronavirus (2019-nCoV). Biosci Trends. 2020;14:69–71. https://doi.org/10.5582/bst.2020.01020.
Shen KL, Yang YH. Diagnosis and treatment of, novel coronavirus infection in children: a pressing issue. World J Pediatr. 2019;2020:1–3. https://doi.org/10.1007/s12519-020-00344-6.
Foster GR. Review article: pegylated interferons: chemical and clinical differences. Aliment Pharmacol Ther. 2004;20(8):825–30. https://doi.org/10.1111/j.1365-2036.2004.02170.x.96.
Russo MW, Fried MW. Side effects of therapy for chronic hepatitis C. Gastroenterology. 2003;124(6):1711–9. https://doi.org/10.1016/s0016-5085(03)00394-9.
Zheng L, Li MP, Gou ZP, Wang Y, Xu N, Cai YM, et al. A pharmacokinetic and pharmacodynamic comparison of a novel pegylated recombinant consensus interferon-α variant with peginterferon-α-2a in healthy subjects. Br J Clin Pharmacol. 2015;79(4):650–9. https://doi.org/10.1111/bcp.12528.
Mainali NR, Bhatt VR, Kedia S, Krishnamurthy J, Wake LM, Akhtari M. Reversible bone marrow aplasia induced by pegylated interferon-α-2a therapy in a patient with primary myelofibrosis. J Oncol Pharm Pract. 2014;20(5):386–92. https://doi.org/10.1177/1078155213504444.
Raison CL, Demetrashvili M, Capuron L, Miller AH. Neuropsychiatric adverse effects of interferon-alpha: recognition and management. CNS Drugs. 2005;19:105–23. https://doi.org/10.2165/00023210-200519020-00002.
Arabi YM, Alothman A, Balkhy HH, Al-Dawood A, AlJohani S, Al Harbi S, et al. Treatment of Middle East Respiratory Syndrome with a combination of lopinavir-ritonavir and interferon-β1b (MIRACLE trial): study protocol for a randomized controlled trial. Trials. 2018;19:81. https://doi.org/10.1186/s13063-017-2427-0.
Chan JF, Yao Y, Yeung ML, Deng W, Bao L, Jia L, et al. Treatment with lopinavir/ritonavir or interferon-β1b improves outcome of MERS-CoV infection in a nonhuman primate model of common marmoset. J Infect Dis. 2015;212:1904–13. https://doi.org/10.1093/infdis/jiv392.
Hung IF, Lung KC, Tso EY, Liu R, Chung TW, Chu MY, et al. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet. 2020;395:1695–704. https://doi.org/10.1016/S0140-6736(20)31042-4.
Belouzard S, Millet JK, Licitra BN, Whittaker GR. Mechanisms of coronavirus cell entry mediated by the viral spike protein. Viruses. 2012;4:1011–33. https://doi.org/10.3390/v4061011.
Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–280.e8. https://doi.org/10.1016/j.cell.2020.02.052.
Devaux CA, Rolain JM, Colson P, Raoult D. New insights on the antiviral effects of chloroquine against coronavirus: what to expect for COVID-19? Int J Antimicrob Agents. 2020;55:105938. https://doi.org/10.1016/j.ijantimicag.2020.105938.
Al-Bari MAA. Targeting endosomal acidification by chloroquine analogs as a promising strategy for the treatment of emerging viral diseases. Pharmacol Res Perspect. 2017;5(1):e00293. https://doi.org/10.1002/prp2.293.
Vincent MJ, Bergeron E, Benjannet S, Erickson BR, Rollin PE, Ksiazek TG, et al. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol J. 2005;2:69. https://doi.org/10.1186/1743-422X-2-69.
Liu J, Cao R, Xu M, Wang X, Zhang H, Hu H, et al. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov. 2020;6:16. https://doi.org/10.1038/s41421-020-0156-0.
Mackenzie AH. Dose refinements in long-term therapy of rheumatoid arthritis with antimalarials. Am J Med. 1983;75:40–5. https://doi.org/10.1016/0002-9343(83)91269-x.
Laaksonen AL, Koskiahde V, Juva K. Dosage of antimalarial drugs for children with juvenile rheumatoid arthritis and systemic lupus erythematosus. A clinical study with determination of serum concentrations of chloroquine and hydroxychloroquine. Scand J Rheumatol. 1974;3:103–8. https://doi.org/10.3109/03009747409115809.
Weniger H, World Health Organization. Review of side effects and toxicity of chloroquine (No. WHO/MAL/79.906). 1979. Geneva: World health Organization.
