Abstract
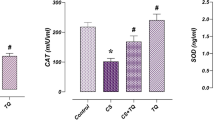
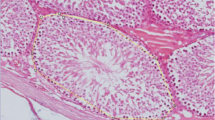
Cancer is one of the world’s major causes of death. The aim of this study is to examine the acute effects of resveratrol on testicular toxicity, oxidative stress, and apoptosis caused by MTX, which is widely used in the treatment of many diseases, especially cancer, histochemically, immunohistochemically, and biochemical methods using different parameters. A total of 32 Wistar albino male rats were randomly divided into 4 groups: control, resveratrol (RES), MTX, and MTX + RES, with 8 animals in each group. At the end of the experiment, tissue and blood samples were taken, and histochemical, immunohistochemical, and biochemical parameters were examined. In this study, where parameters were compared for the first time, total thiol (TT) and native thiol (NT) are the highest in the RES group, disulfide (DS), and ischemia-modified albumin (IMA) are the highest in the MTX group. Total oxidant status (TOS) and oxidative stress index (OSI) are the highest in the MTX group, and total antioxidant status (TAS) is the highest in the RES group. Separation and deterioration in the tunica albuginea, congestion and edema in the interstitial region, vacuolization in the seminiferous epithelium, and spermatogenic serial cells spilling into the lumen without completing their maturation were observed. When examined in terms of histochemical, immunohistochemical, and biochemical examinations, our study revealed that resveratrol has positive effects on methotrexate-induced acute testicular damage, oxidative stress, and apoptosis.



Similar content being viewed by others
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Baykara O. Current modalities in treatment of cancer. Balıkesır Heal Sci J. 2016;5(3):154–65. https://doi.org/10.5505/bsbd.2016.93823.
Mian M, Tinelli M, De March E, Turri G, Meneghini V, Pescosta N, et al. Bortezomib, thalidomide and lenalidomide: have they really changed the outcome of multiple myeloma? Anticancer Res. 2016;36(3):1059–65.
Madhu P, Reddy KP, Reddy PS. Role of melatonin in mitigating chemotherapy-induced testicular dysfunction in Wistar rats. Drug Chem Toxicol. 2016;39(2):137–46. https://doi.org/10.3109/01480545.2015.1055359.
Haghi-Aminjan H, Asghari MH, Farhood B, Rahimifard M, Hashemi Goradel N, Abdollahi M. The role of melatonin on chemotherapy-induced reproductive toxicity. J Pharm Pharmacol. 2018;70(3):291–306. https://doi.org/10.1111/jphp.12855.
Bedoui Y, Guillot X, Sélambarom J, Guiraud P, Giry C, Jaffar-Bandjee MC, et al. Methotrexate an old drug with new tricks. Int J Mol Sci. 2019;20(20):5023. https://doi.org/10.3390/ijms20205023.
Chan ESL, Cronstein BN. Mechanisms of action of methotrexate. Bull Hosp Jt Dis. 2013;71(Suppl 1):S5-8.
Scully CJ, Anderson CJ, Cannon GW. Long-term methotrexate therapy for rheumatoid arthritis. Semin Arthritis Rheum. 1991;20(5):317–31. https://doi.org/10.1016/0049-0172(91)90032-u.
Elango T, Thirupathi A, Subramanian S, Ethiraj P, Dayalan H, Gnanaraj P. Methotrexate treatment provokes apoptosis of proliferating keratinocyte in psoriasis patients. Clin Exp Med. 2017;17(3):371–81. https://doi.org/10.1007/s10238-016-0431-4.
Bath RK, Brar NK, Forouhar FA, Wu GY. A review of methotrexate-associated hepatotoxicity. J Dig Dis. 2014;15(10):517–24. https://doi.org/10.1111/1751-2980.12184.
Nouri HS, Azarmi Y, Movahedin M. Effect of growth hormone on testicular dysfunction induced by methotrexate in rats. Andrologia. 2009;41(2):105–10. https://doi.org/10.1111/j.1439-0272.2008.00897.x.
Gulgun M, Erdem O, Oztas E, Kesik V, Balamtekin N, Vurucu S, et al. Proanthocyanidin prevents methotrexate-induced intestinal damage and oxidative stress. Exp Toxicol Pathol. 2010;62(2):109–15. https://doi.org/10.1016/j.etp.2009.02.120.
Vardi N, Parlakpinar H, Ates B, Cetin A, Otlu A. Antiapoptotic and antioxidant effects of beta-carotene against methotrexate-induced testicular injury. Fertil Steril. 2009;92(6):2028–33. https://doi.org/10.1016/j.fertnstert.2008.09.015.
Shin S, Jeon JH, Park D, Jang M-J, Choi JH, Choi B-H, et al. Trans-resveratrol relaxes the corpus cavernosum ex vivo and enhances testosterone levels and sperm quality in vivo. Arch Pharm Res. 2008;31(1):83–7. https://doi.org/10.1007/s12272-008-1124-7.
Juan ME, González-Pons E, Munuera T, Ballester J, Rodríguez-Gil JE, Planas JM. Trans-resveratrol, a natural antioxidant from grapes, increases sperm output in healthy rats. J Nutr. 2005;135(4):757–60. https://doi.org/10.1093/jn/135.4.757.
Revel A, Raanani H, Younglai E, Xu J, Han R, Savouret JF, et al. Resveratrol, a natural aryl hydrocarbon receptor antagonist, protects sperm from DNA damage and apoptosis caused by benzo(a)pyrene. Reprod Toxicol. 2001;15(5):479–86. https://doi.org/10.1016/s0890-6238(01)00149-6.
