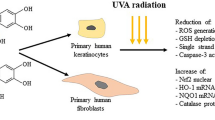
Abstract
Maintaining youthful skin from photoaging with natural products, including essential oils, is a vital strategy that has piqued the interest of researchers in the pharmaceutical and cosmetic industries. This research aimed to investigate the protective properties of Zingiber montanum (J. Koenig) Link ex A. Dietr. essential oil against ultraviolet B (UVB)-induced skin damage and photoaging in normal human dermal fibroblast (HDFn) cells. The essential oil was extracted from fresh plant rhizomes using solvent-free microwave extraction. Its antiphotoaging properties in HDFn cells were investigated using reactive oxygen species (ROS)-scavenging, wound healing, matrix metalloproteinases (MMP-1, MMP-3, and MMP-9) expression, procollagen synthesis, and elastase and tyrosinase inhibitory assays. The results showed that the test oil exhibited no significant toxicity in HDFn at concentrations up to 10 mg/mL, with cell viability exceeding 90%. Following UVB irradiation at 30 mJ/cm2, Z. montanum oil demonstrated time and concentration-dependent ROS radical scavenging capabilities. In a cell migration assay, the essential oil demonstrated wound-healing properties. Z. montanum oil suppressed the expression of MMPs and enhanced the synthesis of type I procollagen at a concentration of 0.1–1 mg/mL. In addition, 0.1–1 mg/mL Z. montanum oil inhibited elastase activity in a concentration-dependent manner but did not affect tyrosinase activity. From these findings, the essential oil of Z. montanum could have potential applications in developing cosmeceutical products to prevent skin photoaging.






Similar content being viewed by others
Abbreviations
- AP-1:
-
Activator protein 1
- GC–MS:
-
Gas chromatography-mass spectrometry
- HDFn:
-
Normal human dermal fibroblast
- IL:
-
Interleukin
- MAPKs:
-
Mitogen-activated protein kinases
- MMPs:
-
Matrix metalloproteinases
- NAC:
-
N-Acetylcysteine
- NF-κB:
-
Nuclear factor-κB
- ROS:
-
Reactive oxygen species
- UVB:
-
Ultraviolet B
- Z. montanum :
-
Zingiber montanum (J. Koenig) Link ex A. Dietr
References
Kang CH, Rhie SJ, Kim YC (2018) Antioxidant and skin anti-aging effects of marigold methanol extract. Toxicol Res 34:31–39. https://doi.org/10.5487/TR.2018.34.1.031
Farage MA, Miller KW, Elsner P, Maibach HI (2008) Intrinsic and extrinsic factors in skin ageing: a review. Int J Cosmet Sci 30:87–95. https://doi.org/10.1111/j.1468-2494.2007.00415.x
Krutmann J, Schikowski T, Morita A, Berneburg M (2021) Environmentally-induced (extrinsic) skin aging: exposomal factors and underlying mechanisms. J Investig Dermatol 141:1096–1103. https://doi.org/10.1016/j.jid.2020.12.011
Ichihashi M, Ando H, Yoshida M, Niki Y, Matsui M (2009) Photoaging of the skin. Anti-Aging Med 6:46–59. https://doi.org/10.3793/jaam.6.46
Matsumura Y, Ananthaswamy HN (2004) Toxic effects of ultraviolet radiation on the skin. Toxicol Appl Pharmacol 195:298–308. https://doi.org/10.1016/j.taap.2003.08.019
Monteiro e Silva AS, Michniak-Kohn B, Leonardi RG (2017) An overview about oxidation in clinical practice of skin aging. An Bras Dermatol 92:367–374. https://doi.org/10.1590/abd1806-4841.20175481
Park KH, Kim J, Jung S, Sung KH, Son YK, Bae JM, Kim BH (2019) Alleviation of ultraviolet B-induced photoaging by 7-MEGATM 500 in hairless mouse skin. Toxicol Res 35:353–359. https://doi.org/10.5487/tr.2019.35.4.353
Pittayapruek P, Meephansan J, Prapapan O, Komine M, Ohtsuki M (2016) Role of matrix metalloproteinases in photoaging and photocarcinogenesis. Int J Mol Sci 17:868. https://doi.org/10.3390/ijms17060868
