Abstract
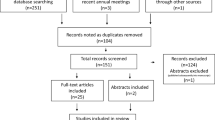
Autoimmune primary ovarian insufficiency (POI) is a devastating disease with limited clinical guidance. The objective of this systematic review was to identify treatments for autoimmune POI and analyze their efficacy. A comprehensive search of CINAHL, Cochrane, Embase, PubMed, Scopus, and Web of Science was performed from inception to April 2022. English language publications that evaluated women with autoimmune POI after a documented intervention were included. Animal models of autoimmune POI were also included. Risk of bias was assessed with the SYRCLE’s risk of bias tool for animal studies or the NIH Quality Assessment Tool for Case Series as appropriate. Twenty-eight studies were included in this review, with 11 RCTs, 15 case reports, and 2 case series. Seventeen studies were in humans, and 11 were in animal models. No completed RCTs, cohort studies, or case-control studies were identified in humans. In observational human studies, corticosteroids were effective in select patients. In many case reports, adequate treatment of comorbid autoimmune conditions resulted in return of menses, hormonal normalization, or spontaneous pregnancy. In terms of assisted reproductive technologies, there was case report evidence for both in vitro fertilization (IVF) and in vitro maturation (IVM) in women wishing to conceive with their own oocytes. Ovulation induction, IVF, and IVM resulted in a total of 15 pregnancies and 14 live births. In animal models, there was additional evidence for stem cell therapies and treatments used in traditional Chinese medicine, although this research may not be generalizable to humans. Furthermore, litter size was not evaluated in any of the animal studies. Additional research is needed to establish the efficacy of current treatments for autoimmune POI with a controlled experimental design and larger sample size. Additionally, there is a critical need to develop novel therapies for this condition, as understanding of its pathophysiology and available tools to modulate the immune response have progressed.

Similar content being viewed by others
Data Availability
The data that support the findings of this study were derived from the following resources available in the public domain: CINAHL, Cochrane, Embase, PubMed, Scopus, and Web of Science databases.
Code Availability
N/A
References
European Society for Human Reproduction and Embryology (ESHRE) Guideline Group on POI, Webber L, Davies M, et al. ESHRE guideline: management of women with premature ovarian insufficiency. Hum Reprod. 2016;31(5):926–37.
Conway GS, Kaltsas G, Patel A, Davies MC, Jacobs HS. Characterization of idiopathic premature ovarian failure. Fertil Steril. 1996;65(2):337–41.
Betterle C, Zanchetta R. Update on autoimmune polyendocrine syndromes (APS). Acta Biomed. 2003;74(1):9–33.
Reato G, Morlin L, Chen S, Furmaniak J, Rees Smith B, Masiero S, Albergoni P, Cervato S, Zanchetta R, Betterle C. Premature ovarian failure in patients with autoimmune Addison’s disease: clinical, genetic, and immunological evaluation. J Clin Endocrinol Metab. 2011;96(8):1255–61.
Grossmann B, Saur S, Rall K, et al. Prevalence of autoimmune disease in women with premature ovarian failure. Eur J Contracept Reprod Health Care. 2020;25(1):72–5.
Cao L, Liu W, Zhu Z. Clinical characteristics and relationship between myasthenia gravis and premature ovarian failure: report of two cases. J Int Med Res. 2019;47(8):3992–7.
Faubion SS, Kuhle CL, Shuster LT, Rocca WA. Long-term health consequences of premature or early menopause and considerations for management. Climacteric. 2015;18(4):483–91.
Singer D, Mann E, Hunter MS, Pitkin J, Panay N. The silent grief: psychosocial aspects of premature ovarian failure. Climacteric. 2011;14(4):428–37.
La Marca A, Marzotti S, Brozzetti A, et al. Primary ovarian insufficiency due to steroidogenic cell autoimmunity is associated with a preserved pool of functioning follicles. J Clin Endocrinol Metab. 2009;94(10):3816–23.
Falorni A, Brozzetti A, Aglietti MC, et al. Progressive decline of residual follicle pool after clinical diagnosis of autoimmune ovarian insufficiency. Clin Endocrinol (Oxf). 2012;77(3):453–8.
Bartmann AK, Silveira LDF, Silva LFI, et al. Autoimmune hypothyroidism and intermittent ovarian failure—case report. JBRA Assist Reprod. 2019;23(3):287–9.
