Abstract
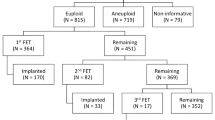
It was suggested that the embryo pooling was an alternative for patients with insufficient number of embryos for preimplantation genetic testing (PGT) in a single ovarian stimulation cycle. However, limited study noticed whether it is an efficient strategy to pool cleavage-stage embryos by vitrification. This study included 71 cycles with vitrified-warmed and fresh embryos simultaneously for PGT between May 2016 and May 2021. The embryos from the same patients were split into two groups based on the origin: warming group and fresh group. Embryo development, sequencing results, clinical and neonatal outcomes were compared. The results showed that the rate of high-quality embryos in the warming group was significantly higher than that in the fresh group (64.53% versus 52.61%, P = 0.011); however, the available blastocyst rate in this group was significantly lower than that in the fresh group (47.29% versus 57.83%, P = 0.026). There were 96 and 144 blastocysts that underwent trophectoderm (TE) biopsy in warming and fresh groups, respectively. The high-quality blastocyst rate was significantly lower in the warming group compared to the fresh group (57.29% versus 70.14%, P = 0.041). The rates of genetic transferable blastocyst were comparable between the two groups (P = 0.956). There were no statistical differences in terms of embryo implantation, clinical pregnancy, miscarriage rates, and neonatal outcomes between the two groups. In conclusion, this study demonstrated that the cleavage-stage embryo pooling strategy might be unfavorable for the maintenance of embryonic development potential. If not necessary, it is not recommended to pool cleavage-stage embryos for PGT.
Similar content being viewed by others
References
Handyside AH, Kontogianni EH, Hardy K, Winston RM. Pregnancies from biopsied. human preimplantation embryos sexed by Y-specific DNA amplification. Nature. 1990;344:768–70. https://doi.org/10.1038/344768a0.
Scott KL, Hong KH, Scott RT. Selecting the optimal time to perform biopsy for preimplantation genetic testing. Fertil Steril. 2013;100:608–14. https://doi.org/10.1016/j.fertnstert.2013.07.004.
Scott RT, Upham KM, Forman EJ, Zhao T, Treff NR. Cleavage-stage biopsy significantly impairs human embryonic implantation potential while blastocyst biopsy does not: a randomized and paired clinical trial. Fertil Steril. 2013;100:624–30. https://doi.org/10.1016/j.fertnstert.2013.04.039.
Xiong S, Liu WW, Wang J, Liu JX, Gao Y, Wu LH, et al. Trophectoderm biopsy protocols may impact the rate of mosaic blastocysts in cycles with pre-implantation genetic testing for aneuploidy. J Assist Reprod Genet. 2021; 04. https://doi.org/10.1007/s10815-021-02137-w.
Chamayou S, Sicali M, Alecci C, Ragolia C, Liprino A, Nibali D, et al. The accumulation of vitrified oocytes is a strategy to increase the number of euploid available blastocysts for transfer after preimplantation genetic testing. J Assist Reprod Genet. 2017;34:479–86. https://doi.org/10.1007/s10815-016-0868-0.
Xiaokun Hu, Chenhui D, DuoDuo Z, Wen Z, Jing W, Yanhong Z, et al. Embryo pooling: a promising strategy for managing insufficient number of embryos in preimplantation genetic diagnosis. Gynecol Endocrinol. 2017;33:867–71. https://doi.org/10.1080/09513590.2017.1347778.
Chatziparasidou A, Nijs M, Moisidou M, Chara O, Ioakeimidou C, Pappas C, et al. Accumulation of oocytes and/or embryos by vitrification: a new strategy for managing poor responder patients undergoing preimplantation diagnosis. F1000Res. 2013;2:240. https://doi.org/10.12688/f1000research.2-240.v2.
Orris JJ, Taylor TH, Gilchrist JW, Hallowell SV, Glassner MJ, Wininger JD. The utility of embryo banking in order to increase the number of embryos available for preimplantation genetic screening in advanced maternal age patients. J Assist Reprod Genet. 2010;27:729–33. https://doi.org/10.1007/s10815-010-9474-8.
Mansoori GA. Kinetics of water loss from cells at subzero centigrade temperatures. Cryobiology. 1975;12:34–45.
Kleinhans FW, Mazur P. Comparison of actual vs. synthesized ternary phase diagrams for solutes of cryobiological interest. Cryobiology. 2007;54:212–22. https://doi.org/10.1016/j.cryobiol.2007.01.007.
Pinborg A, Henningsen AA, Loft A, Malchau SS, Forman J, Andersen AN. Large baby syndrome in singletons born after frozen embryo transfer (FET): is it due to maternal factors or the cryotechnique? Hum Reprod. 2014;29:618–27. https://doi.org/10.1093/humrep/det440.
Stanger J, Wong J, Conceicao J, Yovich J. Vitrification of human embryos previously cryostored by either slow freezing or vitrification results in high pregnancy rates. Reprod Biomed Online. 2012;24:314–20. https://doi.org/10.1016/j.rbmo.2011.11.013.
Taylor TH, Patrick JL, Gitlin SA, Michael Wilson J, Crain JL, Griffin DK, et al. Outcomes of blastocysts biopsied and vitrified once versus those cryopreserved twice for euploid blastocyst transfer. Reprod Biomed Online. 2014;29:59–64. https://doi.org/10.1016/j.rbmo.2014.03.001.
Cimadomo D, Rienzi L, Romanelli V, Alviggi E, Levi-Setti PE, Albani E, et al. Inconclusive chromosomal assessment after blastocyst biopsy: prevalence, causative factors and outcomes after re-biopsy and re-vitrification A multicenter experience. Hum Reprod. 2018;33:1839–46. https://doi.org/10.1093/humrep/dey282.
