Abstract
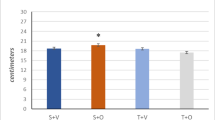
The hypertension incidence and its complication on pregnant women are growing and can lead to adverse consequences on their fetuses. However, it is known that regular exercise practice can be healthful to hypertensive pregnant women but harmful to fetal growth. So, the objective of this study was to evaluate the effects of exercise beginning before pregnancy or during pregnancy on the maternal blood pressure and reproductive outcome and on the fetal development of spontaneously hypertensive rats (SHR). Pregnant SHR were randomly distributed into three experimental groups: (1) SHR-Control, non-exercised; (2) SHR-Ex0, rats submitted to physical exercise (swimming program) from day zero to 20 of pregnancy; (3) and SHR-ExPr, rats submitted to swimming program before and during pregnancy. At end of pregnancy (day 21), the rats were anesthetized, and reproductive parameters and fetal development were assessed. Blood pressure was reduced at the end of pregnancy in all the groups. Regardless of swimming exposure time, there was reduced maternal weight gain. The exercise decreased fetal weight at term pregnancy, with a higher percentage of small for gestational age (SGA) fetuses and lower number ossification sites, indicating intrauterine growth restriction (IUGR). In conclusion, our findings provide insight to support that swimming exercise in pregnant SHR impairs fetal development, causing IUGR and visceral malformations. Therefore, the indication of physical exercise must be defined very carefully, as it can compromise fetal development.


Similar content being viewed by others
Availability of Data and Material (Data Transparency)
Data supporting findings are presented within the manuscript.
Code Availability (Software Application or Custom Code)
Not applicable.
References
Kearney PM, Whelton M, Reynolds K, Muntner P, Whelton PK, He J. Global burden of hypertension : analysis of worldwide data. Lancet. 2005;365:217–23. https://doi.org/10.1016/S0140-6736(05)17741-1.
Mills KT, Bundy JD, Kelly TN, Reed JE, Kearney PM, Reynolds K, et al. Global disparities of hypertension prevalence and control: a systematic analysis of population-based studies from 90 countries. Circulation. 2016;134:441–50. https://doi.org/10.1161/CIRCULATIONAHA.115.018912.
Fraser A, Nelson SM, Macdonald-Wallis C, Cherry L, Butler E, Sattar N, et al. Associations of pregnancy complications with calculated cvd risk and cardiovascular risk factors in middle age: the avon longitudinal study of parents and children. Circulation. 2012;125:1367–80. https://doi.org/10.1161/CIRCULATIONAHA.111.044784.
Vest AR, Cho LS. Hypertension in pregnancy. Curr Atheroscler Rep. 2014;16:395. https://doi.org/10.1007/s11883-013-0395-8.
Ohkuchi A, Hirashima C, Takahashi K, Suzuki H, Matsubara S. Prediction and prevention of hypertensive disorders of pregnancy. Hypertens Res. 2016;40:5–14. https://doi.org/10.1038/hr.2016.107.
Sutton ALM, Harper LM, Tita ATN. Hypertensive disorders in pregnancy. Obstet Gynecol Clin N Am. 2018;45:333–47. https://doi.org/10.1016/j.ogc.2018.01.012.
Clapp JF, Kim H, Burciu B, Lopez B. Beginning regular exercise in early pregnancy : Effect on fetoplacental growth. Am J Obstet Gynecol. 2000;183:1484–8. https://doi.org/10.1067/mob.2000.107096.
ACOG Practice Bulletin No. 202 Summary: gestational hypertension and preeclampsia. Obstet Gynecol. 2019;133:1. https://doi.org/10.1097/AOG.0000000000003019.
Rocha R, Peraçoli JC, Volpato GT, Damasceno DC, Campos KE. Effect of exercise on the maternal outcome in pregnancy of spontaneously hypertensive rats. Acta Cir Bras. 2014;29:55–9. https://doi.org/10.1590/s0102-8650201400150002.