Patel B, Haddad R, Saxena I, Gossain VV. Spontaneous long-term remission in a patient with premature ovarian failure. Endocr Pract. 2003;9(5):380–3.
Gama R, Wright J. Transient primary ovarian and thyroid failure. Ann Clin Biochem. 1997;34(Pt 2):208–9.
Taylor R, Smith NM, Angus B, Horne CH, Dunlop W. Return of fertility after twelve years of autoimmune ovarian failure. Clin Endocrinol (Oxf). 1989;31(3):305–8.
Bakalov VK, Anasti JN, Calis KA, et al. Autoimmune oophoritis as a mechanism of follicular dysfunction in women with 46,XX spontaneous premature ovarian failure. Fertil Steril. 2005;84(4):958–65.
Novosad JA, Kalantaridou SN, Tong ZB, Nelson LM. Ovarian antibodies as detected by indirect immunofluorescence are unreliable in the diagnosis of autoimmune premature ovarian failure: a controlled evaluation. BMC Womens Health. 2003;3(1):2.
Ishizuka B. Current understanding of the etiology, symptomatology, and treatment options in premature ovarian insufficiency (POI). Front Endocrinol (Lausanne). 2021;12:626924.
National Heart, Lung, and Blood Institute. Study Quality Assessment Tools. https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools. Accessed May 18, 2023.
Smith S, Hosid S. Premature ovarian failure associated with autoantibodies to the zona pellucida. Int J Fertil Menopausal Stud. 1994;39(6):316–9.
Rabinowe SL, Berger MJ, Welch WR, Dluhy RG. Lymphocyte dysfunction in autoimmune oophoritis. Resumption of menses with corticosteroids. Am J Med. 1986;81(2):347–50.
Blumenfeld Z, Halachmi S, Peretz BA, et al. Premature ovarian failure—the prognostic application of autoimmunity on conception after ovulation induction. Fertil Steril. 1993;59(4):750–5.
Kalantaridou SN, Braddock DT, Patronas NJ, Nelson LM. Treatment of autoimmune premature ovarian failure. Hum Reprod. 1999;14(7):1777–82.
Coulam CB, Kempers RD. Randall RV Premature ovarian failure: evidence for the autoimmune mechanism. Fertil Steril. 1981;36(2):238–40.
Finer N, Fogelman I, Bottazzo G. Pregnancy in a woman with premature ovarian failure. Postgrad Med J. 1985;61:1079–80.
Jung C, Paspaliaris W, Grill V, Lee D, Ward G. Reversible autoimmune thyroidiitis and oophoritis in a patient with Addison’s disease. Intern Med J. 2007;37(6):422–423.
Edmonds M, Lamki L, Killinger DW, Volpé R. Autoimmune thyroiditis, adrenalitis and oophoritis. Am J Med. 1973;54(6):782–7.
Cakir EDP, Ozdemir O, Eren E, Saglam H, Okan M, Tanm OF. Resolution of autoimmune oophoritis after thymectomy in a myasthenia gravis patient. J Clin Res Pediatr Endocrinol. 2011;3(4):212–5.
Bateman BG, Nunley WC Jr, Kitchin JD 3rd. Reversal of apparent premature ovarian failure in a patient with myasthenia gravis. Fertil Steril. 1983;39(1):108–10.
Ryan MM, Jones HR Jr. Myasthenia gravis and premature ovarian failure. Muscle Nerve. 2004;30(2):231–3.
Chang YS, Lai CC, Chen WS, Wang SH, Chou CT, Tsai CY. Protein-losing enteropathy and premature ovarian failure in a young woman with systemic lupus erythematosus. Lupus. 2012;21(11):1237–9.
Ferrau F, Gangemi S, Vita G, Trimarchi F, Cannavo S. Pregnancy after azathioprine therapy for ulcerative colitis in a woman with autoimmune premature ovarian failure and Addison’s disease: HLA haplotype characterization. Fertil Steril. 2011;95(7):2430.e17.
Rogenhofer N, Pavlik R, Jeschke U, Wypior G, Ochsenkühn R, Thaler CJ. Effective ovarian stimulation in a patient with resistant ovary syndrome and antigonadotrophin antibodies. Am J Reprod Immunol. 2015;73(2):185–91.
Virant-Klun I, Vogler A. In vitro maturation of oocytes from excised ovarian tissue in a patient with autoimmune ovarian insufficiency possibly associated with Epstein-Barr virus infection. Reprod Biol Endocrinol. 2018;16(1):33.