Hwang KA, Yi BR, Chul K (2011) Molecular mechanisms and in vivo mouse models of skin aging associated with dermal matrix alterations. Lab Anim Res 27:1–8. https://doi.org/10.5625/lar.2011.27.1.1
Pillai S, Oresajo C, Hayward J (2005) Ultraviolet radiation and skin aging: roles of reactive oxygen species, inflammation andprotease activation, and strategies for prevention of inflammation-induced matrix degradation - a review. Int J Cosmet Sci 27:17–34. https://doi.org/10.1111/j.1467-2494.2004.00241.x
Chase AJ, Bond M, Crook MF, Newby AC (2002) Role of nuclear factor-κB activation in metalloproteinase-1, -3, and -9 secretion by human macrophages in vitro and rabbit foam cells produced in vivo. Arterioscler Thromb Vasc Biol 22:765–771. https://doi.org/10.1161/01.atv.0000015078.09208.92
Rabe JH, Mamelak AJ, McElgunn PJS, Morison WL, Sauder DN (2006) Photoaging: mechanisms and repair. J Am Acad Dermatol 55:1–19. https://doi.org/10.1016/j.jaad.2005.05.010
Vincenti MP, Brinckerhoff CE (2002) Transcriptional regulation of collagenase (MMP-1, MMP-13) genes in arthritis: integration of complex signaling pathways for the recruitment of gene-specific transcription factors. Arthritis Res 4:157–164. https://doi.org/10.1186/ar401
Correa D (2021) Global cosmetics market to reach $463.5 billion by 2027: Allied Market Research. https://www.globenewswire.com/news-release/2021/02/04/2170144/0/en/Global-Cosmetics-Market-to-Reach-463-5-Billion-by-2027-Allied-Market-Research.html. Accessed 18 Apr 2021
Cavinato M, Waltenberger B, Baraldo G, Grade CVC, Stuppner H, Jansen-Dürr P (2017) Plant extracts and natural compounds used against UVB-induced photoaging. Biogerontology 18:499–516. https://doi.org/10.1007/s10522-017-9715-7
Kostyuk V, Potapovich A, Albuhaydar AR, Mayer W, De Luca C, Korkina L (2018) Natural substances for prevention of skin photoaging: Screening systems in the development of sunscreen and rejuvenation cosmetics. Rejuv Res 21:91–101. https://doi.org/10.1089/rej.2017.1931
Sarkic A, Stappen I (2018) Essential oils and their single compounds in cosmetics—a critical review. Cosmetics 5:11. https://doi.org/10.3390/cosmetics5010011
Chongmelaxme B, Sruamsiri R, Dilokthornsakul P, Dhippayom T, Kongkaew C, Saokaew S, Chuthaputti A, Chaiyakunapruk N (2017) Clinical effects of Zingiber cassumunar (Plai): a systematic review. Complement Ther Med 35:70–77. https://doi.org/10.1016/j.ctim.2017.09.009
Pongprayoon U, Soontornsaratune P, Jarikasem S, Sematong T, Wasuwat S, Claeson P (1997) Topical antiinflammatory activity of the major lipophilic constituents of the rhizome of Zingiber cassumunar. Part I: the essential oil. Phytomedicine 3:319–322. https://doi.org/10.1016/s0944-7113(97)80003-7
Rahman L, Lembang RS, Lallo S, Handayani SR, Usmanengsi Permana AD (2021) Bioadhesive dermal patch as promisingapproach for improved antibacterial activity of bioactive compound of Zingiber cassumunar Roxb in ex vivo Staphylococcusaureus skin infection model. J Drug Deliv Sci Technol 63: https://doi.org/10.1016/j.jddst.2021.102522
Nagano T, Oyama Y, Kajita N, Chikahisa L, Nakata M, Okazaki E, Masuda T (1997) New curcuminoids isolated from Zingiber cassumuar protect cells suffering from oxidative stress: a flow-cytometric study using rat thymocytes and H2O2. Jpn J Pharmacol 75:363–370. https://doi.org/10.1254/jjp.75.363