McChesney EW. Animal toxicity and pharmacokinetics of hydroxychloroquine sulfate. Am J Med. 1983;75:11–8. https://doi.org/10.1016/0002-9343(83)91265-2.
Popert AJ. Chloroquine: a review. Rheumatol Rehabil. 1976;15:235–8. https://doi.org/10.1093/rheumatology/15.3.235.
Alving AS, Eichelberger L, Craige B, Jones R, Whorton CM, Pullman TN. Studies on the chronic toxicity of chloroquine. J Clin Invest. 1948;27:60–5. https://doi.org/10.1172/JCI101974.
Looareesuwan S, White NJ, Chanthavanich P, Edwards G, Nicholl DD, Bunch C, et al. Cardiovascular toxicity and distribution kinetics of intravenous chloroquine. Br J Pharmacol. 1986;22:31–6. https://doi.org/10.1111/j.1365-2125.1986.tb02876.x.
Stokkermans TJ, Trichonas G. Chloroquine and hydroxychloroquine toxicity. [Updated 2019 Jun 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. https://www.ncbi.nlm.nih.gov/books/NBK537086/.
Yusuf IH, Sharma S, Luqmani R, Downes SM. Hydroxychloroquine retinopathy. Eye (Lond). 2017;31:828–45. https://doi.org/10.1038/eye.2016.298.
Falcone PM, Paolini L, Lou PL. Hydroxychloroquine toxicity despite normal dose therapy. Ann Ophthalmol. 1993;25:385–8.
Furst DE. Pharmacokinetics of hydroxychloroquine and chloroquine during treatment of rheumatic diseases. Lupus. 1996;5:11–5. https://doi.org/10.1177/0961203396005001041.
Ducharme J, Farinotti R. Clinical pharmacokinetics and metabolism of chloroquine. Focus on recent advancements. Clin Pharmacokinet. 1996;31(4):257–74. https://doi.org/10.2165/00003088-199631040-00003.
Lim HS, Im JS, Cho JY, Bae KS, Klein TA, Yeom JS, et al. Pharmacokinetics of hydroxychloroquine and its clinical implications in chemoprophylaxis against malaria caused by Plasmodium vivax. Antimicrob Agents Chemother. 2009;53:1468–75. https://doi.org/10.1128/AAC.00339-08.
Browning DJ. Pharmacology of Chloroquine and Hydroxychloroquine. Hydroxychloroquine Chloroquine Retinopathy. 2014;4:35–633. https://doi.org/10.1007/978-1-4939-0597-3_2.
Melles RB, Marmor MF. Pericentral retinopathy and racial differences in hydroxychloroquine toxicity. Ophthalmology. 2015;122:110–6. https://doi.org/10.1016/j.ophtha.2014.07.018.
Wang X, Cao R, Zhang H, Liu J, Xu M, Hu H, et al. The anti-influenza virus drug, arbidol is an efficient inhibitor of SARS-CoV-2 in vitro. Version 2. Cell Discov. 2020;6:28. https://doi.org/10.1038/s41421-020-0169-8.
Leneva IA, Fediakina IT, Gus'kova TA, Glushkov RG. Sensitivity of various influenza virus strains to arbidol. Influence of arbidol combination with different antiviral drugs on reproduction of influenza virus A. Ter Arkh. 2005;77:84–8.
Shi L, Xiong H, He J, Deng H, Li Q, Zhong Q, et al. Antiviral activity of arbidol against influenza A virus, respiratory syncytial virus, rhinovirus, coxsackie virus and adenovirus in vitro and in vivo. Arch Virol. 2007;152:1447–555. https://doi.org/10.1007/s00705-007-0974-5.
Blaising J, Polyak SJ, Pécheur EI. Arbidol as a broad-spectrum antiviral: An update. Antiviral Res. 2014;107:84–94. https://doi.org/10.1016/j.antiviral.2014.04.006.
Khamitov RA, Loginova S, Shchukina VN, Borisevich SV, Maksimov VA, Shuster AM. Antiviral activity of arbidol and its derivatives against the pathogen of severe acute respiratory syndrome in the cell cultures. Vopr Virusol. 2008;53(4):9–13.
Barnard DL, Kumaki Y. Recent developments in anti-severe acute respiratory syndrome coronavirus chemotherapy. Fut Virol. 2011;6(5):615–31. https://doi.org/10.2217/fvl.11.33.
Wang Z, Yang B, Li Q, Wen L, Zhang R. Clinical features of 69 cases with coronavirus disease, in Wuhan, China. Clin Infect Dis. 2019;2020:272. https://doi.org/10.1093/cid/ciaa272.