Wang M, Jiang JH, Xi QS, Li D, Ren XL, Li Z, et al. Repeated cryopreservation process impairs embryo implantation potential but does not affect neonatal outcomes. Reprod Biomed Online. 2021;42:75–82. https://doi.org/10.1016/j.rbmo.2020.11.007.
Zheng XY, Chen Y, Yan J, Wu YQ, Zhuang XJ, Lin SL, et al. Effect of repeated cryopreservation on human embryo developmental potential. Reprod Biomed Online. 2017;35:627–32. https://doi.org/10.1016/j.rbmo.2017.08.016.
Ding X, Yang J, Li L, Yang N, Lan L, Huang G, Ye H. Fertility outcomes in women after controlled ovarian stimulation with gonadotropin releasing hormone agonist long protocol: fresh versus frozen embryo transfer. BMC Pregnancy Childbirth. 2021;21:207. https://doi.org/10.1186/s12884-021-03698-5.
Jingyu Li, Shun X, Yanhua Z, Chong Li, Wei H, Guoning H. Effect of the re-vitrification of embryos at different stages on embryonic developmental potential. Front Endocrinol. 2021;12:653310. https://doi.org/10.3389/fendo.2021.653310.
Xiong S, Han W, Liu JX, Zhang XD, Liu WW, Liu H, et al. Effects of cumulus cells removal after 6 h co-incubation of gametes on the outcomes of human IVF. J Assist Reprod Genet. 2011;28:1205–11. https://doi.org/10.1007/s10815-011-9630-9.
Gardner DK, Lane M. Culture and selection of viable blastocysts: a feasible proposition for human IVF? Hum Reprod Update. 1997;3:367–82. https://doi.org/10.1093/humupd/3.4.367.
Xiong S, Liu JX, Gao Y, Liu WW, Wu LH, Han W, et al. Shortened equilibration time can compromise clinical outcomes in human embryo vitrification. Hum Fertil (Camb). 2016;19:114–9. https://doi.org/10.1080/14647273.2016.1186848.
Capalbo A, Rienzi L, Cimadomo D, Maggiulli R, Elliott T, Wright G, et al. Correlation between standard blastocyst morphology, euploidy and implantation: an observational study in two centers involving 956 screened blastocysts. Hum Reprod. 2014;29:1173–81. https://doi.org/10.1093/humrep/deu033.
Hu XK, Ding CH, Zhang DD, Zhou W, Wang J, Zeng YH, et al. Embryo pooling: a promising strategy for managing insufficient number of embryos in preimplantation genetic diagnosis. Gynecol Endocrinol. 2017;33:867–71. https://doi.org/10.1080/09513590.2017.1347778.
Wilding M, Terribile M, Parisi I, Nargund G. Thaw, biopsy and refreeze strategy for PGT-A on previously cryopreserved embryos. Facts Views Vis Obgyn. 2019;11:223–7.
Anick DV, Lisbet VL, Martine DR, Pieter V, Greta V, Andrea B, et al. Multiple vitrification-warming and biopsy procedures on human embryos: clinical outcome and neonatal follow-up of children. Hum Reprod. 2020;35:2488–96. https://doi.org/10.1093/humrep/deaa236.
Lan Yu, Jia C, Lan Y, Song R, Zhou L, Li Y, et al. Analysis of embryo intactness and developmental potential following slow freezing and vitrification. Syst Biol Reprod Med. 2017;63:285–93. https://doi.org/10.1080/19396368.2017.1362060.
Vanderzwalmen P, Ectors F, Grobet L, Prapas Y, Panagiotidis Y, Vanderzwalmen S, et al. Aseptic vitrification of blastocysts from infertile patients, egg donors and after IVM. Reprod Biomed Online. 2009;19:700–7. https://doi.org/10.1016/j.rbmo.2009.09.011.
Kader A, Choi A, Sharma RK, Falcone T, Agarwal A. Effect of varying equilibration time in a two-step vitrification method on the post-warming DNA integrity of mouse blastocysts. Fertil Steril. 2010;93:2640–5. https://doi.org/10.1016/j.fertnstert.2009.07.991.
Chatzimeletiou K, Morrison EE, Vanderzwalmen P, Prapas N, Prapas Y, Basil C, et al. Cytoskeletal analysis of human blastocysts by confocal laser scanning microscopy following vitrification. Hum Reprod. 2012;27:106–13. https://doi.org/10.1093/humrep/der344.
Zhang SP, Tan K, Gong F, Gu YF, Tan YQ, Lu CF, et al. Blastocysts can be rebiopsied for preimplantation genetic diagnosis and screening. Fertil Steril. 2014;102:1641–5. https://doi.org/10.1016/j.fertnstert.2014.09.018.
Acknowledgements
Our thanks should be given to all the embryologists and biologists in charge of embryo biopsy, cryopreservation, and NGS analysis.
Funding
This work was supported by the Chongqing Health Committee (grant number: 2019MSXM067, 2020MSXM086 and 2021MSXM108).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Shun Xiong and Xiangwei Hao are contributed equally to the research and preparation of the manuscript.
Rights and permissions
About this article
Cite this article
Xiong, S., Hao, X., Gao, Y. et al. Accumulation of Cleavage-Stage Embryos by Vitrification may Compromise Embryonic Developmental Potential in Preimplantation Genetic Testing. Reprod. Sci. 29, 1930–1938 (2022). https://doi.org/10.1007/s43032-022-00880-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-022-00880-8