Abstract
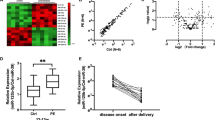
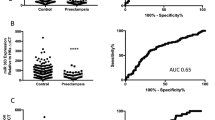
MicroRNA is associated with angiogenesis, invasion, proliferation, and vascular endothelial remodeling of various diseases. We aimed to investigate serum MicroRNA (miRNA) levels in preeclampsia (PE) and to determine whether any changes in miRNA levels are useful in predicting early onset preeclampsia (EOPE) and adverse perinatal outcomes. A total of 89 pregnant patients were enrolled in this prospective case–control study (55 PE and 34 healthy controls). miR-17, miR-20a, miR-20b, miR126, miR155, miR-200, miR-222, and miR-210 levels were studied in maternal serum in preeclamptic pregnant women. Multiple logistic regression analyses analyzed the risk factors which are associated with EOPE and adverse maternal outcomes. The Real-time RT-PCR method was used to determine maternal serum miRNA levels. Serum miR-17, miR-20a, miR-20b, miR126, and miR-210 levels were significantly higher in PE than the control group (p < .001, p < .001, p < .001, p < .001 and p = .047 respectively). Increased miR-17, miR-20a, and miR-20b levels were independently associated with PE (OR: 0.642, 95%Cl: 0.486–0.846, p = .002; OR: 0.899, 95%Cl: 0.811–0.996, p = .042 and OR: 0.817, 95%Cl: 0.689–0.970, p = .021). Increased miR-17 and miR-126 levels were negatively correlated with serum EOPE in PE (r = -.313, p = .020), and increased miR-210 levels were significantly positively correlated with EOPE in PE (r = .285, p = .005). Increased expression of serum miR-17, miR-20a, miR-20b, miR126, and miR-210 were found to be associated with PE, also increased expression of miR-17, miR-20a, and miR-20b were to be predicted with PE, also increased maternal serum miR-17 and miR-126 expressions were negatively correlated and increased miR-210 expression was positively correlated with EOPE in PE women.

Similar content being viewed by others
Data Availability
Data transparency.
Code Availability
Not applicable.
References
Ventura W, Koide K, Hori K, et al. Placental expression of microRNA-17 and -19b is down-regulated in early pregnancy loss. Eur J Obstet Gynecol Reprod Biol. 2013;169(1):28–32.
Flor I, Neumann A, Freter C, et al. Abundant expression and hemimethylation of C19MC in cell cultures from placenta-derived stromal cells. Biochem Biophys Res Commun. 2012;422(3):411–6.
Gao Y, She R, Wang Q, et al. Up-regulation of miR- 299 suppressed the invasion and migration of HTR-8/SVneo trophoblast cells partly via targeting HDAC2 in pre-eclampsia. Biomed Pharmacother. 2018;97:1222–8.
Bartel DP. Metazoan microRNAs. Cell. 2018;173(1):20–51.
Belgardt BF, Ahmed K, Spranger M, et al. The microRNA-200 family regulates pancreatic beta cell survival in type 2 diabetes. Nat Med. 2015;21(6):619–27.
Qu Y, Zhang H, Sun W, et al. MicroRNA-155 promotes gastric cancer growth and invasion by negatively regulating transforming growth factor-β receptor 2. Cancer Sci. 2018;109(3):618–28.
Pung KJ, Halberg N, Yoshida M, et al. A microRNA regulon that mediates endothelial recruitment and metastasis by cancer cells. Nature. 2012;481(190):194.
Wang W, Qu A, Liu W, et al. Circulating miR-210 as a diagnostic and prognostic biomarker for colorectal cancer. Eur J Cancer Care. 2017;26:e12448.
Zhou C, Zou Q, Li H, et al. Preeclampsia downregulates microRNAs in fetal endothelial cells: Roles of miR-29a/c-3p in endothelial function. J Clin Endocrinol Metab. 2017;102(9):3470–9.
Wang S, Olson EN. AngiomiRs— key regulators of angiogenesis. Curr Opin Genet Dev. 2017;19(205–211):15.