Rosa SGV, Santos WC. Clinical trials on drug repositioning for COVID-19 treatment. Rev Panam Salud Publica. 2020;44:e40. https://doi.org/10.26633/RPSP.2020.40.
Deng L, Li C, Zeng Q, Liu X, Li X, Zhang H, et al. Arbidol combined with LPV/r versus LPV/r alone against Corona Virus Disease 2019: a retrospective cohort study. J Infect. 2020;81:e1–e5. https://doi.org/10.1016/j.jinf.2020.03.002.
Titova ON, Petrova MA, Shklyarevich NA, Kuzubova NA, Aleksandrov AL, Kovaleva LF, et al. Efficacy of Arbidol in the prevention of virus-induced exacerbations of bronchial asthma and chronic obstructive pulmonary disease. Ter Arkh. 2018;90(8):48–52. https://doi.org/10.26442/terarkh201890848-52.
Li Y, Xie Z, Lin W, Cai W, Wen C, Guan Y, et al. An exploratory randomized, controlled study on the efficacy and safety of lopinavir/ritonavir or arbidol treating adult patients hospitalized with mild/moderate COVID-19 (ELACOI). MedRxiv. https://www.medrxiv.org. doi: https://doi.org/10.1101/2020.03.19.20038984.
Deng P, Zhong D, Yu K, Zhang Y, Wang T, Chen X. Pharmacokinetics, metabolism, and excretion of the antiviral drug arbidol in humans. Antimicrob Agents Chemother. 2013;57:1743–55. https://doi.org/10.1128/AAC.02282-12.
Fedson DS, Opal SM, Rordam OM. Hiding in plain sight: an approach to treating patients with severe COVID-19 infection. mBio. 2020;11:e00398-20. https://doi.org/10.1128/mBio.00398–20.
Wösten-van Asperen RM, Bos AP, Bem RA, Dierdorp BS, Dekker T, van Goor H, et al. Imbalance between pulmonary angiotensin- converting enzyme and angiotensin-converting enzyme 2 activity in acute respiratory distress syndrome. Pediatr Crit Care Med. 2013;2013(14):e438–e441441. https://doi.org/10.1097/PCC.0b013e3182a55735.
Vaduganathan M, Vardeny O, Michel T, McMurray JJV, Pfeffer MA, Solomon SD. Renin-angiotensin-aldosterone system inhibitors in patients with Covid-19. N Engl J Med. 2020;382:1653–9. https://doi.org/10.1056/NEJMsr2005760.
Wan Y, Shang J, Graham R, Baric RS, Li F. Receptor recognition by novel coronavirus fromWuhan: An analysis based on decade-long structural studies of SARS. J Virol. 2020;94:e00127–e220. https://doi.org/10.1128/JVI.00127-20.
Steinberg BE, Goldenberg NM, Lee WL. Do viral infections mimic bacterial sepsis? The role of microvascular permeability: a review of mechanisms and methods. Antiviral Res. 2012;93:2–15. https://doi.org/10.1016/j.antiviral.2011.10.019.
Marin GH. Facts and reflections on COVID-19 and anti-hypertensives drugs. Drug Discov Ther. 2020;14:105–6. https://doi.org/10.5582/ddt.2020.01017.
Sun ML, Yang JM, Sun YP, Su GH. Inhibitors of RAS might be a good choice for the therapy of COVID-19 pneumonia. Zhonghua Jie He He Hu Xi Za Zhi. 2020;43:219–22. https://doi.org/10.3760/cma.j.issn.1001-0939.2020.03.016.
Meng J, Xiao G, Zhang J, He X, Ou M, Bi J, et al. Renin-angiotensin system inhibitors improve the clinical outcomes of COVID-19 patients with hypertension. Emerg Microbes Infect. 2020;9:757–60. https://doi.org/10.1080/22221751.2020.1746200.
Peng YD, Meng K, Guan HQ, Leng L, Zhu RR, Wang BY, et al. Clinical characteristics and outcomes of 112 cardiovascular disease patients infected by 2019-nCoV. Zhonghua Xin Xue Guan Bing Za Zhi. 2020;48:E004. https://doi.org/10.3760/cma.j.cn112148-20200220-00105.
D'Ardes D, Boccatonda A, Rossi I, Guagnano MT, Santilli F, Cipollone F, et al. COVID-19 and RAS: unravelling an unclear relationship. Int J Mol Sci. 2020;21:3003. https://doi.org/10.3390/ijms21083003.
