Abstract
Purpose
Best Practice Guidelines (BPGs) were published one decade ago to decrease surgical site infection (SSI) in pediatric spinal deformity. Successful implementation has not been established. This study evaluated surgeon compliance with items on the BPG. We hypothesized that BPG authors and surgeons with more experience, higher caseload, and awareness of the BPG would have higher compliance.
Methods
We queried North American and European surgeons, authors and non-authors, and members of various spine study groups on adherence to BPGs using an anonymous survey. Mean compliance scores (MCSs) were developed by correlating Likert responses with MCSs (“None of the time” = no compliance = MCS 0, “Sometimes” = weak to moderate = MCS 1, “Most of the time” = high = MCS 2, “All the time” = perfect = MCS 3).
Results
Of the 142 respondents, 73.7% reported high or perfect compliance. Average compliance scores for all guidelines was 2.2 ± 0.4. There were significantly different compliance scores between North American and European surgeons (2.3 vs 1.8, p < 0.001), authors and non-authors (2.5 vs. 2.2, p = 0.023), and surgeons with and without knowledge of the BPGs (2.3 vs. 1.8, p < 0.001). There was a weak correlation between BPG awareness and compliance (r = 0.34, p < 0.001) and no correlation between years in practice (r = 0.0, p = 0.37) or yearly caseload (r = 0.2, p = 0.78) with compliance.
Conclusions
Compliance among our cohort of surgeons surveyed was high. North American surgeons, authors of the BPGs and those aware of the guidelines had increased compliance. Participation in a spine study group, years in practice, and yearly caseload were not associated with compliance.
Level of evidence
Level V—expert opinion.

Similar content being viewed by others
Data availability
Raw data were generated at Columbia University Medical Center, Department of Orthopaedics. Derived data supporting the findings of this study are available from the corresponding author AC-G on request.
References
Mcleod L, Flynn J, Erickson M, Miller N, Keren R, Dormans J (2015) Variation in 60-day Readmission for Surgical-site Infections (SSIs) and Reoperation Following Spinal Fusion Operations for Neuromuscular Scoliosis [Online]. Available: http://www.pedorthopaedics.com. Accessed 25 Sept 2022
Salsgiver E et al (2016) Surgical Site Infections following Spine Surgery for Non-idiopathic Scoliosis [Online]. Available: http://www.pedorthopaedics.com. Accessed 5 Oct 2022
Spina NT, Aleem IS, Nassr A, Lawrence BD (2018) Surgical site infections in spine surgery: preoperative prevention strategies to minimize risk. Global Spine J 8(4_suppl):31S-36S. https://doi.org/10.1177/2192568217752130
Matsumoto H et al (2017) Paper #45: development of a risk severity score (RSS) predicting surgical site infection in early onset scoliosis: identifying high-risk patients. Spine Deform 5(6):464–465. https://doi.org/10.1016/j.jspd.2017.09.048
Salsgiver E et al (2017) Surgical site infections following spine surgery for non-idiopathic scoliosis. J Pediatr Orthop 37(8):e476–e483. https://doi.org/10.1097/BPO.0000000000000727
Floccari LV, Milbrandt TA (2016) Surgical site infections after pediatric spine surgery. Orthop Clin N Am 47(2):387–394. https://doi.org/10.1016/j.ocl.2015.09.001
Meza BC, Talwar D, Flynn JM (2020) Measures to reduce end-of-case wound contamination: the impact of intra-wound vancomycin powder and betadine irrigation on surgical site infections in posterior spinal fusion. Spine Deform 8(1):45–50. https://doi.org/10.1007/s43390-020-00033-4
Hammoor B et al (2021) Surgical site infections in pediatric spinal surgery after implementation of a quality assurance program. Spine Deform 9(1):125–133. https://doi.org/10.1007/s43390-020-00192-4
