Abstract
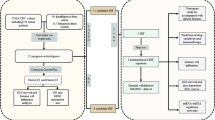
Cervical cancer (CC) remains a prevalent gynecological malignancy, posing a significant health burden among women worldwide. With the remarkable discoveries of cellular pyroptosis and cuproptosis, there has been a growing focus on exploring the intricate relationship between these two forms of cell death and their impact on tumor progression. In recent years, alternative splicing has emerged as a significant field in cancer research. Thus, the integration of alternative splicing, pyroptosis, and cuproptosis holds immense value in studying their collective impact on the occurrence and progression of cervical cancer. In this study, alternative splicing data of pyroptosis- and cuproptosis-associated genes were integrated with public databases, including TCGA, to establish a prognostic model for cervical cancer based on COX regression modeling. Subsequently, the tumor microenvironment (TME) phenotypes in the high-risk and low-risk patient groups were characterized through a comprehensive bioinformatics analysis. The findings of this study revealed that the low-risk group exhibited a predominant immune-active TME phenotype, while the high-risk group displayed a tumor-favoring metabolic phenotype. These results indicate that the alternative splicing of pyroptosis- and cuproptosis-associated genes plays a pivotal role in remodeling the phenotypic landscape of the cervical cancer TME by modulating immune responses and metabolic pathways. This study provides valuable insights into the interplay between alternative splicing variants involved in pyroptosis and cuproptosis and the TME, contributing to a deeper understanding of cervical cancer pathogenesis and potential therapeutic avenues.






Similar content being viewed by others
Data Availability
The data used in this study could be accessed from freely public-accessible database including databases of TCGA (https://portal.gdc.cancer.gov/), Molecular Signatures (https://www.gsea-msigdb.org/gsea/msigdb/), and TCGA SpliceSeq (https://bioinformatics.mdanderson.org/TCGASpliceSeq/).
Code Availability
Raw data and codes are available on request.
References
Fernandes A, et al. Human papillomavirus-independent cervical cancer. Int J Gynecol Cancer. 2022;32(1):1–7.
Ferrall L, et al. Cervical cancer immunotherapy: facts and hopes. Clin Cancer Res. 2021;27(18):4953–73.
Ge EJ, et al. Connecting copper and cancer: from transition metal signalling to metalloplasia. Nat Rev Cancer. 2022;22(2):102–13.
Song Y, et al. HPV E7 inhibits cell pyroptosis by promoting TRIM21-mediated degradation and ubiquitination of the IFI16 inflammasome. Int J Biol Sci. 2020;16(15):2924–37.
Li L, et al. Pyroptosis, a new bridge to tumor immunity. Cancer Sci. 2021;112(10):3979–94.
Lee C, et al. Inflammasome as a promising therapeutic target for cancer. Life Sci. 2019;231:116593.
Tong X, et al. Targeting cell death pathways for cancer therapy: recent developments in necroptosis, pyroptosis, ferroptosis, and cuproptosis research. J Hematol Oncol. 2022;15(1):174.
Zhou C, et al. Identification of pyroptosis-related signature for cervical cancer predicting prognosis. Aging (Albany NY). 2021;13(22):24795–814.
Sharma BR, Kanneganti TD. NLRP3 inflammasome in cancer and metabolic diseases. Nat Immunol. 2021;22(5):550–9.
Hamarsheh S, Zeiser R. NLRP3 inflammasome activation in cancer: a double-edged sword. Front Immunol. 2020;11:1444.
Dey Sarkar R, Sinha S, Biswas N. Manipulation of inflammasome: a promising approach towards immunotherapy of lung cancer. Int Rev Immunol. 2021;40(3):171–82.
Yu SY, Li XL. Pyroptosis and inflammasomes in obstetrical and gynecological diseases. Gynecol Endocrinol. 2021;37(5):385–91.
Huang Y, Li R, Yang Y. Role of pyroptosis in gynecological oncology and its therapeutic regulation. Biomolecules. 2022;12(7):924.
Lei L, Tan L, Sui L. A novel cuproptosis-related gene signature for predicting prognosis in cervical cancer. Front Genet. 2022;13:957744.
Chen SJ, et al. Mechanistic basis of a combination D-penicillamine and platinum drugs synergistically inhibits tumor growth in oxaliplatin-resistant human cervical cancer cells in vitro and in vivo. Biochem Pharmacol. 2015;95(1):28–37.
Lelièvre P, et al. The multifaceted roles of copper in cancer: a trace metal element with dysregulated metabolism, but also a target or a bullet for therapy. Cancers (Basel). 2020;12(12):3594.
Bandmann O, Weiss KH, Kaler SG. Wilson’s disease and other neurological copper disorders. Lancet Neurol. 2015;14(1):103–13.
Zhang M, Shi M, Zhao Y. Association between serum copper levels and cervical cancer risk: a meta-analysis. Biosci Rep 2018;38(4):BSR20180161
Tsvetkov P, et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science. 2022;375(6586):1254–61.
Zhang Z, et al. Cuproptosis-related risk score predicts prognosis and characterizes the tumor microenvironment in hepatocellular carcinoma. Front Immunol. 2022;13:925618.
Bao JH, et al. Identification of a novel cuproptosis-related gene signature and integrative analyses in patients with lower-grade gliomas. Front Immunol. 2022;13:933973.
Xu S, et al. Cuproptosis-associated lncRNA establishes new prognostic profile and predicts immunotherapy response in clear cell renal cell carcinoma. Front Genet. 2022;13:938259.
Voli F, et al. Intratumoral copper modulates PD-L1 expression and influences tumor immune evasion. Cancer Res. 2020;80(19):4129–44.
Wang ET, et al. Alternative isoform regulation in human tissue transcriptomes. Nature. 2008;456(7221):470–6.
Keren H, Lev-Maor G, Ast G. Alternative splicing and evolution: diversification, exon definition and function. Nat Rev Genet. 2010;11(5):345–55.
Oltra SS, et al. Distinct GSDMB protein isoforms and protease cleavage processes differentially control pyroptotic cell death and mitochondrial damage in cancer cells. Cell Death Differ. 2023;30(5):1366–81.
Gupta A, Lutsenko S. Human copper transporters: mechanism, role in human diseases and therapeutic potential. Future Med Chem. 2009;1(6):1125–42.
Ouyang D, et al. Comprehensive analysis of prognostic alternative splicing signature in cervical cancer. Cancer Cell Int. 2020;20:221.
Shao XY, et al. Prognostic value and potential role of alternative mRNA splicing events in cervical cancer. Front Genet. 2020;11:726.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
The databases of TCGA and websites used in this work are open free to the public, the data of patients deposited in TCGA have obtained ethical approval, and this study is based on open-source data, so there are no ethical issues and other conflicts of interest.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information

Supplementary Figure 1
Correlation analysis of tumor-infiltrating immune cells (TICs) and risk score. (PNG 380 kb)

Supplementary Figure 2
Correlation analysis of immune checkpoint-associated genes and risk score. (PNG 683 kb)
ESM 3
(DOCX 17 kb)
ESM 4
(XLSX 9 kb)
ESM 5
(XLSX 13 kb)
ESM 6
(XLSX 35 kb)
ESM 7
(XLSX 10 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bi, K., Yang, J. & Wei, X. Alternative splicing variants involved in pyroptosis and cuproptosis contribute to phenotypic remodeling of the tumor microenvironment in cervical cancer. Reprod. Sci. 30, 3648–3660 (2023). https://doi.org/10.1007/s43032-023-01284-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01284-y