Abstract
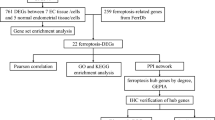
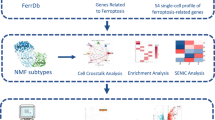
Ferroptosis, a recently discovered form of cell death, has been implicated in various diseases. However, the genetic relationship between ferroptosis and ovarian aging has not been thoroughly investigated through informatics analysis. In this study, we conducted bioinformatics analysis using ovarian aging and ferroptosis datasets to identify potential ferroptosis-related genes using R software. The expression levels of these genes at different ages were analyzed using the GTEx public database. To validate these findings at the genetic level, we performed clinical infertility biopsies. Bioinformatics analysis of a mouse ovary dataset revealed significantly higher expression of Tfrc, Ncoa4, and Slc3a2 in the aging group compared to the young group, while Gpx4 showed the opposite pattern. Consistent results were observed in biopsies from clinically aged infertile patients. This study is the first to identify a ferroptosis-related gene associated with ovarian aging, highlighting its potential as a diagnostic biomarker.




Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Finch CE, Holmes DJ. Ovarian aging in developmental and evolutionary contexts. Ann N Y Acad Sci. 2010;1204:82–94.
Giorgi C, Marchi S, Simoes ICM, Ren Z, Morciano G, Perrone M, Patalas-Krawczyk P, Borchard S, Jedrak P, Pierzynowska K, Szymanski J, Wang DQ, Portincasa P, Wegrzyn G, Zischka H, Dobrzyn P, Bonora M, Duszynski J, Rimessi A, Karkucinska-Wieckowska A, Dobrzyn A, Szabadkai G, Zavan B, Oliveira PJ, Sardao VA, Pinton P, Wieckowski MR. Mitochondria and reactive oxygen species in aging and age-related diseases. Int Rev Cell Mol Biol. 2018;340:209–344.
Li CJ, Lin LT, Tsai HW, Chern CU, Wen ZH, Wang PH, Tsui KH. The molecular regulation in the pathophysiology in ovarian aging. Aging Dis. 2021;12(3):934–49.
de Vet A, Laven JS, de Jong FH, Themmen AP, Fauser BC. Antimullerian hormone serum levels: a putative marker for ovarian aging. Fertil Steril. 2002;77(2):357–62.
Chen X, Yu C, Kang R, Tang D. Iron metabolism in ferroptosis. Front Cell Dev Biol. 2020;8:590226.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22(4):266–82.
Koppula P, Zhuang L, Gan B. Cystine transporter SLC7A11/xCT in cancer: ferroptosis, nutrient dependency, and cancer therapy. Protein Cell. 2021;12(8):599–620.
Ju J, Song YN, Wang K. Mechanism of ferroptosis: a potential target for cardiovascular diseases treatment. Aging Dis. 2021;12(1):261–76.
Gao H, Bai Y, Jia Y, Zhao Y, Kang R, Tang D, Dai E. Ferroptosis is a lysosomal cell death process. Biochem Biophys Res Commun. 2018;503(3):1550–6.
Shin D, Kim EH, Lee J, Roh JL. Nrf2 inhibition reverses resistance to GPX4 inhibitor-induced ferroptosis in head and neck cancer. Free Radic Biol Med. 2018;129:454–62.
Feng H, Schorpp K, Jin J, Yozwiak CE, Hoffstrom BG, Decker AM, Rajbhandari P, Stokes ME, Bender HG, Csuka JM, Upadhyayula PS, Canoll P, Uchida K, Soni RK, Hadian K, Stockwell BR. Transferrin receptor is a specific ferroptosis marker. Cell Rep. 2020;30(10):3411-3423 e7.
Maiorino M, Conrad M, Ursini F. GPx4, lipid peroxidation, and cell death: discoveries, rediscoveries, and open issues. Antioxid Redox Signal. 2018;29(1):61–74.
Sun L, Dong H, Zhang W, Wang N, Ni N, Bai X, Liu N. Lipid peroxidation, GSH depletion, and SLC7A11 inhibition are common causes of EMT and ferroptosis in A549 cells, but different in specific mechanisms. DNA Cell Biol. 2021;40(2):172–83.
Bannai S, Kitamura E. Transport interaction of L-cystine and L-glutamate in human diploid fibroblasts in culture. J Biol Chem. 1980;255(6):2372–6.
Xu X, Zhang X, Wei C, Zheng D, Lu X, Yang Y, Luo A, Zhang K, Duan X, Wang Y. Targeting SLC7A11 specifically suppresses the progression of colorectal cancer stem cells via inducing ferroptosis. Eur J Pharm Sci. 2020;152:105450.
Mancias JD, Wang X, Gygi SP, Harper JW, Kimmelman AC. Quantitative proteomics identifies NCOA4 as the cargo receptor mediating ferritinophagy. Nature. 2014;509(7498):105–9.