Bachmann KR et al (2020) The variability in the management of acute surgical site infections: an opportunity for the development of a best practice guideline. Spine Deform 8(3):463–468. https://doi.org/10.1007/s43390-020-00079-4
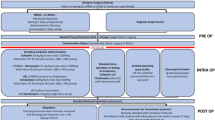
Glotzbecker MP, St Hilaire TA, Pawelek JB, Thompson GH, Vitale MG (2019) Best practice guidelines for surgical site infection prevention with surgical treatment of early onset scoliosis. J Pediatr Orthop 39(8):e602–e607. https://doi.org/10.1097/BPO.0000000000001079
Ryan SL, Sen A, Staggers K, Luerssen TG, Jea A (2014) A standardized protocol to reduce pediatric spine surgery infection: a quality improvement initiative: clinical article. J Neurosurg Pediatr 14(3):259–265. https://doi.org/10.3171/2014.5.PEDS1448
Glotzbecker MP, Garg S, Akbarnia BA, Vitale M, Hillaire TS, Joshi A (2014) Surgeon practices regarding infection prevention for growth friendly spinal procedures. J Child Orthop 8(3):245–250. https://doi.org/10.1007/s11832-014-0584-1
Vandenberg C et al (2018) Compliance with a comprehensive antibiotic protocol improves infection incidence in pediatric spine surgery. J Pediatr Orthop 38(5):287–292. https://doi.org/10.1097/BPO.0000000000000812
Mackenzie WGS et al (2018) Team approach: preventing surgical site infections in pediatric scoliosis surgery. JBJS Rev 6(2):E2. https://doi.org/10.2106/JBJS.RVW.16.00121
Solla F et al (2021) Prevention of surgical site infections in pediatric spines: a single-center experience. Child's Nerv Syst. https://doi.org/10.1007/500381-021-05095-x
Stephan SR, Illingworth KD, Gupta K, Andras LM, Skaggs DL (2021) Surgical site infection following neuromuscular posterior spinal fusion fell 72% after adopting the 2013 best practice guidelines. Spine (Phila Pa 1976) 46(17):1147–1153. https://doi.org/10.1097/BRS.0000000000004050
Ramo BA et al (2014) Surgical site infections after posterior spinal fusion for neuromuscular scoliosis: a thirty year experience at a single institution. J Bone Jt Surg Am Vol 96(24):2038–2048. https://doi.org/10.2106/JBJS.N.00277
Vitale MG et al (2013) Building consensus: development of a best practice guideline (BPG) for surgical site infection (SSI) prevention in high-risk pediatric spine surgery [Online]. Available: http://www.pedorthopaedics.com. Accessed 15 Nov 2022
Linstone HA, Turoff M, Helmer O (2002) The Delphi Method Techniques and Applications
Glotzbecker MP et al (2013) What’s the evidence? Systematic literature review of risk factors and preventive strategies for surgical site infection following pediatric spine surgery. J Pediatr Orthop 33(5):479–487
Jevsevar DS, Karlin LI (1993) The relationship between preoperative nutritional status and complications after an operation for scoliosis in patients who have cerebral palsy. J Bone Jt Surg 75(6):880–884. https://doi.org/10.2106/00004623-199306000-00008
Obana KK et al (2021) Pre-operative nutrition assessments do not improve outcomes in cerebral palsy patients undergoing varus derotational osteotomy. Med (United States) 100(47):E27776. https://doi.org/10.1097/MD.0000000000027776
Hatlen T, Song K, Shurtleff D, Duguay S (2010) Contributory factors to postoperative spinal fusion complications for children with myelomeningocele. Spine (Phila Pa 1976) 35(13):1294–1299. https://doi.org/10.1097/BRS.0b013e3181bf8efe
Shenoy ES, Giuriato MA, Song Z (2021) Prevalence, costs, and consequences of low-value preprocedural urinalyses in the US. JAMA Intern Med. https://doi.org/10.1001/jamainternmed.2021.4075
Piantoni L et al (2020) Antibiotic prophylaxis in high-risk pediatric spine surgery: is cefazolin enough? Spine Deform 8(4):669–676. https://doi.org/10.1007/s43390-020-00092-7
Karamian BA et al (2022) Cefazolin prophylaxis in spine surgery: patients are frequently underdosed and at increased risk for infection. Spine J 22(9):1442–1450. https://doi.org/10.1016/j.spinee.2022.05.018
