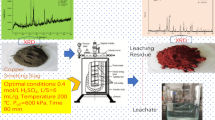
Abstract
This study investigated sulfuric acid leaching of Zn and/or Cu from brass ashes. The material consists of a heterogeneous powder containing ZnO, CuO, SiO2, Zn2SiO4, Zn, and Cu as main phases. Due to the strong exothermic behavior exhibited by changing operating conditions (L/S ratio = 3‒6 mL/g, pH 1‒5), temperature with time behavior was monitored to determine the heat released by leaching reactions. Eh–pH diagrams were drawn to identify regions to leach Zn selectively or simultaneously with Cu. A rich-Zn liquor (62 g/L) with low Cu (0.97 g/L) content was obtained using L/S ratio 3 mL/g and pH 5; a second leaching stage at pH 5 resulted in a liquor with only 10.2 g/L of Zn and 1.5 g/L of Cu. The low efficiency was attributed to the predominance of the refractory Zn2SiO4 phase. To increase leaching yield of Zn and Cu, the leaching residue was leached at pH 1 and L/S ratio = 3 mL/g, resulting in a liquor containing 90.5 g/L Zn and 14.4 g/L Cu. Quartz was refractory to H2SO4 in all tests, and silica gel formation was not observed. The acquired liquors must be submitted to adequate treatment to produce purified Zn and Cu solutions.
Graphical abstract














Similar content being viewed by others
References
Adhia JD (1969) Effect and control of impurities in electrolytic zinc production. In: Symposium on non-ferrous metals technology, V. III—Nickel, lead, zinc, rare earth and nuclear metals, Jamshedpur, pp 1‒10. http://eprints.nmlindia.org/3910
Agarwal JC, Flood HW, Beecher N, Sharma SN (1974) Preliminary economic analysis for hydrometallurgical processes. JOM 26(1):26–33. https://doi.org/10.1007/BF03355859
Ahmed IM, Nayl AA, Daoud JA (2016) Leaching and recovery of zinc and copper from brass slag by sulfuric acid. J Saudi Chem Soc 20(1):S280–S285. https://doi.org/10.1016/j.jscs.2012.11.003
Andrianne P, Scoyer J, Winand R (1980) Zinc electrowinning—a comparison of adherence-reducing pretreatments for aluminum cathode blanks. Hydrometallurgy 6(1–2):159–169. https://doi.org/10.1016/0304-386X(80)90015-8
Annoni R, Souza PS, Petrániková M, Miskufova A, Havlík T, Mansur MB (2013) Submerged-arc welding slags: characterization and leaching strategies for the removal of aluminum and titanium. J Hazard Mater 244–245:335–341. https://doi.org/10.1016/j.jhazmat.2012.11.053
Basir SMA, Rabah MA (1999) Hydrometallurgical recovery of metal values from brass melting slag. Hydrometallurgy 53(1):31–44. https://doi.org/10.1016/S0304-386X(99)00030-4
Brouwer D (2010) Smart processing of brass smelter residues. MSc thesis, Report nº AES/RE/10-39, Delft University of Technology, The Netherlands
Buarzaiga M (1999) An investigation of the failure mechanisms of aluminum cathodes in zinc electrowinning cells. PhD thesis, University of British Columbia, Vancouver, Canada. https://open.library.ubc.ca/soa/cIRcle/collections/ubctheses/831/items/1.0078757
Cheng CY (2000) Purification of synthetic laterite leach solution by solvent extraction using D2EHPA. Hydrometallurgy 56(3):369–386. https://doi.org/10.1016/S0304-386X(00)00095-5
Cheremisinoff NP (2001) Handbook of pollution prevention practices. CRC Press, Boca Raton. https://doi.org/10.1201/9781482270877
Cole PM, Sole KC (2003) Zinc solvent extraction in the process industries. Miner Process Extr Metall Rev 24(2):91–137. https://doi.org/10.1080/08827500306897
Conard BR (1992) The role of hydrometallurgy in achieving sustainable development. Hydrometallurgy 30(1–3):1–28. https://doi.org/10.1016/0304-386X(92)90074-A
Díaz G, Martín D (1994) Modified Zincex process: the clean, safe and profitable solution to the zinc secondaries treatment. Res Cons Recycl 10(1–2):43–57. https://doi.org/10.1016/0921-3449(94)90037-X