Koontongkaew S, Meesuk L, Aupaphong V, Dechatiwongse Na Ayudhaya T, Poachanukoon O (2013) Inhibitory effect of Zingiber cassumunar extracts on lipopolysaccharide-induced cyclooxygenase-2 and matrix metalloproteinase expression in human gingival fibroblasts. J Periodontal Res 48:507–516. https://doi.org/10.1111/jre.12033
Jeenapongsa R, Yoovathaworn K, Sriwatanakul KM, Pongprayoon U, Sriwatanakul K (2003) Anti-inflammatory activity of (E)-1-(3,4-dimethoxyphenyl) butadiene from Zingiber cassumunar Roxb. J Ethnopharmacol 87:143–148. https://doi.org/10.1016/s0378-8741(03)00098-9
Jitapunkul K, Poachanukoon O, Hannongbua S, Toochinda P, Lawtrakul L (2017) Simulation study of interactions between two bioactive components from Zingiber cassumunar and 5-lipoxygenase. Cell Mol Bioeng 11:77–89. https://doi.org/10.1007/s12195-017-0515-6
Pongprayoon U, Tuchinda P, Claeson P, Sematong T, Reutrakul V, Soontornsaratune P (1997) Topical antiinflammatoryactivity of the major lipophilic constituents of the rhizome of Zingiber cassumunar. Part II: Hexane extractives. Phytomedicine 3:323–326. https://doi.org/10.1016/s0944-7113(97)80004-9
Nakamura S, Iwami J, Matsuda H, Wakayama H, Pongpiriyadacha Y, Yoshikawa M (2009) Structures of new phenylbutanoids and nitric oxide production inhibitors from the rhizomes of Zingiber cassumunar. Chem Pharm Bull 57:1267–1272. https://doi.org/10.1248/cpb.57.1267
Tewtrakul S, Subhadhirasakul S (2007) Anti-allergic activity of some selected plants in the Zingiberaceae family. J Ethnopharmacol 109:535–538. https://doi.org/10.1016/j.jep.2006.08.010
Poachanukoon O, Meesuk L, Pattanacharoenchai N, Monthanapisut P, Dechatiwongse Na Ayudhya T, Koontongkaew S (2015) Zingiber cassumunar ROXb and its active constituent inhibit MMP-9 direct activation by house dust mite allergens and MMP-9 expression in PMA-stimulated human airway epithelial cells. Asian Pac J Allergy Immunol 33:42–51 https://doi.org/10.12932/ap0490.33.1.2015
Chaiwongsa R, Ongchai S, Boonsing P, Kongtawelert P, Panthong A, Reutrakul V (2013) Active compound of Zingiber Cassumunar Roxb. down-regulates the expression of genes involved in joint erosion in a human synovial fibroblast cell line. Afr J Tradit Complement Altern Med 10:40–48. https://doi.org/10.4314/ajtcam.v10i1.7
Li MX, Bai X, Ma YP, Zhang HX, Nama N, Pei SJ, Du ZZ (2019) Cosmetic potentials of extracts and compounds from Zingiber cassumunar Roxb. rhizome. Ind Crops Prod 141:111764. https://doi.org/10.1016/j.indcrop.2019.111764
Leelarungrayub J, Manorsoi J, Manorsoi A (2017) Anti-inflammatory activity of niosomes entrapped with Plai oil (Zingiber cassumunar Roxb.) by therapeutic ultrasound in a rat model. Int J Nanomed 12:2469–2476. https://doi.org/10.2147/ijn.s129131
Yingngam B, Brantner A (2018) Boosting the essential oil yield from the rhizomes of cassumunar ginger by an eco-friendly solvent-free microwave extraction combined with central composite design. J Essent Oil Res 30:409–420. https://doi.org/10.1080/10412905.2018.1503099
Yingngam B, Brantner A, Treichler M, Brugger N, Navabhatra A, Nakonrat P (2021) Optimization of the eco-friendly solvent-free microwave extraction of Limnophila aromatica essential oil. Ind Crops Prod 165:113443. https://doi.org/10.1016/j.indcrop.2021.113443
WebBook, NIST Chemistry (2020) NIST standard reference database number 69. 2018. https://webbook.nist.gov/chemistry. Accessed 9 Mar 2020
Mueller H, Kassack UM, Wiese M (2004) Comparison of the usefulness of the MTT, ATP, and calcein assays to predict the potency of cytotoxic agents in various human cancer cell lines. J Biomol Screen 9:506–515. https://doi.org/10.1177/1087057104265386
