Abstract
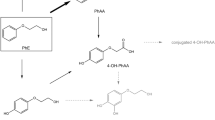
Although 2-amino-5-nitrophenol (2A5NP) is one of the ingredients of hair dye, there has been no information on the dermal absorption rate of 2A5NP. 2A5NP is managed at less than 1.5% in Korea and Japan. In this study, analytical methods were developed and validated using high-performance liquid chromatography (HPLC) in various matrices of wash, swab, stratum corneum (SC), skin (dermis + epidermis), and receptor fluid (RF). Validation results were acceptable based on Korea Ministry of Food and Drug Safety (MFDS) guideline. The HPLC analysis showed a good linearity (r2 = 0.9992–0.9999), a high accuracy (93.1–110.2%), and a good precision (1.1–8.1%) in accordance with the validation guideline. Franz diffusion cell was used to determine dermal absorption of 2A5NP using mini pig skin. 2A5NP (1.5%) was applied to skin at 10 μl/cm2. For certain cosmetic ingredients such as hair dye with short exposure time, an interim wash step (after 30 min) was added during the study. After application for 30 min and 24 h, skin was wiped off with swab and SC was collected using tape stripping. RF was sampled at 0, 1, 2, 4, 8, 12, and 24 h. Total dermal absorption rate of 2A5NP (1.5%) was determined to be 13.6 ± 2.9%.



Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article.
Abbreviations
- 2A5NP:
-
2-Amino-5-nitrophenol
- ACN:
-
Acetonitrile
- CV:
-
Coefficient of variation
- DW:
-
Distilled water
- FDCA:
-
US Federal Food, Drug and Cosmetic Act
- HOQ:
-
High of quantification
- HPLC:
-
High-performance liquid chromatography
- Js:
-
Equilibrium flux
- LLOQ:
-
Lower limit of quantitation
- LOQ:
-
Limit of quantification
- MFDS:
-
Ministry of Food and Drug safety
- MOQ:
-
Minimum of quantification
- NOAEL:
-
No observed adverse effect level
- PEG:
-
Polyethylene glycol
- PTFE:
-
Polytetrafluoroethylene
- RF:
-
Receptor fluid
- SC:
-
Stratum corneum
- SED:
-
Systemic exposure dose
- TFA:
-
Trifluoroacetic acid
References
KHIDI (Korea Health Industry Development Institute) (2019) Cosmetic industry analysis. https://www.khidi.or.kr/board/view?linkId=48809330&menuId=MENU00085
KCA (Korea Cosmetic Association) (2021) Cosmetics market information. https://kcia.or.kr/home/industry/industry_01.php?type=view&no=13559&ss=page%3D%26skind%3D%26sword%3D%26ob%3D
Kim K, Kabir E, Jahan SA (2016) The use of personal hair dye and its implications for human health. Environ Int 89:222–227. https://doi.org/10.1016/j.envint.2016.01.018
Hamann D, Yazar K, Hamann CR, Thyssen JP, Lidén C (2014) p-Phenylenediamine and other allergens in hair dye products in the U nited S tates: A consumer exposure study. Contact Dermatitis 70:213–218. https://doi.org/10.1111/cod.12164
MFDS (Ministry of Food and Drug Safety) (2019) Common guidelines for risk assessment of human products. https://www.mfds.go.kr/brd/m_210/view.do?seq=14364
Lee JD, Kim J, Jang HJ, Lee B, Kim K (2019) Percutaneous permeability of 1-phenoxy-2-propanol, a preservative in cosmetics. Regul Toxicol Pharmacol 103:56–62. https://doi.org/10.1016/j.yrtph.2019.01.002
Poet TS, McDougal JN (2002) Skin absorption and human risk assessment. Chem Biol Interact 140:19–34. https://doi.org/10.1016/S0009-2797(02)00013-3
PubChem (2021) Chemical and physical properties of 2-amino-5-nitrrophenol. https://pubchem.ncbi.nlm.nih.gov/compound/2-Amino-5-nitrophenol
IARC (International Agency for Research on Cancer) (1993) Occupational exposures of hairdressers and barbers and personal use of hair colourants; some hair dyes, cosmetic colourants, industrial dyestuffs and aromatic amines. IARC monographs on the evaluation of carcinogenic risks to humans, p. 57
Society of Dyers and Colourists (1971) Colour Index, 3rd edn, Vol. 4. Bradford, Yorkshire, p. 4647.