Wu F, Yang Z, Li G. Role of specific microRNAs for endothelial function and angiogenesis. Biochem Biophys Res Commun. 2009;386(4):549–53.
Wang W, Feng L, Zhang H, et al. Preeclampsia up‐regulates angiogenesis‐associated microRNA (i.e., miR‐17, −20a, and −20b) that target ephrin‐B2 and EPHB4 in the human placenta. J Clin Endocrinol Metab. 2012;97(6):E1051–9.
Wang Y, Zhang Y, Wang H, et al. Aberrantly up-regulated miR-20a in pre-eclampsic placenta compromised the proliferative and invasive behaviors of trophoblast cells by targeting forkhead box protein A1. Int J Biol Sci. 2014;10(9):973–82.
Liu B, Liu L, Cui S, et al. Expression and significance of microRNA-126 and VCAM-1 in placental tissues of women with early-onset preeclampsia. J Obstet Gynaecol Res. 2012;47(6):2042–50.
Luo R, Shao X, Xu P, et al. MicroRNA-210 contributes to preeclampsia by downregulating potassium channel modulatory factor 1. Hypertension. 2014;64(4):839–45.
Sibai B, Dekker G, Küfemin M. Pre-eclampsia. Lancet. 2005;365(9461):785–99.
Tranquilli AL, Brown MA, Zeeman GG, et al. The definition of severe and early-onset preeclampsia. Statements from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Pregnancy Hypertens. 2013;3(1):44–7.
Maynard SE, Min JY, Merchan J, et al. Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Invest. 2003;111(5):649–58.
Lycoudi A, Mavreli D, Mavrou A, et al. miRNAs in pregnancy-related complications. Expert Rev Mol Diagn. 2015;15:999–1010.
Hua Z, Lv Q, Ye W, et al. MiRNA-directed regulation of VEGF and other angiogenic factors under hypoxia. PLoS One. 2006;1(1):e116. https://doi.org/10.1371/journal.pone.0000116.
Dews M, Homayouni A, Yu D, et al. Augmentation of tumor angiogenesis by a Myc-activated microRNA cluster. Nat Genet. 2006;38(9):1060–5. https://doi.org/10.1038/ng1855.
Martin MM, Buckenberger JA, Jiang J, et al. The human angiotensin II type 1 receptor +1166 A/C polymorphism attenuates microRNA-155 binding. J Biol Chem. 2007;288(6):4227. https://doi.org/10.1074/jbc.A112.701050.
Fasanaro P, D’Alessandra Y, Di Stefano V, et al. MicroRNA-210 modulates endothelial cell response to hypoxia and inhibits the receptor tyrosine-kinase ligand Ephrin-A3. J Biol Chem. 2008;83(23):15878–83.
Harris TA, Yamakuchi M, Ferlito M, et al. MicroRNA-126 regulates endothelial expression of vascular cell adhesion molecule 1. Proc Natl Acad Sci U S A. 2008;105(5):1516–21. https://doi.org/10.1073/pnas.0707493105.
Tili E, Michaille JJ, Cimino A, et al. Modulation of miR-155 and miR-125b levels following lipopolysaccharide/TNF-alpha stimulation and their possible roles in regulating the response to endotoxin shock. J Immunol. 2007;179(8):5082–9. https://doi.org/10.4049/jimmunol.179.8.5082.
O’Connell RM, Rao DS, Chaudhuri AA, et al. Sustained expression of microRNA-155 in hematopoietic stem cells causes a myeloproliferative disorder. J Exp Med. 2008;205(3):585–94. https://doi.org/10.1084/jem.20072108.
Mistry HD, Gill CA, Kurlak LO, et al. Association between maternal micronutrient status, oxidative stress, and common genetic variants in antioxidant enzymes at 15 weeks gestation in nulliparous women who subsequently develop preeclampsia. Free Radic Biol Med. 2015;78:147–55.
Ali SM, Khalil RA. Genetic, immune, and vasoactive factors in the vascular dysfunction associated with hypertension in pregnancy. Expert Opin Ther Targets. 2015;19(11):1495–515.