Kalyanaraman B, Darley-Usmar V, Davies KJA, Dennery PA, Forman HJ, Grisham MB, Mann GE, Moore K, Roberts LJ, Ischiropoulos H (2012) Measuring reactive oxygen and nitrogen species with fluorescent probes: challenges and limitations. Free Radic Biol Med 52:1–6. https://doi.org/10.1016/j.freeradbiomed.2011.09.030
Suarez-Arnedo A, Torres Figueroa F, Clavijo C, Arbeláez P, Cruz JC, Muñoz-Camargo C (2020) An image J plugin for the high throughput image analysis of in vitro scratch wound healing assays. PLoS ONE 15: https://doi.org/10.1371/journal.pone.0232565
Muniandy K, Gothai S, Tan WS, Kumar SS, Mohd Esa N, Chandramohan G, Al-Numair KS, Arulselvan P (2018) In vitro wound healing potential of stem extract of Alternanthera sessilis. J Evid Based Complementary Altern Med 2018:3142073. https://doi.org/10.1155/2018/3142073
Momtaz S, Lall N, Basson A (2008) Inhibitory activities of mushroom tyrosine and DOPA oxidation by plant extracts. S Afr J Bot 74:577–582. https://doi.org/10.1016/j.sajb.2008.02.005
An YA, Hwang JY, Lee JS, Kim YC (2015) Cornus officinalis methanol extract upregulates melanogenesis in Melan-a cells. Toxicol Res 31:165–172. https://doi.org/10.5487/tr.2015.31.2.165
Chang TS (2012) Natural melanogenesis inhibitors acting through the down-regulation of tyrosinase activity. Materials (Basel) 5:1661–1685. https://doi.org/10.3390/ma5091661
Kim SE, Lee CM, Kim YC (2017) Anti-melanogenic effect of Oenothera laciniata methanol extract in Melan-a cells. Toxicol Res 33:55–62. https://doi.org/10.5487/tr.2017.33.1.055
Fisher GJ, Datta S, Wang Z, Li XY, Quan T, Chung JH, Kang S, Voorhees JJ (2000) c-Jun–dependent inhibition of cutaneous procollagen transcription following ultraviolet irradiation is reversed by all-trans retinoic acid. J Clin Investig 106:663–670. https://doi.org/10.1172/jci9362
Kim MS, Kim YK, Cho KH, Chung JH (2006) Regulation of type I procollagen and MMP-1 expression after single or repeated exposure to infrared radiation in human skin. Mech Ageing Dev 127:875–882. https://doi.org/10.1016/j.mad.2006.09.007
Park MA, Sim MJ, Kim YC (2017) Anti-photoaging effects of Angelica acutiloba root ethanol extract in human dermal fibroblasts. Toxicol Res 33:125–134. https://doi.org/10.5487/tr.2017.33.2.125
Sriram G, Bigliardi PL, Bigliardi-Qi M (2015) Fibroblast heterogeneity and its implications for engineering organotypic skin models in vitro. Eur J Cell Biol 94:483–512. https://doi.org/10.1016/j.ejcb.2015.08.001
Wang Y, Wang Y, Cheng C, Wang L, Yang J, Li H, Liu H, Cai L (2016) Establishment of a photoaging model in human dermal fibroblasts and study on photoaging-related biomarkers. Int J Clin Exp Med 9(5):8846–8851 http://www.ijcem.com/files/ijcem0015786.pdf
Cooper S, Bowden G (2007) Ultraviolet B regulation of transcription factor families: Roles of nuclear factor-kappa B (NF-κB) and activator protein-1 (AP-1) in UVB-induced skin carcinogenesis. Curr Cancer Drug Targets 7:325–334. https://doi.org/10.2174/156800907780809714
Kwon KR, Alam MB, Park JH, Kim TH, Lee SH (2019) Attenuation of UVB-induced photo-aging by polyphenolic-rich Spatholobus suberectus stem extract via modulation of MAPK/AP-1/MMPs signaling in human keratinocytes. Nutrients 11:1341. https://doi.org/10.3390/nu11061341
Subedi L, Lee TH, Wahedi HM, Baek SH, Kim SY (2017) Resveratrol-enriched rice attenuates UVB-ROS-induced skin aging via downregulation of inflammatory cascades. Oxid Med Cell Longev 2017:8379539. https://doi.org/10.1155/2017/8379539
Fisher GJ, Datta SC, Talwar HS, Wang ZQ, Varani J, Kang S, Voorhees JJ (1996) Molecular basis of sun-induced premature skin ageing and retinoid antagonism. Nature 379:335–339. https://doi.org/10.1038/379335a0