Jiang Y, Peng T, Luo Y, Li MLY. Resveratrol reestablishes spermatogenesis after testicular injury in rats caused by 2, 5-hexanedione. Chin Med J. 2008;121(13):1204–9.
Yulug E, Turedi S, Alver A, Turedi S, Kahraman C. Effects of resveratrol on methotrexate-ınduced testicular damage in rats. Bratislava Med J. 2015;116(11):676–80. https://doi.org/10.4149/bll_2015_132.
Leonard SS, Xia C, Jiang BH, Stinefelt B, Klandorf H, Harris GK, et al. Resveratrol scavenges reactive oxygen species and effects radical-induced cellular responses. Biochem Biophys Res Commun. 2003Oct 3;309(4):1017–26. https://doi.org/10.1016/j.bbrc.2003.08.105.
Savas HB, Gultekin FCI. Positive effects of meal frequency and calorie restriction on antioxidant systems in rats. North Clin Istanb. 2017;26(4):109–16. https://doi.org/10.14744/nci.2017.21548.
Abdel-Wahhab MA, Nada SA, Arbid MS. Ochratoxicosis: prevention of developmental toxicity by L-methionine in rats. J Appl Toxicol. 1999;19(1):7–12. https://doi.org/10.1002/(sici)1099-1263(199901/02)19:1%3c7::aid-jat529%3e3.0.co;2-g.
Buyukozturk S. Sosyal Bilimler için Veri Analizi El Kitabı İstatistik, Araştırma Deseni SPSS Uygulamaları ve Yorum. 28th ed. Ankara: Pegem Akademi; 2012. 210 p.
Shapiro S, Wilk M. An analysis of variance test for normality (complete samples). Biometrika. 1965;52(3–4):591–611. https://doi.org/10.2307/2333709.
Argyriou AA, Bruna J, Marmiroli P, Cavaletti G. Chemotherapy-induced peripheral neurotoxicity (CIPN): an update. Crit Rev Oncol Hematol. 2012;82(1):51–77. https://doi.org/10.1016/j.critrevonc.2011.04.012.
Blumenfeld Z. Chemotherapy and fertility. Best Pr Res Clin Obs Gynaecol. 2012;26(3):379–90. https://doi.org/10.1016/j.bpobgyn.2011.11.008.
Shrestha S, Dhungel S, Saxena AK, Bhattacharya S, Maskey D. Effect of methotrexate (MTX) administration on spermatogenesis: an experimental on animal model. Nepal Med Coll J. 2007;9(4):230–3.
Padmanabhan S, Tripathi DN, Vikram A, Ramarao P, Jena GB. Cytotoxic and genotoxic effects of methotrexate in germ cells of male Swiss mice. Mutat Res - Genet Toxicol Environ Mutagen. 2008;655(1–2):59–67. https://doi.org/10.1016/j.mrgentox.2008.07.003.
Daggulli M, Dede O, Utangac MM, Bodakci MN, Hatipoglu NK, Penbegul N, et al. Protective effects of carvacrol against methotrexate-induced testicular toxicity in rats. Int J Clin Exp Med. 2014;7(12):5511–6.
Karbandi S, Momenizadeh A, Hydarzadeh M, Mazlom R, Hasanzadeh M. The effect of mother empowerment program on mothers’ attachment to their hospitalized premature neonates. Evid Based Care. 2015;5(2):7–14.
León J, Acuña-Castroviejo D, Escames G, Tan DX, Reiter RJ. Melatonin mitigates mitochondrial malfunction. J Pineal Res. 2005;38(1):1–9. https://doi.org/10.1111/j.1600-079X.2004.00181.x.
Hartl FU. Molecular chaperones in cellular protein folding. Nature. 1996;381(6583):571–9. https://doi.org/10.1038/381571a0.
Kim H-E, Jiang X, Du F, Wang X. PHAPI, CAS, and Hsp70 promote apoptosome formation by preventing Apaf-1 aggregation and enhancing nucleotide exchange on Apaf-1. Mol Cell. 2008;30(2):239–47. https://doi.org/10.1016/j.molcel.2008.03.014.
Güvenç M, Aksakal M. Ameliorating effect of kisspeptin-10 on methotrexate-induced sperm damages and testicular oxidative stress in rats. Andrologia. 2018;50(8):e13057. https://doi.org/10.1111/and.13057.
Wang Y, Chen Y, Guan L, Zhang H, Huang Y, Johnson CH, et al. Carnitine palmitoyltransferase 1C regulates cancer cell senescence through mitochondria-associated metabolic reprograming. Cell Death Differ. 2018;25(4):733–46. https://doi.org/10.1038/s41418-017-0013-3.
Acknowledgements
The authors thank the Suleyman Demirel University Scientific Research Projects Coordination Unit.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this publication was supported by a grant from this work was supported by the Research Fund of Suleyman Demirel University. Suleyman Demirel University Faculty of Medicine (Ethics Committee No. 13–2/2019), and it was supported by the SDU Scientific Research Projects Unit with the project number TDK-2020–8034. The content is solely the responsibility of the authors and does not necessarily represent the official views of Suleyman Demirel University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
In this study, an experimental animal (rat) was used. The necessary permissions were obtained from the Animal Experiments Local Ethics Committee of Suleyman Demirel University’s (SDU) Faculty of Medicine (Ethics Committee No. 13–2/2019).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is based on the first author’s doctoral thesis under the supervision of the second author.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sarman, E., Gulle, K. & Ilhan, I. Histochemical, Immunohistochemical, and Biochemical Investigation of the Effect of Resveratrol on Testicular Damage Caused by Methotrexate (MTX). Reprod. Sci. 30, 3315–3324 (2023). https://doi.org/10.1007/s43032-023-01269-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01269-x