Abstract
Purpose
Previous reports have demonstrated the effectiveness of casting for EOS. Brace treatment for EOS has not been studied. The purpose of this multicenter retrospective study was to compare radiographic outcomes, complications, and rates of conversion to surgery in children with EOS treated with casting or bracing.
Methods
Children aged 2–6 years with idiopathic or neuromuscular EOS treated with casting or bracing with minimum follow-up of 2 years were identified.
Results
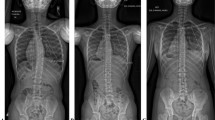
68 patients (36 cast, 32 brace) were analyzed. Diagnosis, age at start of treatment, and duration of follow-up were similar. Although the cast patients had a larger pre-treatment major curve magnitude (50° vs 31°, p < 0.001), both groups had a similar major curve magnitude at most recent follow-up (36° vs 32°, p = 0.456). T1–T12 and T1–S1 length increased in both groups. The cast and brace patients had similar complications and conversions to surgery. Sub-analysis showed that while casting resulted in curve improvement regardless of etiology, bracing was able to prevent curve progression in patients with idiopathic EOS but not in patients with non-idiopathic EOS (Δ− 15° vs 27°, p = 0.006). Regression analysis (significance p = 0.10) controlling for baseline age, major curve magnitude, and T1–T12 and T1–S1 length showed that treatment method was associated with difference in major curve magnitude (p = 0.090) and T1–T12 length (p = 0.024).
Conclusion
In our study, serial casting led to curve improvement in children with idiopathic and neuromuscular EOS, whereas brace treatment appeared to prevent curve progression in patients with idiopathic EOS but did not appear to control the curve in neuromuscular EOS.
Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Pehrsson K, Larsson S, Oden A, Nachemson A (1992) Long-term follow-up of patients with untreated scoliosis. A study of mortality, causes of death, and symptoms. Spine (Phila Pa 1976) 17(9):1091–1096
Branthwaite MA (1986) Cardiorespiratory consequences of unfused idiopathic scoliosis. Br J Dis Chest 80(4):360–369
Campbell RM Jr, Smith MD, Mayes TC et al (2003) The characteristics of thoracic insufficiency syndrome associated with fused ribs and congenital scoliosis. J Bone Jt Surg Am 85(3):399–408
Karol LA, Johnston C, Mladenov K, Schochet P, Walters P, Browne RH (2008) Pulmonary function following early thoracic fusion in non-neuromuscular scoliosis. J Bone Jt Surg Am 90(6):1272–1281
Bess S, Akbarnia BA, Thompson GH et al (2010) Complications of growing-rod treatment for early-onset scoliosis: analysis of one hundred and forty patients. J Bone Jt Surg Am 92(15):2533–2543
Upasani VV, Parvaresh KC, Pawelek JB et al (2016) Age at initiation and deformity magnitude influence complication rates of surgical treatment with traditional growing rods in early-onset scoliosis. Spine Deform 4(5):344–350
Teoh KH, Winson DM, James SH et al (2016) Do magnetic growing rods have lower complication rates compared with conventional growing rods? Spine J 16(4 Suppl):S40–44
Murphy RF, Moisan A, Kelly DM, Warner WC Jr, Jones TL, Sawyer JR (2016) Use of vertical expandable prosthetic titanium rib (VEPTR) in the treatment of congenital scoliosis without fused ribs. J Pediatr Orthop 36(4):329–335
Lucas G, Bollini G, Jouve JL et al (2013) Complications in pediatric spine surgery using the vertical expandable prosthetic titanium rib: the French experience. Spine (Phila Pa 1976) 38(25):E1589–1599
Sankar WN, Skaggs DL, Yazici M et al (2011) Lengthening of dual growing rods and the law of diminishing returns. Spine (Phila Pa 1976) 36(10):806–809
Cotrel Y, Morel G (1964) The Elongation-Derotation-flexion technic in the correction of scoliosis. Rev Chir Orthop Reparatrice Appar Mot 50:59–75
Mehta MH (2005) Growth as a corrective force in the early treatment of progressive infantile scoliosis. J Bone Jt Surg Br 87(9):1237–1247
Sanders JO, D'Astous J, Fitzgerald M, Khoury JG, Kishan S, Sturm PF (2009) Derotational casting for progressive infantile scoliosis. J Pediatr Orthop 29(6):581–587