Chansel-Debordeaux L, Rault E, Depuydt C, et al. Successful live birth after in vitro maturation treatment in a patient with autoimmune premature ovarian failure: a case report and review of the literature. Gynecol Endocrinol. 2021;37(12):1138–42.
Grynberg M, Jacquesson L, Sifer C. In vitro maturation of oocytes for preserving fertility in autoimmune premature ovarian insufficiency. Fertil Steril. 2020;114(4):848–53.
Hooijmans CR, Rovers MM, de Vries RB, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE’s risk of bias tool for animal studies. BMC Med Res Methodol. 2014;26(14):43. https://doi.org/10.1186/1471-2288-14-43.
Chen S, Lu Y, Chen Y, et al. The effect of Bu Shen Huo Xue Tang on autoimmune premature ovarian insufficiency via modulation of the Nrf2/Keap1 signaling pathway in mice. J Ethnopharmacol. 2021;273:113996.
Wang P, Lu Y, Chen S, Chen Y, Hu C, Zuo Y. Protective function of Bu Shen Huo Xue formula on the immunity of B6AF1 mice with experimental autoimmune premature ovarian failure. Exp Ther Med. 2018;15(4):3302–10.
Chen H, Song L, Xu X, Han Z, Peng F, Zhang Q, Liu C, Liang X. The effect of icariin on autoimmune premature ovarian insufficiency via modulation of Nrf2/HO-1/Sirt-1 pathay in mice. Reprod Biol. 2022;22:100638.
Ding Q, Wang Y, Li N, et al. Bidirectional estrogen-like effects of genistein on murine experimental autoimmune ovarian disease. Int J Mol Sci. 2016;17(11):1855.
He X, Wang SY, Yin CH, Wang T, Jia CW, Ma YM. Hydrogen-rich water exerting a protective effect on ovarian reserve function in a mouse model of immune premature ovarian failure induced by zona pellucida 3. Chin Med J (Engl). 2016;129(19):2331–7.
Berberian V, Sánchez S, Sánchez-Borzone M, Attademo AM, Lasaga M, Celis ME. Effect of alpha-melanotropin hormone on serum levels of luteinizing hormone and progesterone in experimental rat autoimmune oophoritis. Peptides. 2006;27(9):2295–9.
Li H, Zhao W, Wang L, et al. Human placenta-derived mesenchymal stem cells inhibit apoptosis of granulosa cells induced by IRE1α pathway in autoimmune POF mice. Cell Biol Int. 2019;43(8):899–909.
Lu X, Cui J, Cui L, et al. The effects of human umbilical cord-derived mesenchymal stem cell transplantation on endometrial receptivity are associated with Th1/Th2 balance change and uNK cell expression of uterine in autoimmune premature ovarian failure mice. Stem Cell Res Ther. 2019;10(1):214.
Yin N, Wang Y, Lu X, et al. hPMSC transplantation restoring ovarian function in premature ovarian failure mice is associated with change of Th17/Tc17 and Th17/Treg cell ratios through the PI3K/Akt signal pathway. Stem Cell Res Ther. 2018;9(1):37.
Yin N, Zhao W, Luo Q, Yuan W, Luan X, Zhang H. Restoring ovarian function with human placenta-derived mesenchymal stem cells in autoimmune-induced premature ovarian failure mice mediated by Treg cells and associated cytokines. Reprod Sci. 2018;25(7):1073–82.
Wang Z, Wei Q, Wang H, et al. Mesenchymal stem cell therapy using human umbilical cord in a rat model of autoimmune-induced premature ovarian failure. Stem Cells Int. 2020;2020:3249495.
Lim J, Ortiz L, Nakamura BN, et al. Effects of deletion of the transcription factor Nrf2 and benzo [a]pyrene treatment on ovarian follicles and ovarian surface epithelial cells in mice. Reprod Toxicol. 2015;58:24–32.
Hu X, Roberts JR, Apopa PL, Kan YW, Ma Q. Accelerated ovarian failure induced by 4-vinyl cyclohexene diepoxide in Nrf2 null mice. Mol Cell Biol. 2006;26(3):940–54.
Brozzetti A, Marzotti S, La Torre D, et al. Autoantibody responses in autoimmune ovarian insufficiency and in Addison's disease are IgG1 dominated and suggest a predominant, but not exclusive, Th1 type of response. Eur J Endocrinol. 2010;163(2):309–17.