Peyriere H, Eiden C, Macia JC, Reynes J. Antihypertensive drugs in patients treated with antiretrovirals. Ann Pharmacother. 2012;46:703–9. https://doi.org/10.1345/aph.1Q546.
Foy M, Sperati CJ, Lucas GM, Estrella MM. Drug interactions and antiretroviral drug monitoring. Curr HIV/AIDS Rep. 2014;11:212–22. https://doi.org/10.1007/s11904-014-0212-1.
Barreras A, Gurk-Turner C. Angiotensin II receptor blockers. Proc (Bayl Univ Med Cent). 2003;16:123–6. https://doi.org/10.1080/08998280.2003.11927893.
Israili ZH. Clinical pharmacokinetics of angiotensin II (AT1) receptor blockers in hypertension. J Hum Hypertens. 2000;14:S73–86. https://doi.org/10.1038/sj.jhh.1000991.
Domenic A. Sica, Pharmacology and clinical efficacy of angiotensin receptor blockers. Am J Hypertens. 2001;14:242–7. https://doi.org/10.1016/S0895-7061(01)02134-3.
Phadke M, Saunik S. COVID-19 treatment by repurposing drugs until the vaccine is in sight. Drug Dev Res. 2020. https://doi.org/10.1002/ddr.21666.10.1002/ddr.21666.
Tikoo K, Patel G, Kumar S, Karpe PA, Sanghavi M, Malek V, et al. Tissue specific up regulation of ACE2 in rabbit model of atherosclerosis by atorvastatin: role of epigenetic histone modifications. Biochem Pharmacol. 2015;93:343–51. https://doi.org/10.1016/j.bcp.2014.11.013.
Ferrario CM. ACE 2: more of Ang 1–7 or less Ang II? Curr Opin Nephrol Hypertens. 2011;20:1–6. https://doi.org/10.1097/MNH.0b013e3283406f57.
Fedson DS (2016) Treating the host response to emerging virus diseases:lessons learned from sepsis, pneumonia, influenza and Ebola. Ann Transl Med. 4:421. https://doi.org/10.21037/atm.2016.11.03.
Wösten-van Asperen RM, Lutter R, Specht PA, Moll GN, van Woensel JB, van der Loos CM, et al. Acute respiratory distress syndrome leads to reduced ratio of ACE/ACE2 activities and is prevented by angiotensin-(1–7) or an angiotensin II receptor antagonist. J Pathol. 2011;225:618–27. https://doi.org/10.1002/path.2987.
Yuan S (2015) Statins may decrease the fatality rate of Middle East respiratory syndrome infection. mBio. 6:e01120-15. doi: 10.1128/mBio.01120-15.
Totura AL, Baric RS. Reply to “statins may decrease the fatality rate of MERS infection”. mBio. 2015;6:e01303-15. doi: 10.1128/mBio.01303-15.
Schachter M. Chemical, pharmacokinetic and pharmacodynamic properties of statins: an update. Fundam Clin Pharmacol. 2005;19:117–25. https://doi.org/10.1111/j.1472-8206.2004.00299.x.
Bełtowski J, Wójcicka G, Jamroz-Wiśniewska A. Adverse effects of statins-mechanisms and consequences. Curr Drug Saf. 2009;4:209–28. https://doi.org/10.2174/157488609789006949.
Mancini GB, Baker S, Bergeron J, Fitchett D, Frohlich J, Genest J, et al. Diagnosis, prevention, and management of statin adverse effects and intolerance: canadian consensus working group update. Can J Cardiol. 2016;32:S35–65. https://doi.org/10.1016/j.cjca.2016.01.003.
Burger D, Back D, Buggisch P, Buti M, Craxí A, Foster G, et al. Clinical management of drug-drug interactions in HCV therapy: challenges and solutions. J Hepatol. 2013;58:792–800. https://doi.org/10.1016/j.jhep.2012.10.027.
Chauvin B, Drouot S, Barrail-Tran A, Taburet AM. Drug-drug interactions between HMG-CoA reductase inhibitors (statins) and antiviral protease inhibitors. Clin Pharmacokinet. 2013;52:815–31. https://doi.org/10.1007/s40262-013-0075-4.
Sheppard M, Laskou F, Stapleton PP, Hadavi S, Dasgupta B. Tocilizumab (Actemra). Hum Vaccin Immunother. 2017;13:1972–88. https://doi.org/10.1080/21645515.2017.1316909.
Bersanelli M. Controversies about COVID-19 and anticancer treatment with immune checkpoint inhibitors. Immunotherapy. 2020;12:269–73. https://doi.org/10.2217/imt-2020-0067.