Flett DS (2005) Solvent extraction in hydrometallurgy: the role of organophosphorus extractants. J Organometallic Chem 690(10):2426–2438. https://doi.org/10.1016/j.jorganchem.2004.11.037
Fosnacht DR, O’Keefe TJ (1983) The effects of certain impurities and their interactions on zinc electrowinning. Metall Trans B 14:645–655. https://doi.org/10.1007/BF02653950
Gerlach RW, Dobb DE, Raab GA, Nocerino JM (2002) Gy sampling theory in environmental studies. 1. Assessing soil splitting protocols. J Chemom 16(7):321–328. https://doi.org/10.1002/cem.705
Grimes S, Donaldson J, Gomez GC (2008) Report on the environmental benefits of recycling. Bureau of International Recycling. https://www.mgg-recycling.com/wp-content/uploads/2013/06/BIR_CO2_report.pdf
Guimarães AS, Mansur MB (2018) Selection of a synergistic solvent extraction system to remove calcium and magnesium from concentrated nickel sulfate solutions. Hydrometallurgy 175:250–256. https://doi.org/10.1016/j.hydromet.2017.12.001
Guimarães AS, Silva PS, Mansur MB (2014) Purification of nickel from multicomponent aqueous sulfuric solutions by synergistic solvent extraction using Cyanex 272 and Versatic 10. Hydrometallurgy 150:173–177. https://doi.org/10.1016/j.hydromet.2014.10.005
Guimarães AS, Resende GPS, Santos ID, Vera YM, Mansur MB (2024) Evaluating cationic extractant systems containing Versatic 10 on the solvent extraction of metals from an Ni lateritic sulfuric solution. Sep Pur Technol 328:125060. https://doi.org/10.1016/j.seppur.2023.125060
Han JS, O’Keefe TJ (1992) Electrochemical evaluation of the adherence of zinc to aluminum cathodes. Surf Coat Technol 53(3):231–238. https://doi.org/10.1016/0257-8972(92)90381-J
Jha MK, Kumar V, Singh RJ (2001) Review of hydrometallurgical recovery of zinc from industrial wastes. Res Cons Recycl 33(1):1–22. https://doi.org/10.1016/S0921-3449(00)00095-1
Jiachao J, Youcai Z (2009) A process for the production of Zn powder by alkaline treatment of brass smelting ash at industrial scale. In: Proceedings of the 3rd international conference on bioinformatics and biomedical engineering, Beijing, China, pp 1‒4
Kahvecioglu Ö, Derin B, Yücel O (2003) Carbothermal recovery of zinc from brass ash. Min Process Extr Metall Trans Inst Min Metall Sect C 112(2):95–101. https://doi.org/10.1179/037195503225002790
Kazadi DM, Groot DR, Steenkamp JD, Pöllmann H (2016) Control of silica polymerisation during ferromanganese slag sulphuric acid digestion and water leaching. Hydrometallurgy 166:214–221. https://doi.org/10.1016/j.hydromet.2016.06.024
Khan NZ, Khan A, Shakoor A, Azam K (2015) Manufacturing defects of brass products and suggested remedies. IJISET 2(9):497–514
Kilicarslan A, Saridede MN (2015) Recovery of metallic values from brass waste using Brønsted acidic ionic liquid as leachate. JOM 67:2739–2746. https://doi.org/10.1007/s11837-015-1510-9
Kilicarslan A, Saridede MN, Stopic S, Friedrich B (2014) Use of ionic liquid in leaching process of brass wastes for copper and zinc recovery. Int J Min Metall Mater 21:138–143. https://doi.org/10.1007/s12613-014-0876-y
Kim Y, Lee J (2016) Leaching kinetics of zinc from metal oxide varistors (MOVs) with sulfuric acid. Metals 6(8):192. https://doi.org/10.3390/met6080192
Kukurugya F, Vindt T, Havlík T (2015) Behavior of zinc, iron and calcium from electric arc furnace (EAF) dust in hydrometallurgical processing in sulfuric acid solutions: thermodynamic and kinetic aspects. Hydrometallurgy 154:20–32. https://doi.org/10.1016/j.hydromet.2015.03.008
Lee TH, Joo SH, Nersisyan HH, Kong MS, Lee JW, Park KW, Lee JH (2016) Reduction kinetics of zinc powder from brass converter slag by pyrometallurgical method using hydrogen gas. KONA Powder Part J 33:278–286. https://doi.org/10.14356/kona.2016009