Quan T, Qin Z, Xia W, Shao Y, Voorhees JJ, Fisher GJ (2009) Matrix-degrading metalloproteinases in photoaging. J Investig Dermatol 14:20–24. https://doi.org/10.1038/jidsymp.2009.8
Lee GH, Jin SW, Kim SJ, Pham TH, Choi JH, Jeong HG (2019) Tetrabromobisphenol A induces MMP-9 expression via NADPH oxidase and the activation of ROS, MAPK, and Akt pathways in human breast cancer MCF-7 cells. Toxicol Res 35:93–101. https://doi.org/10.5487/tr.2019.35.1.093
Shin JW, Kwon SH, Choi JY, Na JI, Huh CH, Choi HR, Park KC (2019) Molecular mechanisms of dermal aging and antiaging approaches. Int J Mol Sci 20:2126. https://doi.org/10.3390/ijms20092126
Tu Y, Quan T (2016) Oxidative stress and human skin connective tissue aging. Cosmetics 3:28. https://doi.org/10.3390/cosmetics3030028
Cho JW, Jeong YS, Han JW, Chun YJ, Kim HK, Kim MY, Kim BJ, Park KM, Kim JK, Kim JH, Cho SM (2011) Skin hydration and collagen synthesis of AF-343 in HS68 cell line and NC/Nga mice by filaggrin expression and suppression of matrix metallopreteinase. Toxicol Res 27:225–229. https://doi.org/10.5487/tr.2011.27.4.225
Ha BG, Park MA, Lee CM, Kim YC (2015) Antioxidant activity and anti-wrinkle effects of Aceriphyllum rossii leaf ethanol extract. Toxicol Res 31:363–369. https://doi.org/10.5487/tr.2015.31.4.363
Kim SR, Jung YR, An HJ, Kim DH, Jang EJ, Choi YJ, Moon KM, Park MH, Park CH, Chung KW, Bae HR, Choi YW, Kim ND, Chung HY (2013) Anti-wrinkle and anti-inflammatory effects of active garlic components and the inhibition of MMPs via NF-κB signaling. PLoS ONE 8: https://doi.org/10.1371/journal.pone.0073877
Pham QL, Jang HJ, Kim KB (2017) Anti-wrinkle effect of fermented black ginseng on human fibroblasts. Int J Mol Med 39:681–686. https://doi.org/10.3892/ijmm.2017.2858
Aziz J, Shezali H, Radzi Z, Yahya NA, Abu Kassim NH, Czernuszka J, Rahman MT (2016) Molecular mechanisms of stress-responsive changes in collagen and elastin networks in skin. Skin Pharmacol Physiol 29:190–203. https://doi.org/10.1159/000447017
Minami A, Fujita Y, Goto J, Iuchi A, Fujita K, Mikami Y, Shiratori M, Ishii A, Mitragotri S, Iwao Y, Kanazawa H, Kurebayashi Y, Takahashi T, Otsubo T, Ikeda K, Suzuki T (2021) Enhancement of elastin expression by transdermal administration of sialidase isozyme Neu2. Sci Rep 11:3302. https://doi.org/10.1038/s41598-021-82820-1
Sparavigna A (2020) Role of the extracellular matrix in skin aging and dedicated treatment - state of the art. Plast Aesthet Res 7:14. https://doi.org/10.20517/2347-9264.2019.73
Imokawa G, Ishida K (2015) Biological mechanisms underlying the ultraviolet radiation-induced formation of skin wrinkling and sagging I: Reduced skin elasticity, highly associated with enhanced eermal elastase activity, triggers wrinkling and sagging. Int J Mol Sci 16:7753–7775. https://doi.org/10.3390/ijms16047753
Tsukahara K, Takema Y, Moriwaki S, Tsuji N, Suzuki Y, Fujimura T, Imokawa G (2001) Selective inhibition of skin fibroblast elastase elicits a concentration-dependent prevention of ultraviolet B-induced wrinkle formation. J Investig Dermatol 117:671–677. https://doi.org/10.1046/j.0022-202x.2001.01450.x
Guo S, DiPietro LA (2010) Factors affecting wound healing. J Dent Res 89:219–229. https://doi.org/10.1177/0022034509359125
Makrantonaki E, Wlaschek M, Scharffetter-Kochanek K (2017) Pathogenesis of wound healing disorders in the elderly. J Dtsch Dermatol Ges 15:255–275. https://doi.org/10.1111/ddg.13199