Santana-Codina N, Gikandi A, Mancias JD. The role of NCOA4-mediated ferritinophagy in ferroptosis. Adv Exp Med Biol. 2021;1301:41–57.
Gao M, Monian P, Pan Q, Zhang W, Xiang J, Jiang X. Ferroptosis is an autophagic cell death process. Cell Res. 2016;26(9):1021–32.
Li CJ, Chang CH, Tsang YL, Fang SH, Chen SN, Chiang AJ. Prognostic significance of ferroptosis pathway gene signature and correlation with macrophage infiltration in cervical squamous cell carcinoma. Int Immunopharmacol. 2022;112:109273.
Han X, Wang R, Zhou Y, Fei L, Sun H, Lai S, Saadatpour A, Zhou Z, Chen H, Ye F, Huang D, Xu Y, Huang W, Jiang M, Jiang X, Mao J, Chen Y, Lu C, Xie J, Fang Q, Wang Y, Yue R, Li T, Huang H, Orkin SH, Yuan GC, Chen M, Guo G. Mapping the mouse cell atlas by microwell-seq. Cell. 2018;173(5):1307.
Russ JE, Haywood ME, Lane SL, Schoolcraft WB, Katz-Jaffe MG. Spatially resolved transcriptomic profiling of ovarian aging in mice. iScience. 2022;25(8):104819.
Li CJ, Lin LT, Tsai HW, Wen ZH, Tsui KH. Phosphoglycerate mutase family member 5 maintains oocyte quality via mitochondrial dynamic rearrangement during aging. Aging Cell. 2022;21(2):e13546.
Hansen KR, Knowlton NS, Thyer AC, Charleston JS, Soules MR, Klein NA. A new model of reproductive aging: the decline in ovarian non-growing follicle number from birth to menopause. Hum Reprod. 2008;23(3):699–708.
Tsang YL, Kao CL, Lin SA, Li CJ. Mitochondrial dysfunction and oxidative stress in aging and disease. Biomedicines. 2022;10(11):2872.
Goldman KN. The quest for biomarkers linking ovarian aging and longevity. Fertil Steril. 2022;118(1):134–5.
Mazhar M, Din AU, Ali H, Yang G, Ren W, Wang L, Fan X, Yang S. Implication of ferroptosis in aging. Cell Death Discov. 2021;7(1):149.
Cao JY, Dixon SJ. Mechanisms of ferroptosis. Cell Mol Life Sci. 2016;73(11–12):2195–209.
Friedmann Angeli JP, Schneider M, Proneth B, Tyurina YY, Tyurin VA, Hammond VJ, Herbach N, Aichler M, Walch A, Eggenhofer E, Basavarajappa D, Radmark O, Kobayashi S, Seibt T, Beck H, Neff F, Esposito I, Wanke R, Forster H, Yefremova O, Heinrichmeyer M, Bornkamm GW, Geissler EK, Thomas SB, Stockwell BR, O’Donnell VB, Kagan VE, Schick JA, Conrad M. Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol. 2014;16(12):1180–91.
Ng SW, Norwitz SG, Norwitz ER. The impact of iron overload and ferroptosis on reproductive disorders in humans: implications for preeclampsia. Int J Mol Sci. 2019;20(13):3283.
Yureneva S, Averkova V, Silachev D, Donnikov A, Gavisova A, Serov V, Sukhikh G. Searching for female reproductive aging and longevity biomarkers. Aging (Albany NY). 2021;13(12):16873–94.
Ng SW, Norwitz SG, Taylor HS, Norwitz ER. Endometriosis: the role of iron overload and ferroptosis. Reprod Sci. 2020;27(7):1383–90.
Kelsey T. Models and biomarkers for ovarian ageing. Subcell Biochem. 2023;103:185–99.
Funding
This research was funded by the Ministry of Science Technology (MOST 111–2314-B-075B-014-MY3, 111–2314-B-075B-004-MY3), Kaohsiung Veterans General Hospital (VGHKS111-144, 111–145, 111–147, and 111-D06-1), and Yen Tjing Ling Medical Foundation (CI-111–16 and CI-112–10).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics Approval
This study was approved by the Institutional Review Board of the Kaohsiung Veterans General Hospital (KSVGH21-CT1-43). All procedures conducted in this study were in accordance with the principles stated in the Declaration of Helsinki.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Identification of ferroptosis-related genes as diagnostic markers for ovarian aging.
• Predictive value of ferroptosis-related genes in infertility patients.
• Clinical biopsy validates the link between ferroptosis-related genes and ovarian aging.
• Ferroptosis-related genes as potential biomarkers for ovarian aging.
• The role of ferroptosis-related genes in assessing ovarian aging through clinical biopsy.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, PH., Li, CJ., Lin, LT. et al. Unraveling the Clinical Relevance of Ferroptosis-Related Genes in Human Ovarian Aging. Reprod. Sci. 30, 3529–3536 (2023). https://doi.org/10.1007/s43032-023-01310-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01310-z