Zhang X, Song K, Tong F, Fei M, Guo H, Lu Z, et al. First case of COVID-19 in a patient with multiple myeloma successfully treated with tocilizumab. Blood Adv. 2020;4:1307–10. https://doi.org/10.1182/bloodadvances.2020001907.
Ferrey AJ, Choi G, Hanna RM, Chang Y, Tantisattamo E, Ivaturi K, et al. Case of novel coronavirus disease 19 in a chronic hemodialysis patient presenting with gastroenteritis and developing severe pulmonary disease. Am J Nephrol. 2020;28:1–6. https://doi.org/10.1159/000507417.
Mihai C, Dobrota R, Schröder M, Garaiman A, Jordan S, Becker MO, et al. COVID-19 in a patient with systemic sclerosis treated with tocilizumab for SSc-ILD. Ann Rheum Dis. 2020;79:668–9. https://doi.org/10.1136/annrheumdis-2020-217442.
Michot JM, Albiges L, Chaput N, Saada V, Pommeret F, Griscelli F, et al. Tocilizumab, an anti-IL6 receptor antibody, to treat Covid-19-related respiratory failure: a case report. Ann Oncol. 2020;31:961–4. https://doi.org/10.1016/j.annonc.2020.03.300.
Xu X, Han M, Li T, Sun W, Wang D, Fu B, et al. Effective treatment of severe COVID-19 patients with tocilizumab. Proc Natl Acad Sci USA. 2020;117:10970–5. https://doi.org/10.1073/pnas.2005615117.
Toniati P, Piva S, Cattalini M, Garrafa E, Regola F, Castelli F, et al. Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: a single center study of 100 patients in Brescia, Italy. Autoimmun Rev. 2020;19:102568. https://doi.org/10.1016/j.autrev.2020.102568.
Luo P, Liu Y, Qiu L, Liu X, Liu D, Li J. Tocilizumab treatment in COVID-19: a single center experience. J Med Virol. 2020;92:814–8. https://doi.org/10.1002/jmv.25801.
Abdallah H, Hsu JC, Lu P, Fettner S, Zhang X, Douglass W, et al. Pharmacokinetic and pharmacodynamic analysis of subcutaneous tocilizumab in patients with rheumatoid arthritis from 2 randomized, controlled trials: SUMMACTA and BREVACTA. J Clin Pharmacol. 2017;57:459–68. https://doi.org/10.1002/jcph.826.
Jones G, Ding C. Tocilizumab: a review of its safety and efficacy in rheumatoid arthritis. Clin Med Insights Arthritis Musculoskelet Disord. 2010;3:81–9. https://doi.org/10.4137/CMAMD.S4864.
Mahamid M, Mader R, Safadi R. Hepatotoxicity of tocilizumab and anakinra in rheumatoid arthritis: management decisions. Clin Pharmacol. 2011;3:39–433. https://doi.org/10.2147/CPAA.S24004.
Nishimoto N, Miyasaka N, Yamamoto K, Kawai S, Takeuchi T, Azuma J. Long-term safety and efficacy of tocilizumab, an anti-IL-6 receptor monoclonal antibody, in monotherapy, in patients with rheumatoid arthritis (the STREAM study): evidence of safety and efficacy in a 5-year extension study. Ann Rheum Dis. 2009;68:1580–4. https://doi.org/10.1136/ard.2008.092866.
Rosenthal PJ (2017) Antiprotozoal drugs. In: Katzung BG (eds) Basic & clinical pharmacology, 14e. McGraw-Hill Education; New York.
Kelleni MT. Nitazoxanide/azithromycin combination for COVID-19: a suggested new protocol for early management. Pharmacol Res. 2020;157:104874. https://doi.org/10.1016/j.phrs.2020.104874.
Rossignol JF. Nitazoxanide, a new drug candidate for the treatment of Middle East respiratory syndrome coronavirus. J Infect Public Health. 2016;9:227–30. https://doi.org/10.1016/j.jiph.2016.04.001.
Şimşek Yavuz S, Ünal S. Antiviral treatment of COVID-19. Turk J Med Sci. 2020;50:611–9. https://doi.org/10.3906/sag-2004-145.
Caly L, Druce JD, Catton MG, Jans DA, Wagstaff KM. The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro. Antiviral Res. 2020;3:104787. https://doi.org/10.1016/j.antiviral.2020.104787.