Mansur MB, Slater MJ, Biscaia EC Jr (2003) Reactive extraction of zinc sulfate with bis(2-ethylhexyl)phosphoric acid in a short Kühni column used in batch mode. Ind Eng Chem Res 42(17):4068–4076. https://doi.org/10.1021/ie020883y
Martins JM, Guimarães AS, Dutra AJB, Mansur MB (2020) Hydrometallurgical separation of zinc and copper from waste brass ashes using solvent extraction with D2EHPA. J Mater Res Technol 9(2):2319–2330. https://doi.org/10.1016/j.jmrt.2019.12.063
Martins JM, Dutra AJB, Mansur MB, Guimarães AS (2021) Comparison of oxidative roasting and alkaline leaching for removing chloride and fluoride from brass ashes. Hydrometallurgy 202:105619. https://doi.org/10.1016/j.hydromet.2021.105619
Matinde E, Simate GS, Ndlovu S (2018) Mining and metallurgical wastes: a review of recycling and re-use practices. J South Afr Inst Min Metall 118(8):825–844
Neto AP, Mansur MB (2013) Transient modeling of zinc extraction with D2EHPA in a Kühni column. Chem Eng Res Des 91(12):2323–2332. https://doi.org/10.1016/j.cherd.2013.05.014
Nicol MJ (2007) Electrowinning of base metals. A course presented to UFMG. Parker Center, Australia
Rudnik E (2019a) Recovery of zinc from zinc ash by leaching in sulphuric acid and electrowinning. Hydrometallurgy 188:256–263. https://doi.org/10.1016/j.hydromet.2019.07.006
Rudnik E (2019b) Investigation of industrial waste materials for hydrometallurgical recovery of zinc. Hydrometallurgy 139:105871. https://doi.org/10.1016/j.mineng.2019.105871
Santos LH, Carvalho PLG, Rodrigues GD, Mansur MB (2015) Selective removal of calcium from sulfate solutions containing magnesium and nickel using aqueous two-phase systems (ATPS). Hydrometallurgy 156:259–263. https://doi.org/10.1016/j.hydromet.2015.06.010
Silva MF, Oliveira MRS, Santos ID, Radino-Rouse P, Mansur MB (2022) Iron precipitation strategies from nickel laterite ore sulfuric acid leach liquor. Min Proc Extract Metall Rev 43(1):28–39. https://doi.org/10.1080/08827508.2020.1809392
Timur S, Gürmen S, Orhan G, Arslan C, Duman I (2000) Beneficiation of brass ashes. Dev Min Process 13:C12a-27-C12a−33. https://doi.org/10.1016/S0167-4528(00)80095-2
Tozawa K, Nishimura T, Akahori M, Malaga MA (1992) Comparison between purification processes for zinc leach solutions with arsenic and antimony trioxides. Hydrometallurgy 30:445–461. https://doi.org/10.1016/0304-386X(92)90099-L
Vilarinho C, Davim JP, Soares D, Castro F, Barbosa J (2005) Influence of the chemical composition on the machinability of brasses. J Mater Process Technol 170(1–2):441–447. https://doi.org/10.1016/j.jmatprotec.2005.05.035
Wang Q (2006) Industrial solid waste disposal and recycling, 4th edn. China Environmental Science Press, Beijing
Zhang Y, Hua Y, Gao X, Xu C, Li J, Li Y, Zhang Q, Xiong L, Su Z, Wang M, Ru J (2016) Recovery of zinc from a low-grade zinc oxide ore with high silicon by sulfuric acid curing and water leaching. Hydrometallurgy 166:16–21. https://doi.org/10.1016/j.hydromet.2016.08.01
Zhu Z, Cheng CY (2012) A study on zinc recovery from leach solutions using Ionquest 801 and its mixtures with D2EHPA. Miner Eng 39:117–123. https://doi.org/10.1016/j.mineng.2012.07.012
Acknowledgements
The authors acknowledge NEXA Resources, CAPES (finance code 001), and CNPq (processes 304018/2020-1 and 303998/2022-9) for financial support, and the Electron Microscopy Nucleus of PEMM/COPPE/UFRJ for their support with the use of microscopes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Martins, J.M., Guimarães, A.S., Dutra, A.J.B. et al. The evaluation of zinc and/or copper sulfuric acid leaching from brass ashes. Braz. J. Chem. Eng. (2024). https://doi.org/10.1007/s43153-024-00447-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s43153-024-00447-x