Cornelissen VA, Smart NA. Exercise training for blood pressure: a systematic review and meta-analysis. J Am Heart Assoc. 2013;2:e004473. https://doi.org/10.1161/JAHA.112.004473.
Sosa EG. Trastornos hipertensivos durante el embarazo. Rev Cuba Obstet Ginecol. 2000;26:99–114.
Abate DT, Barbosa Neto O, Rossi e Silva RC, Faleiros AC, Correa RR, da Silva VJ, et al. Exercise-training reduced blood pressure and improve placental vascularization in pregnant spontaneously hypertensive rats pilot study. Fetal Pediatr Pathol. 2012;31:423–31. https://doi.org/10.3109/15513815.2012.659535.
Artal R. Exercise in pregnancy: Guidelines. Clin Obstet Gynecol. 2016;59:639–44. https://doi.org/10.1097/GRF.0000000000000223.
Negrao CE, Moreira ED, Santos MC, Farah VM, Krieger EM. Vagal function impairment after exercise training. J Appl Physiol. 1992;72:1749–53. https://doi.org/10.1152/jappl.1992.72.5.1749.
Katona PG, McLean M, Dighton DH, Guz A. Sympathetic and parasympathetic cardiac control in athletes and nonathletes at rest. J Appl Physiol Respir Environ Exerc Physiol. 1982;52:1652–7. https://doi.org/10.1152/jappl.1982.52.6.1652.
Ahokas RA, Sibai BM. The relationship between experimentally determined litter size and maternal blood pressure in spontaneously hypertensive rats. Am J Obstet Gynecol. 1990;162:841–7. https://doi.org/10.1016/0002-9378(90)91021-4.
Corrêa MS, Gelaleti RB, Bento GF, Damasceno DC, Peraçoli JC. DNA damage in Wistar Kyoto rats exercised during pregnancy. Acta Cir Bras. 2017;32:388–95. https://doi.org/10.1590/s0102-865020170050000008.
Li S, Chen Y, Zhang Y, Zhang H, Wu Y, He H, et al. Exercise during pregnancy enhances vascular function via epigenetic repression of CaV1.2 channel in offspring of hypertensive rats. Life Sci. 2019;231:116576. https://doi.org/10.1016/j.lfs.2019.116576.
Damasceno DC, Silva HP, Vaz GF, Vasques-Silva FA, Calderon IM, Rudge MV, et al. Diabetic rats exercised prior to and during pregnancy: Maternal reproductive outcome, biochemical profile, and frequency of fetal anomalies. Reprod Sci. 2013;20:730–8. https://doi.org/10.1177/1933719112461186.
Volpato GT, Damasceno DC, Sinzato YK, Ribeiro VM, Rudge MV, Calderon IM. Oxidative stress status and placental implications in diabetic rats undergoing swimming exercise after embryonic implantation. Reprod Sci. 2015;22:602–8. https://doi.org/10.1177/1933719114556485.
Castardeli E, Paiva SA, Matsubara BB, Matsubara LS, Minicucci MF, Azevedo PS, et al. Chronic cigarette smoke exposure results in cardiac remodeling and impaired ventricular function in rats. Arq Bras Cardiol. 2005;84:320–4. https://doi.org/10.1590/s0066-782x2005000400009.
Okamoto K, Aoki K. Development of a strain of spontaneously hypertensive rats. Chem Pharm Bull. 1963;27:282–93.
Damasceno DC, Kiss ACI, Sinzato YK, de Campos KE, Rudge MVC, Calderon IMP, et al. Maternal-fetal outcome, lipid profile, and oxidative stress of diabetic rats neonatally exposed to streptozotocin. Exp Clin Endocrinol Diabetes. 2011;119:408–13. https://doi.org/10.1055/s-0030-1269886.