Fan HH, Wang LQ, Liu WL, An XP, Liu ZD, He XQ, et al. Repurposing of clinically approved drugs for treatment of coronavirus disease 2019 in a 2019-novel coronavirus (2019-nCoV) related coronavirus model. Chin Med J (Engl). 2020;133:1051–6. https://doi.org/10.1097/CM9.0000000000000797.
Rothan HA, Stone S, Natekar J, Kumari P, Arora K, Kumar M. The FDA- approved gold drug auranofin inhibits novel coronavirus (SARSCOV-2) replication and attenuates inflammation in human cells. Virology. 2020;547:7–11. https://doi.org/10.1016/j.virol.2020.05.002.
Fung TS, Liu DX. Coronavirus infection, ER stress, apoptosis and innate immunity. Front Microbiol. 2014;5:296. https://doi.org/10.3389/fmicb.2014.00296.
Siu KL, Chan CP, Kok KH, Woo PC, Jin DY. Comparative analysis of the activation of unfolded protein response by spike proteins of severe acute respiratory syndrome coronavirus and human coronavirus HKU1. Cell Biosci. 2014;4(1):3. https://doi.org/10.1186/2045-3701-4-3.
Sung SC, Chao CY, Jeng KS, Yang JY, Lai MM. The 8ab protein of SARS-CoV is a luminal ER membrane-associated protein and induces the activation of ATF6. Virology. 2009;387:402–13. https://doi.org/10.1016/j.virol.2009.02.021.
Rothan HA, Kumar M. Role of endoplasmic reticulum-associated proteins in flavivirus replication and assembly complexes. Pathogens. 2019;8(3):148. https://doi.org/10.3390/pathogens8030148.
Mehta P, Ciurtin C, Scully M, Levi M, Chambers RC. JAK inhibitors in COVID-19: need for vigilance regarding increased inherent thrombotic risk. Eur Respir J. 2020:2001919. doi: 10.1183/13993003.01919-2020.
La Rosée F, Bremer HC, Gehrke I, Kehr A, Hochhaus A, Birndt S, et al. The Janus kinase 1/2 inhibitor ruxolitinib in COVID-19 with severe systemic hyperinflammation. Leukemia. 2020;34:1805–15. https://doi.org/10.1038/s41375-020-0891-0.
Cantini F, Niccoli L, Matarrese D, Nicastri E, Stobbione P, Goletti D. Baricitinib therapy in COVID-19: a pilot study on safety and clinical impact. J Infect. 2020;81(2):318–56. https://doi.org/10.1016/j.jinf.2020.04.017.
Bronte V, Ugel S, Tinazzi E, Vella A, Sanctis FDe, Canè S, et al. Baricitinib restrains the immune dysregulation in COVID-19 patients. Preprint at medRxiv, https://doi.org/10.1101/2020.06.26.20135319.
RECOVERY Collaborative Group, Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, et al. Dexamethasone in hospitalized patients with Covid-19 - preliminary report. N Engl J Med. 2020: NEJMoa2021436 doi: 10.1056/NEJMoa2021436.
Johnson RM, Vinetz JM. Dexamethasone in the management of covid -19. BMJ 2020; 370. https://doi.org/10.1136/bmj.m2648.
Theoharides TC, Conti P. Dexamethasone for COVID-19? Not so fast. J Biol Regul Homeost Agents. 2020;34(3). https://doi.org/10.23812/20-EDITORIAL_1–5.
Brotherton H, Usuf E, Nadjm B, Forrest K, Bojang K, Samateh AL, et al. Dexamethasone for COVID-19: data needed from randomised clinical trials in Africa. Lancet Glob Health. 2020. https://doi.org/10.1016/S2214-109X(20)30318-1.
Li Y, Zhang J, Wang N, Li H, Shi Y, Guo G, et al. Therapeutic drugs targeting 2019-nCoV main protease by high-throughput screening. BioRxiv. Preprint at https://www.biorxiv.org. https://doi.org/10.1101/2020.01.28.922922.
Elfiky AA. Ribavirin, remdesivir, sofosbuvir, galidesivir, and tenofovir against SARS-CoV-2 RNA dependent RNA polymerase (RdRp): a molecular docking study. Life Sci. 2020;25:117592. https://doi.org/10.1016/j.lfs.2020.117592.
Balasubramaniam M, Reis RS. Computational target-based drug repurposing of elbasvir, an antiviral drug predicted to bind multiple SARS-CoV-2 proteins. ChemRxiv. https://doi.org/10.26434/chemrxiv.12084822.v2.