Kobayashi M, Nakashima A, Yoshino O, et al. Decreased effector regulatory T cells and increased activated CD4+ T cells in premature ovarian insufficiency. Am J Reprod Immunol. 2019;81(6):e13125.51.
Kawamura K, Cheng Y, Suzuki N, Deguchi M, Sato Y, Takae S, Ho CH, Kawamura N, Tamura M, Hashimoto S, Sugishita Y, Morimoto Y, Hosoi Y, Yoshioka N, Ishizuka B, Hsueh AJ. Hippo signaling disruption and Akt stimulation of ovarian follicles for infertility treatment. Proc Natl Acad Sci U S A. 2013;110(43):17474–9.
Funding
This research was supported in part by the Howard and Georgeanna Seegar Jones Endowment to JHS.
Author information
Authors and Affiliations
Contributions
JS conceived of the idea and supervised the findings of this work and edited the manuscript. EN searched databases for relevant publications. EL interpreted the data and wrote the manuscript with help from BS. All authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
N/A
Consent to Participate
N/A
Consent for Publication
N/A
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1 Search terms
Database | Search statements |
|---|---|
CINAHL Plus with Full Text (EBSCOhost) | TI ( (autoantibody OR autoimmune OR autoimmunity) AND ("ovarian insufficiency" OR "ovarian failure") ) OR AB ( (autoantibody OR autoimmune OR autoimmunity) AND ("ovarian insufficiency" OR "ovarian failure") ) OR SU ( (autoantibody OR autoimmune OR autoimmunity) AND ("ovarian insufficiency" OR "ovarian failure") ) OR TI ( "inflammatio ovarica" OR "inflammation of the ovary" OR "inflammation of the ovaries" OR "gonadotropin resistant ovary*" OR "gonadotropin-resistant ovary*" OR "gonadotropin resistant ovaries*" OR "gonadotropin-resistant ovaries*" OR "oophoritides" OR "oophroritis" OR "ovarial inflammation*" OR "ovarian autoimmune disease" OR "ovaritis" OR "ovary inflammation*" OR "resistance ovary syndrome*" OR "resistant ovary*" OR "resistant ovaries*" ) OR AB ( "inflammatio ovarica" OR "inflammation of the ovary" OR "inflammation of the ovaries" OR "gonadotropin resistant ovary*" OR "gonadotropin-resistant ovary*" OR "gonadotropin resistant ovaries*" OR "gonadotropin-resistant ovaries*" OR "oophoritides" OR "oophroritis" OR "ovarial inflammation*" OR "ovarian autoimmune disease" OR "ovaritis" OR "ovary inflammation*" OR "resistance ovary syndrome*" OR "resistant ovary*" OR "resistant ovaries*" ) OR SU ( "inflammatio ovarica" OR "inflammation of the ovary" OR "inflammation of the ovaries" OR "gonadotropin resistant ovary*" OR "gonadotropin-resistant ovary*" OR "gonadotropin resistant ovaries*" OR "gonadotropin-resistant ovaries*" OR "oophoritides" OR "oophroritis" OR "ovarial inflammation*" OR "ovarian autoimmune disease" OR "ovaritis" OR "ovary inflammation*" OR "resistance ovary syndrome*" OR "resistant ovary*" OR "resistant ovaries*" ) |
Cochrane Library (Wiley) | [mh Oophoritis] OR ((autoantibody OR autoimmune OR autoimmunity) AND ("ovarian insufficiency" OR "ovarian failure")):ti,ab,kw OR ("inflammatio ovarica" OR "inflammation of the ovary" OR "inflammation of the ovaries" OR "gonadotropin resistant ovary*" OR "gonadotropin-resistant ovary*" OR "gonadotropin resistant ovaries*" OR "gonadotropin-resistant ovaries*" OR "oophoritides" OR "oophroritis" OR "ovarial inflammation*" OR "ovarian autoimmune disease" OR "ovaritis" OR "ovary inflammation*" OR "resistance ovary syndrome*" OR "resistant ovary*" OR "resistant ovaries*"):ti,ab,kw |