NCATS (National Center for Advancing Translational Sciences) (2021) Properties of 2-amino-5-nitrrophenol. https://drugs.ncats.io/drug/0149H05U82#:~:text=2%2DAmino%2D5%2Dnitrophenol%20is%20used%20as%20an%20intermediate,lacquers%2C%20inks%20and%20wood%20stains.&text=It%20has%20been%20used%20(and,in%20permanent%20hair%20colouring%20products
Chung K (2016) Azo dyes and human health: a review. J Environ Sci Health C 34:233–261. https://doi.org/10.1080/10590501.2016.1236602
SCCS (Scientific Committee on Consumer Safety) (2010) Opinion on basic criteria for the In vitro assessment of dermal absorption of cosmetic ingredients. Updated 22 June 2010. SCCS/1358/10
Burnett C, Bergfeld W, Belsito D, Klaassen C, Marks J, Shank R, Andersen F (2009) Final report on the safety assessment of amino nitrophenols as used in hair dyes. Int J Toxicol 28:217S. https://doi.org/10.1177/109158180935465
Franco JH, Silva BF, Zanoni MV (2015) Using ionic liquid combined with HPLC-DAD to analyze semi-permanent hair dyes in commercial formulations. Anal Methods 7:1115–1122. https://doi.org/10.1039/C4AY02311K
Ahmed HAM, Maaboud RMA, Latif FFA, El-Dean AMK, El-Shaieb KM, Vilanova E, Estevan C (2013) Different analytical methods of para-phenylenediamine based hair dye. J Cosmet Dermatol Sci Appl 3:17-25. https://doi.org/10.4236/jcdsa.2013.33A1003
Andrisano V, Gotti R, Di Pietra AM, Cavrini V (1994) HPLC analysis of oxidation hair dyes in permanent hair colorants. J Liq Chromatogr Relat Technol 17:2919–2937. https://doi.org/10.1080/10826079408013510
Shao BH, Xu XZ, Yan JW, Fu XY (2001) Quantitative determination of commercial oxidation hair dyes by reversed-phase HPLC. J Liq Chromatogr Relat Technol 24:241–249. https://doi.org/10.1081/JLC-100001485
MFDS (Ministry of Food and Drug Safety) (2013) Guidelines for validation of bio-sampling methods. http://www.mfds.go.kr/index.do?mid=687andpageNo=2andseq=8003andcmd=v
Betz JM, Brown PN, Roman MC (2011) Accuracy, precision, and reliability of chemical measurements in natural products research. Fitoterapia 82:44–52. https://doi.org/10.1016/j.fitote.2010.09.011
OECD (Organisation for Economic Cooperation and Development) (2004) Guideline for the testing of chemicals skin absorption: in vitro method. 427. https://ntp.niehs.nih.gov/iccvam/suppdocs/feddocs/oecd/oecdtg428-508.pdf
MFDS (Ministry of Food and Drug Safety) (2009) Guideline for in vitro skin absorption method. B1-2009-4-001. http://www.nifds.go.kr/brd/m_15/view.do?seq=11430
Kim J, Im JE, Lee JD, Kim K (2021) Analytical method development and percutaneous absorption of propylidene phthalide, a cosmetic ingredient. J Toxicol Environ Health A 84:811–820. https://doi.org/10.1080/15287394.2021.1944941
MFDS (The Ministry of Food and Drug Safety) (2014) Regulations on cosmetic safety standards. Ministry of Food and Drug Safety 2014–118 issue. http://www.mfds.go.kr/index.do?mid=687andpageNo=2andseq=8003andcmd=v
SCCP (Scientific Committee on Consumer Products) (2009) Opinion on intermediates and reaction products of oxidative hair dye ingredients formed during hair dyeing. https://www.semanticscholar.org/paper/ON-Intermediates-and-reaction-products-of-oxidative-Dubakiene-Ja%C5%BAwiec-Kanyion/a789f24b1b9e99b2adefbb4df6c40ace1ca85162
Im J, Kim HY, Lee JD, Park J, Kang K, Kim K (2021) Effect of application amounts on in vitro dermal absorption test using caffeine and testosterone. Pharmaceutics 13:641. https://doi.org/10.3390/pharmaceutics13050641
Moosavi SM, Ghassabian S (2018) Linearity of calibration curves for analytical methods: A review of criteria for assessment of method reliability. IntechOpen Limited London, UK
WHO (World Health Organization) (2006) Dermal absorption. World Health Organization. https://apps.who.int/iris/handle/10665/43542
Lee Y, Kim H, Pham Q, Lee J, Kim K (2022) Pharmacokinetics and the dermal absorption of bromochlorophene, a cosmetic preservative ingredient, in rats. Toxics 10:329. https://doi.org/10.3390/toxics10060329
Pel E, Bordin G, Rodriguez AR (1998) HPLC candidate reference method for oxidative hair dye analysis. I. separation and stability testing. Journal of Liquid Chromatography & Related Technologies 21:883–901. https://doi.org/10.1080/10826079808000516
SCCS (Scientific Committee on Consumer Safety) (2009) Opinion on 5- amino-6-chloro-o-cresol. https://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_o_005.pdf
EFSA (European Food Safety Authority), Buist H, Craig P, Dewhurst I, Hougaard Bennekou S, Kneuer C et al (2017) Guidance on dermal absorption. EFSA J 15: https://doi.org/10.2903/j.efsa.2017.4873
Acknowledgements
This work was supported by Grants (19172MFDS221 and 22202MFDS158) from the Ministry of Food and Drug Safety, Republic of Korea in 2019 and 2022. This work was supported by the Korea Environment Industry & Technology Institute (KEITI) through the Technology Program for Establishing Biocide Safety Management, funded by the Korea Environment (MOE) (2021002970001, 1485017976).
Funding
This work was supported by Grants (19172MFDS221 and 22202MFDS158) from the Ministry of Food and Drug Safety, Republic of Korea in 2019 and 2022. This work was supported by the Korea Environment Industry & Technology Institute (KEITI) through the Technology Program for Establishing Biocide Safety Management, funded by the Korea Environment (MOE) (2021002970001, 1485017976).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by YJK, HYK, JEI, HYK and JDL. Writing-original draft preparation was performed YJK. And writing-review, editing and supervision were performed K-BK. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Consent to participate
Informed consent was obtained from all participants.
Ethics approval
No ethical approval is required.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, Y.J., Kim, H.Y., Lee, J.D. et al. Analytical method development and dermal absorption of 2-amino-5-nitrophenol (2A5NP), a hair dye ingredient under oxidative condition. Toxicol Res. 39, 231–238 (2023). https://doi.org/10.1007/s43188-022-00159-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43188-022-00159-9