Fletcher ND, McClung A, Rathjen KE, Denning JR, Browne R, Johnston CE 3rd (2012) Serial casting as a delay tactic in the treatment of moderate-to-severe early-onset scoliosis. J Pediatr Orthop 32(7):664–671
Baulesh DM, Huh J, Judkins T, Garg S, Miller NH, Erickson MA (2012) The role of serial casting in early-onset scoliosis (EOS). J Pediatr Orthop 32(7):658–663
Demirkiran HG, Bekmez S, Celilov R, Ayvaz M, Dede O, Yazici M (2015) Serial derotational casting in congenital scoliosis as a time-buying strategy. J Pediatr Orthop 35(1):43–49
Gussous YM, Tarima S, Zhao S et al (2015) Serial derotational casting in idiopathic and non-idiopathic progressive early-onset scoliosis. Spine Deform 3(3):233–238
Cao J, Zhang XJ, Sun N et al (2017) The therapeutic characteristics of serial casting on congenital scoliosis: a comparison with non-congenital cases from a single-center experience. J Orthop Surg Res 12(1):56
Stasikelis PJ, Carpenter AM (2018) Results of casting in severe curves in infantile scoliosis. J Pediatr Orthop 38(4):e186–e189
FDA Drug Safety Communication. FDA review results in new warnings about using general anesthetics and sedation drugs in young children and pregnant women. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-review-results-new-warnings-about-using-general-anesthetics-and. Accessed 11 Aug 2019.
Katz DE, Herring JA, Browne RH, Kelly DM, Birch JG (2010) Brace wear control of curve progression in adolescent idiopathic scoliosis. J Bone Jt Surg Am 92(6):1343–1352
Weinstein SL, Dolan LA, Wright JG, Dobbs MB (2013) Effects of bracing in adolescents with idiopathic scoliosis. N Engl J Med 369(16):1512–1521
Thorsness RJ, Faust JR, Behrend CJ, Sanders JO (2015) Nonsurgical management of early-onset scoliosis. J Am Acad Orthop Surg 23(9):519–528
Altman D (1991) Practical statistics for medical research. Chapman and Hall, London
Gomez JA, Grzywna A, Miller PE et al (2017) Initial cast correction as a predictor of treatment outcome success for infantile idiopathic scoliosis. J Pediatr Orthop 37(8):e625–e630
Welborn MC, D'Astous J, Bratton S, Heflin J (2018) Infantile idiopathic scoliosis: factors affecting EDF casting success. Spine Deform 6(5):614–620
Landauer F, Wimmer C, Behensky H (2003) Estimating the final outcome of brace treatment for idiopathic thoracic scoliosis at 6-month follow-up. Pediatr Rehabil 6(3–4):201–207
Miller A, Temple T, Miller F (1996) Impact of orthoses on the rate of scoliosis progression in children with cerebral palsy. J Pediatr Orthop 16(3):332–335
Olafsson Y, Saraste H, Al-Dabbagh Z (1999) Brace treatment in neuromuscular spine deformity. J Pediatr Orthop 19(3):376–379
Halawi MJ, Lark RK, Fitch RD (2015) Neuromuscular scoliosis: current concepts. Orthopedics 38(6):e452–456
Gillingham BL, Fan RA, Akbarnia BA (2006) Early onset idiopathic scoliosis. J Am Acad Orthop Surg 14(2):101–112
Acknowledgements
Dr. Li has received a research grant and non-financial support from the Scoliosis Research Society. Dr. Thompson has received personal fees from Shriners Hospitals for Children and Wolters Kluwer, personal fees and royalties from OrthoPediatrics, and non-financial support from NuVasive and the Scoliosis Research Society. Dr. Sturm has received personal fees from Depuy Synthes/Johnson and Johnson and NuVasive, and is a shareholder of Green Sun Medical. Dr. Emans has received personal fees from Depuy Synthes/Johnson and Johnson and Zimmer Biomet. Dr. Sponseller has received a grant from Depuy Synthes/Johnson and Johnson, and reports other relationships with Depuy Synthes/Johnson and Johnson, Globus, OrthoPediatrics, and Journal of Bone and Joint Surgery. Dr. Glotzbecker has received personal fees and non-financial support from Medtronic, Depuy Synthes/Johnson and Johnson, Zimmer Biomet, NuVasive, and Orthobullets. The Pediatric Spine Study Group has received research grants from the Pediatric Orthopaedic Society of North America, Food and Drug Administration, Depuy Synthes/Johnson and Johnson, NuVasive, Children’s Spine Foundation, and Growing Spine Foundation. The remaining authors declare that they have no conflicts of interest.