Scaltriti S, Cencetti S, Rovesti S, Marchesi I, Bargellini A, Borella P (2007) Risk factors for particulate and microbial contamination of air in operating theatres. J Hosp Infect 66(4):320–326. https://doi.org/10.1016/j.jhin.2007.05.019
DiBartola AC et al (2019) Decreasing room traffic in orthopedic surgery: a quality improvement initiative. Am J Med Qual 34(6):561–568. https://doi.org/10.1177/1062860618821180
White EM, Wetle TF, Reddy A, Baier RR (2021) Front-line nursing home staff experiences during the COVID-19 pandemic. J Am Med Dir Assoc 22(1):199–203. https://doi.org/10.1016/j.jamda.2020.11.022
Darouiche RO et al (2010) Chlorhexidine-alcohol versus povidone-iodine for surgical-site antisepsis. N Engl J Med. https://doi.org/10.1016/S0749-4041(10)79279-4
The Joint Commission, the joint commission’s implementation guide for NPSG.07.05.01 on Surgical Site Infections: the SSI change project (2013) [Online]. Available: http://www.jointcommission.org. Accessed 7 Oct 2022
Tkatschenko D et al (2022) Prevention of surgical site infections in spine surgery: an international survey of clinical practices among expert spine surgeons. Global Spine J. https://doi.org/10.1177/21925682211068414
Melling AC, Ali B, Scott EM, Leaper DJ (2001) Effects of preoperative warming on the incidence of wound infection after clean surgery: a randomised controlled trial. Lancet 358:876–880
Molinari RW, Khera OA, Molinari WJ (2012) Prophylactic intraoperative powdered vancomycin and postoperative deep spinal wound infection: 1,512 consecutive surgical cases over a 6-year period. Eur Spine J. https://doi.org/10.1007/s00586-011-2104-z
Mallet C et al (2022) Does the use of intrawound povidone-iodine irrigation and local vancomycin powder impact surgical site infection rate in adolescent idiopathic scoliosis surgery? Eur Spine J 31(11):3020–3028. https://doi.org/10.1007/s00586-022-07340-6
Acknowledgements
The authors thank Regina Wood, MPH from PSSG and Michelle Marks, PT MA from the Harms study group for their help with the distribution of the survey. They thank also Drs. Carol Hasler, René Castelein, Sébastien Pesenta, Ilkka Helenius, Dror Ovadia, David Farrington, Muharrem Yazici, Moyo Kruyt, Jorge Mineiro, and Oliana Madeira for helping distribute our survey among the EPOS Spine study group.
Funding
This study was not conducted with financial support.
Author information
Authors and Affiliations
Consortia
Contributions
AC-G, JMS, CCR, CE, RS, HL, KL, AZB, PG, BDR, BI, ANL, MGV, European Pediatric Orthopaedic Society Spine Study Group, Pediatric Spine Study Group, and Harms Study Group made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data; or the creation of new software used in the work; they drafted the work or revised it critically for important intellectual content; approved the version to be published; and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Columbia University Institutional Review Board (Protocol AAAU3145) and was performed in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
This study qualifies for a waiver of consent because it is a survey of surgeons and does not involve patient participation. There is no potential to adversely affect the rights or welfare of subjects since this is a survey to gather surgeon opinion, with no patient data being collected or intervention being tested.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Concepción-González, A., Sarmiento, J.M., Rymond, C.C. et al. Evaluating compliance with the 2013 best practice guidelines for preventing surgical site infection in high-risk pediatric spine surgery. Spine Deform 12, 47–56 (2024). https://doi.org/10.1007/s43390-023-00755-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43390-023-00755-1