Embase (Elsevier) | 'ovary inflammation'/de OR ((autoantibody:ti,ab,kw OR autoimmune:ti,ab,kw OR autoimmunity:ti,ab,kw) AND ('ovarian insufficiency':ti,ab,kw OR 'ovarian failure':ti,ab,kw)) OR 'inflammatio ovarica':ti,ab,kw OR 'inflammation of the ovary':ti,ab,kw OR 'inflammation of the ovaries':ti,ab,kw OR 'gonadotropin resistant ovary*':ti,ab,kw OR 'gonadotropin-resistant ovary*':ti,ab,kw OR 'gonadotropin resistant ovaries*':ti,ab,kw OR 'gonadotropin-resistant ovaries*':ti,ab,kw OR 'oophoritides':ti,ab,kw OR 'oophroritis':ti,ab,kw OR 'ovarial inflammation*':ti,ab,kw OR 'ovarian autoimmune disease':ti,ab,kw OR 'ovaritis':ti,ab,kw OR 'ovary inflammation*':ti,ab,kw OR 'resistance ovary syndrome*':ti,ab,kw OR 'resistant ovary*':ti,ab,kw OR 'resistant ovaries*':ti,ab,kw |
PubMed | "Autoimmune oophoritis" [Supplementary Concept] OR "Oophoritis"[Mesh] OR ((autoantibody[Text Word] OR autoimmune[Text Word] OR autoimmunity[Text Word]) AND ("ovarian insufficiency"[Text Word] OR "ovarian failure"[Text Word])) OR "inflammatio ovarica"[Text Word] OR "inflammation of the ovary"[Text Word] OR "inflammation of the ovaries"[Text Word] OR "gonadotropin resistant ovary*"[Text Word] OR "gonadotropin-resistant ovary*"[Text Word] OR "gonadotropin resistant ovaries*"[Text Word] OR "gonadotropin-resistant ovaries*"[Text Word] OR "oophoritides"[Text Word] OR "oophroritis"[Text Word] OR "ovarial inflammation*"[Text Word] OR "ovarian autoimmune disease"[Text Word] OR "ovaritis"[Text Word] OR "ovary inflammation*"[Text Word] OR "resistance ovary syndrome*"[Text Word] OR "resistant ovary*"[Text Word] OR "resistant ovaries*"[Text Word] |
Scopus (Elsevier) | TITLE-ABS-KEY ( ( autoantibody OR autoimmune OR autoimmunity ) AND ( "ovarian insufficiency" OR "ovarian failure" ) ) OR TITLE-ABS-KEY ( "inflammatio ovarica" OR "inflammation of the ovary" OR "inflammation of the ovaries" OR "gonadotropin resistant ovary*" OR "gonadotropin-resistant ovary*" OR "gonadotropin resistant ovaries*" OR "gonadotropin-resistant ovaries*" OR "oophoritides" OR "oophroritis" OR "ovarial inflammation*" OR "ovarian autoimmune disease" OR "ovaritis" OR "ovary inflammation*" OR "resistance ovary syndrome*" OR "resistant ovary*" OR "resistant ovaries*" ) |
Web of Science Core Collection (Clarivate) | ts=(((autoantibody or autoimmune or autoimmunity) and ("ovarian insufficiency" or "ovarian failure")) or "inflammatio ovarica" or "inflammation of the ovary" or "inflammation of the ovaries" or "gonadotropin resistant ovary*" or "gonadotropin-resistant ovary*" or "gonadotropin resistant ovaries*" or "gonadotropin-resistant ovaries*" or "oophoritides" or "oophroritis" or "ovarial inflammation*" or "ovarian autoimmune disease" or "ovaritis" or "ovary inflammation*" or "resistance ovary syndrome*" or "resistant ovary*" or "resistant ovaries*") |
Appendix 2 Criteria for inclusion and exclusion of studies
Inclusion criteria | Exclusion criteria | |
|---|---|---|
Population | Women with diagnosis of autoimmune oophoritis or autoimmune primary ovarian insufficiency (POI). Animal models of autoimmune oophoritis. | Non-autoimmune etiologies of POI. Cellular/in vitro studies. |
Diagnosis specificity | Patients have clinical diagnosis of autoimmune oophoritis or autoimmune POI | Study includes patients with other causes of POI |
Treatment | Documented treatment intervention | No documented treatment intervention |
Results | Documented clinical or experimental outcome after treatment intervention | No documented clinical or experimental outcome after treatment intervention |
Study design | Randomized controlled trials, observational studies | Literature reviews, conference proceedings, unpublished data |
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Levit, E., Singh, B., Nylander, E. et al. A Systematic Review of Autoimmune Oophoritis Therapies. Reprod. Sci. 31, 1–16 (2024). https://doi.org/10.1007/s43032-023-01299-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01299-5