Funding
The study was funded by departmental resources.
Author information
Authors and Affiliations
Consortia
Contributions
YL: made substantial contributions to the conception or design of the work, and performed analysis and interpretation of data; drafted the work; approved the version to be published; agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. JS: performed acquisition of data; revised the work critically for important intellectual content; approved the version to be published; agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. JG: performed data analysis; revised the work critically for important intellectual content; approved the version to be published; agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. GHT: performed interpretation of data; revised the work critically for important intellectual content; approved the version to be published; agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. PFS: performed interpretation of data; revised the work critically for important intellectual content; approved the version to be published; agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. JBE: performed interpretation of data; revised the work critically for important intellectual content; approved the version to be published; agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. PDS: performed interpretation of data; revised the work critically for important intellectual content; approved the version to be published; agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. MPG: made substantial contributions to the conception or design of the work and performed interpretation of data; revised the work critically for important intellectual content; approved the version to be published; agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Pediatric Spine Study Group: performed acquisition of data; revised the work critically for important intellectual content; approved the version to be published; agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Li has received a research grant and non-financial support from the Scoliosis Research Society. Dr. Thompson has received personal fees from Shriners Hospitals for Children and Wolters Kluwer, personal fees and royalties from OrthoPediatrics, and non-financial support from NuVasive and the Scoliosis Research Society. Dr. Sturm has received personal fees from Depuy Synthes/Johnson and Johnson and NuVasive, and is a shareholder of Green Sun Medical. Dr. Emans has received personal fees from Depuy Synthes/Johnson and Johnson and Zimmer Biomet. Dr. Sponseller has received a grant from Depuy Synthes/Johnson and Johnson, and reports other relationships with Depuy Synthes/Johnson and Johnson, Globus, OrthoPediatrics, and Journal of Bone and Joint Surgery. Dr. Glotzbecker has received personal fees and non-financial support from Medtronic, Depuy Synthes/Johnson and Johnson, Zimmer Biomet, NuVasive, and Orthobullets. The Pediatric Spine Study Group has received research grants from the Pediatric Orthopaedic Society of North America, Food and Drug Administration, Depuy Synthes/Johnson and Johnson, NuVasive, Children’s Spine Foundation, and Growing Spine Foundation. The remaining authors declare that they have no conflicts of interest.
Ethics approval
IRB approval, University of Michigan, HUM 00082789, 1/7/14.
Consent to participate
Informed consent to participate in this study was obtained from the parent or legal guardian of all participants.
Consent for publication
Not applicable.
Code availability
The software application and code used for the statistical analysis are available from the corresponding author on reasonable request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, Y., Swallow, J., Gagnier, J. et al. A report of two conservative approaches to early onset scoliosis: serial casting and bracing. Spine Deform 9, 595–602 (2021). https://doi.org/10.1007/s43390-020-00213-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43390-020-00213-2