Abdelazim SA, Shaker OG, Aly YAH, Senousy M. Uncovering serum placental-related non-coding RNAs as possible biomarkers of preeclampsia risk, onset, and severity revealed MALAT-1, miR-363 and miR-17. Sci Rep. 2022;12(1):1249.
Kumar P, Luo Y, Tudela C, et al. The c-Myc-regulated microRNA-17~92 (miR-17~92) and miR-106a~363 clusters target hCYP19A1 and hGCM1 to inhibit human trophoblast differentiation. Mol Cell Biol. 2013;33(9):1782–96.
Chen DB, Wang W. Human placental microRNAs, and preeclampsia. Biol Reprod. 2013;88(5):130.
Frazier S, McBride MW, Mulvana H, et al. From animal models to patients: the role of placental microRNAs, miR-210, miR-126, and miR-148a/152 in preeclampsia. Clin Sci (Lond). 2020;134(8):1001–25.
Hu TX, Wang G, Guo XJ, et al. MiR 20a,-20b, and -200c are involved in hydrogen sulfide stimulation of VEGF production in human placental trophoblasts. Placenta. 2016;39:101–10.
Hromadnikova I, Kotlabova K, Hympanova L, et al. Gestational hypertension, preeclampsia, and intrauterine growth restriction induce dysregulation of cardiovascular and cerebrovascular disease-associated microRNAs in maternal whole peripheral blood. Thromb Res. 2016;137:126–40.
Hromadnikova I, Kotlabova K, Dvorakova L, et al. Postpartum profiling of microRNAs involved in the pathogenesis of cardiovascular/cerebrovascular diseases in women exposed to pregnancy-related complications. Int J Cardiol. 2019;291:158–67.
Hromadnikova I, Kotlabova K, Krofta L. Cardiovascular Disease-Associated MicroRNAs as Novel Biomarkers of First-Trimester Screening for Gestational Diabetes Mellitus in the Absence of Other Pregnancy-Related Complications. Int J Mol Sci. 2022;23(18):10635.
Hromadnikova I, Kotlabova K, Krofta L. First-Trimester Screening for Fetal Growth Restriction and Small-for-Gestational-Age Pregnancies without Preeclampsia Using Cardiovascular Disease-Associated MicroRNA Biomarkers. Biomedicines. 2022;10(3):718.
Hromadnikova I, Kotlabova K, Hympanova L, et al. Cardiovascular and Cerebrovascular Disease Associated microRNAs Are Dysregulated in Placental Tissues Affected with Gestational Hypertension, Preeclampsia, and Intrauterine Growth Restriction. PLoS One. 2015;9:e0138383.
Staszel T, Zapała B, Polus A, et al. Role of microRNAs in endothelial cell pathophysiology. Pol Arch Med Wewn. 2011;121(10):361–6.
Howe CG, Foley HB, Kennedy EM, et al. Extracellular vesicle microRNA in early versus late pregnancy with birth outcomes in the MADRES study. Epigenetics. 2022;17(3):269–85.
Acknowledgements
The authors are thankful to the Selcuk University in Turkey for funding under the network scheme of the Project. (Ref no.2020/94).
Funding
This study was financially supported by Selcuk University.
Author information
Authors and Affiliations
Contributions
SO and AK conceived the idea; SO, MM, and MNA collected the data; EMO provided intellectual inputs; SO, AK, EMO, MM, and CC wrote.
Corresponding author
Ethics declarations
Ethics Approval
The study was approved by the Clinical Research Ethics Committee of Selcuk University in Turkey (approval date/ number: 19.02.2020/04).
Consent to participate and publication
Informed consent was obtained from all patients for being included in the study.
Conflicts of Interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ozler, S., Kebapcilar, A., Ozdemir, E.M. et al. Are Vascular Endothelium and Angiogenesis Effective MicroRNA Biomarkers Associated with the Prediction of Early-Onset Preeclampsia (EOPE) and Adverse Perinatal Outcomes?. Reprod. Sci. 31, 803–810 (2024). https://doi.org/10.1007/s43032-023-01367-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01367-w