Sargiacomo C, Sotgia F, Lisanti MP. COVID-19 and chronological aging: senolytics and other anti-aging drugs for the treatment or prevention of corona virus infection? Aging (Albany NY). 2020;12:6511–7. https://doi.org/10.18632/aging.103001.
Elfiky AA. SARS-CoV-2 RNA dependent RNA polymerase (RdRp) targeting: an in silico perspective. J Biomol Struct Dyn. 2020;1–9. https://doi.org/10.1080/07391102.2020.1761882.
Cai Q, Yang M, Liu D, et al. Experimental treatment with favipiravir for COVID-19: an open-label control study. Engineering (Beijing). 2020. https://doi.org/10.1016/j.eng.2020.03.007.10.1016/j.eng.2020.03.007.
Chen C, Zhang Y, Huang J, Yin P, Cheng Z, Wu J et al. Favipiravir versus arbidol for COVID-19: a randomized clinical trial. medRxiv. 2020:2020.03.17.20037432.
Chen ZR, Zhou Y, Liu J, Peng HW, Zhou J, Zhong HL, et al. Pharmacotherapics advice in guidelines for COVID-19. Front Pharmacol. 2020;11:950. https://doi.org/10.3389/fphar.2020.00950.
Wang J. Fast identification of possible drug treatment of coronavirus disease-19 (COVID-19) through computational drug repurposing study. J Chem Inf Model. 2020;60:3277–86. https://doi.org/10.1021/acs.jcim.0c00179.
Ruan Z, Liu C, Guo Y, He Z, Huang X, Jia X, et al. SARS-CoV-2 and SARS-CoV: virtual screening of potential inhibitors targeting RNA-dependent RNA polymerase activity (NSP12). J Med Virol. 2020. https://doi.org/10.1002/jmv.26222.
Alméciga-Díaz CJ, Pimentel-Vera LN, Caro A, Mosquera A, Moreno CAC, Rojas JPM, et al. Virtual screening of potential inhibitors for SARS-CoV-2 main protease, (2020). https://www.preprints.org/manuscript/202004.0146/v1.
Ohashi H, Watashi K, Saso W, Shionoya K, Iwanami S, Hirokawa T, et al. Multidrug treatment with nelfinavir and cepharanthine against COVID-19. bioRxiv 2020.04.14.039925; https://doi.org/10.1101/2020.04.14.039925.
Sayad B, Sobhani M, Khodarahmi R. Sofosbuvir as repurposed antiviral drug against COVID-19: Why were we convinced to evaluate the drug in a registered/approved clinical trial? Arch Med Res. 2020;S0188–4409(20):30551–8. https://doi.org/10.1016/j.arcmed.2020.04.018.
Chien M, Anderson TK, Jockusch S, Tao C, Kumar S, Li X, et al. Nucleotide Analogues as Inhibitors of SARS-CoV-2 Polymerase. Preprint. bioRxiv. 2020;2020.03.18.997585. Published 2020 Mar 20. doi:10.1101/2020.03.18.997585.
Choy KT, Wong AY, Kaewpreedee P, Sia SF, Chen D, Hui KPY, et al. Remdesivir, lopinavir, emetine, and homoharringtonine inhibit SARS-CoV-2 replication in vitro. Antiviral Res. 2020;178:104786. https://doi.org/10.1016/j.antiviral.2020.104786.
Liu X, Wang XJ. Potential inhibitors against 2019-nCoV coronavirus M protease from clinically approved medicines. J Genet Genomics. 2020;47(2):119–21. https://doi.org/10.1016/j.jgg.2020.02.001.
De Meyer S, Bojkova D, Cinatl J, Van Damme E, Buyck C, Van Loock M, et al. Lack of antiviral activity of darunavir against SARS-CoV-2. Int J Infect Dis. 2020;97:7–10. https://doi.org/10.1016/j.ijid.2020.05.085.
Vankadari N. Arbidol: A potential antiviral drug for the treatment of SARS-CoV-2 by blocking trimerization of the spike glycoprotein. Int J Antimicrob Agents. 2020;56(2):105998. https://doi.org/10.1016/j.ijantimicag.2020.105998.
Zhu Z, Lu Z, Xu T, Chen C, Yang G, Zha T, et al. Arbidol monotherapy is superior to lopinavir/ritonavir in treating COVID-19. J Infect. 2020;81:e21–e2323. https://doi.org/10.1016/j.jinf.2020.03.060.
Hoffmann M, Schroeder S, Kleine-Weber H, Müller MA, Drosten C, Pöhlmann S. Nafamostat mesylate blocks activation of SARS-CoV-2: new treatment option for COVID-19. Antimicrob Agents Chemother. 2020;64:e00754–e820. https://doi.org/10.1128/AAC.00754-20.