Swain D, Gupta A (2015) Interfacial growth during closure of a cutaneous wound: stress generation and wrinkle formation. Soft Matter 11:6499–6508. https://doi.org/10.1039/c5sm01135c
Zucker S, Mancuso P, DiMassimo B, Lysik RM, Conner C, Wu CL (1994) Comparison of techniques for measurement of gelatinases/type IV collagenases: enzyme-linked immunoassays versus substrate degradation assays. Clin Exp Metast 12:13–23. https://doi.org/10.1007/bf01784329
Heo DH, Choi H, Yeom MY, Song BJ, Oh SJ (2014) Serum levels of matrix metalloproteinase-9 predict lymph nodemetastasis in breast cancer patients. Oncol Rep 31:1567–1572. https://doi.org/10.3892/or.2014.3001
Prescimone T, Tognotti D, Caselli C, Cabiati M, D’Amico A, Ry SD, Giannessi D (2014) Reappraisal of quantitative gelzymography for matrix metalloproteinases. J Clin Lab Anal 28:374–380. https://doi.org/10.1002/jcla.21696
Brenner M, Hearing VJ (2008) The protective role of melanin against UV damage in human skin. Photochem Photobiol 84:539–549. https://doi.org/10.1111/j.1751-1097.2007.00226.x
Cestari TF, Dantas LP, Boza JC (2014) Acquired hyperpigmentations. An Bras Dermatol 89:11–25. https://doi.org/10.1590/abd1806-4841.20142353
Stulberg DL, Clark N Tovey D (2003) Common hyperpigmentation disorders in adults: Part I. Diagnostic approach, Café au Lait Macules, diffuse hyperpigmentation, sun exposure, and phototoxic reactions. Am Fam Phys 68:1955–1960. https://pubmed.ncbi.nlm.nih.gov/14655804/
Stulberg DL, Clark N, Tovey D (2003) Common hyperpigmentation disorders in adults: Part II. Melanoma, seborrheic keratoses, acanthosis nigricans, melasma, diabetic dermopathy, tinea versicolor, and postinflammatory hyperpigmentation. Am Fam Phys 68:1963–1968. https://pubmed.ncbi.nlm.nih.gov/14655805/
Park J, Chung H, Bang SH, Han AR, Seo EK, Chang SE, Kang DH, Oh ES (2015) (E)-4-(3,4-Dimethoxyphenyl)but-3-en-1-ol enhances melanogenesis through increasing upstream stimulating factor-1-mediated tyrosinase expression. PLoS ONE 10: https://doi.org/10.1371/journal.pone.0141988
Adam RP (2007) Identification of essential oil components by Gas Chromatography/Mass Spectrometry, 4th edn. Allured Publishing Corporation, Illinois
Acknowledgments
This work was supported by the Research Fund 2018 [Grant number 12/2018] of the Research Institute, Rangsit University, Thailand, and partly supported by the Research, Development, and Engineering Fund for National Nanotechnology, National Science and Technology Development Agency (NSTDA), Thailand. This manuscript was proofread and edited by Cambridge Proofreading LLC under the support of the Research Institute of Rangsit University.
Author information
Authors and Affiliations
Contributions
AN funding acquisition, conceived the idea and design of the study, carried out the experimental studies and data analysis, wrote and final revised manuscript. BY participated in the conceptualization of the design, advised for statistical analysis, supervised the research project, prepared the sample extract, performed GC–MS analysis, and collected and interpreted data. RM participated in the conceptualization of the design and supervised the research project. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Navabhatra, A., Maniratanachote, R. & Yingngam, B. Antiphotoaging properties of Zingiber montanum essential oil isolated by solvent-free microwave extraction against ultraviolet B-irradiated human dermal fibroblasts. Toxicol Res. 38, 235–248 (2022). https://doi.org/10.1007/s43188-021-00107-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43188-021-00107-z