Jang S, Rhee JY. Three cases of treatment with nafamostat in elderly patients with COVID-19 pneumonia who need oxygen therapy. Int J Infect Dis. 2020;S1201–9712(20):30379–89. https://doi.org/10.1016/j.ijid.2020.05.072.
Yao X, Ye F, Zhang M, Cui C, Huang B, Niu P, et al. In Vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin Infect Dis. 2020;ciaa237. doi:10.1093/cid/ciaa237.
Encinar JA, Menendez JA. Potential drugs targeting early innate immune evasion of sars-coronavirus 2 via 2'-O-methylation of viral RNA. Viruses. 2020;12:525. https://doi.org/10.3390/v12050525.
Musarrat F, Chouljenko V, Dahal A, Nabi R, Chouljenko T, Jois SD, et al. The anti-HIV drug nelfinavir mesylate (Viracept) is a potent inhibitor of cell fusion caused by the SARSCoV-2 spike (S) glycoprotein warranting further evaluation as an antiviral against COVID-19 infections. J Med Virol. 2020. https://doi.org/10.1002/jmv.25985.10.1002/jmv.25985.
Khan RJ, Jha RK, Amera GM, Jain M, Singh E, Pathak A, et al. Targeting SARS-CoV-2: a systematic drug repurposing approach to identify promising inhibitors against 3C-like proteinase and 2'-O-ribose methyltransferase. J Biomol Struct Dyn. 2020;1–14. doi:10.1080/07391102.2020.1753577.
Poschet JF, Perkett EA, Timmins GS, Deretic V. Azithromycin and ciprofloxacin have a chloroquine-like effect on respiratory epithelial cells. bioRxiv. 2020;3(29). doi: 10.1101/2020.03.29.008631.
Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, Mailhe M, et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents. 2020;105949. https://doi.org/10.1016/j.ijantimicag.2020.105949.
Marinho EM, Batista de Andrade Neto J, Silva J, Rocha da Silva C, Cavalcanti BC, Marinho ES, et al. Virtual screening based on molecular docking of possible inhibitors of Covid-19 main protease. Microb Pathog. 2020;104365. doi: 10.1016/j.micpath.2020.104365.
Cao Y, Wei J, Zou L, Jiang T, Wang G, Chen L, et al. Ruxolitinib in treatment of severe coronavirus disease 2019 (COVID-19): a multicenter, single-blind, randomized controlled trial. J Allergy Clin Immunol. 2020;146(137–146):e3. https://doi.org/10.1016/j.jaci.2020.05.019.
Cantini F, Niccoli L, Nannini C, Matarrese D, Natale MED, Lotti P, et al. Beneficial impact of baricitinib in COVID-19 moderate pneumonia; multicentre study. J Infect. 2020;S0163–4453(20):30433–43. https://doi.org/10.1016/j.jinf.2020.06.052.
Khan SU, Htar TT. Deciphering the binding mechanism of dexamethasone against SARS-CoV-2 main protease: computational molecular modelling approach. Priprint at ChemRxiv. 23/06/2020. https://doi.org/10.26434/chemrxiv.12517535.v1.
Shah B, Modi P, Sagar SR. In silico studies on therapeutic agents for COVID-19: Drug repurposing approach. Life Sci. 2020;252:117652. https://doi.org/10.1016/j.lfs.2020.117652.
Khater S, Das G. Repurposing ivermectin to inhibit the activity of SARS CoV2 helicase: possible implications for COVID 19 therapeutics. OSF Preprints. 2020. https://doi.org/10.31219/osf.io/8dseq.
Anastasiou IA, Eleftheriadou I, Tentolouris A, Tsilingiris D, Tentolouris N. In vitro data of current therapies for SARS-CoV-2. Curr Med Chem. 2020. https://doi.org/10.2174/0929867327666200513075430.10.2174/0929867327666200513075430.
Calderón JM, Zerón HM, Padmanabhan S. Treatment with Hydroxychloroquine vs Hydroxychloroquine + Nitazoxanide in COVID-19 patients with risk factors for poor prognosis: a structured summary of a study protocol for a randomised controlled trial. Trials. 2020;21:504. https://doi.org/10.1186/s13063-020-04448-2.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Singh, T.U., Parida, S., Lingaraju, M.C. et al. Drug repurposing approach to fight COVID-19. Pharmacol. Rep 72, 1479–1508 (2020). https://doi.org/10.1007/s43440-020-00155-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43440-020-00155-6