Moraes-Souza RQ, Sinzato YK, Antunes BT, Umeoka EHL, Oliveira JAC, Garcia-Cairasco N, et al. Evaluation of maternal reproductive outcomes and biochemical analysis from Wistar Audiogenic Rats (WAR) and repercussions in their offspring. Reprod Sci. 2020;27:2223–31. https://doi.org/10.1002/bdr2.1818.
Staples RE, Schnell VL. Refinements in rapid clearing technique in the KOHalizarin red S method for fetal bone. Stain Technol. 1964;39:61–3.
Wilson JG. Methods for administering agents and detecting malformations in experimental animal. In: Wilson JC, Warkany J, editors. Teratology: Principles and techniques. Chicago: University of Chicago Press; 1965. p. 47–74.
Aliverti V, Bonanomi L, Giavini E, Leone VG, Mariani L. The extent of fetal ossification as an index of delayed development in teratogenic studies. Teratology. 1979;20:237–42. https://doi.org/10.1002/tera.1420200208.
Paula VG, Cruz LL, Sene LB, Gratão TB, Soares TS, Moraes-Souza RQ, et al. Maternal-fetal repercussions of Phyllanthus niruri L. treatment during rat pregnancy. J Ethnopharmacol. 2020;254:112728. https://doi.org/10.1016/j.jep.2020.112728.
Kim K, Chung E, Kim C, Lee S. Swimming exercise during pregnancy alleviates pregnancy-associated long-term memory impairment. Physiol Behav. 2012;107:82–6. https://doi.org/10.1016/j.physbeh.2012.06.004.
Weissgerber TL, Wolfe LA, Davies GA, Mottola MF. Exercise in the prevention and treatment of maternal-fetal disease : a review of the literature. Appl Physiol Nutr Metab. 2006;31:661–74. https://doi.org/10.1139/h06-060.
Roberts CL, Ford JB, Algert CS, Antonsen S, Chalmers J, Cnattingius S, et al. Population-based trends in pregnancy hypertension and pre-eclampsia : an international comparative study. BMJ Open. 2011;1:e000101. https://doi.org/10.1136/bmjopen-2011-000101.
Mudd LM, Owe KM, Morrola MF, Pivarnik JM. Health benefits of physical activity during pregnancy: an international perspective. Med Sci Sports Exerc. 2012;45:268–77. https://doi.org/10.1249/MSS.0b013e31826cebcb.
Dempsey JC, Butler CL, Sorensen TK, Lee IM, Thompson ML, Miller RS, et al. A case-control study of maternal recreational physical activity and risk of gestational diabetes mellitus. Diabetes Res Clin Pract. 2004;66:203–15. https://doi.org/10.1016/j.diabres.2004.03.010.
Robinson AM, Bucci DJ. Physical exercise during pregnancy improves object recognition memory in adult offspring. Neuroscience. 2014;256:53–60. https://doi.org/10.1016/j.neuroscience.2013.10.012.
Peraçoli JC, Rudge MV, Sartori MS, da Silva Franco RJ. Effects of hypertension on maternal adaptations to pregnancy: experimental study on spontaneously hypertensive rats. Sao Paulo Med J. 2001;119:54–8. https://doi.org/10.1590/s1516-31802001000200003.
Scott JN, Goecke JC. Profile of pregnancy in young spontaneously hypertensive rats. Jpn Heart J. 1984;25:263–6. https://doi.org/10.1536/ihj.25.263.
Carbillon L, Uzan M, Uzan S. Pregnancy, vascular tone, and maternal hemodynamics: a crucial adaptation. Obstet Gynecol Surv. 2000;55:574–81. https://doi.org/10.1097/00006254-200009000-00023.
Chahoud I, Paumgartten FJR. Relationships between fetal body weight of Wistar rats at term and the extent of skeletal ossification. Braz J Med Biol Res. 2005;38:565–75. https://doi.org/10.1590/s0100-879x2005000400010.
Lima FR, Oliveira N. Pregnancy and exercise. Rev Bras Reumatol. 2005;45:188–90. https://doi.org/10.1590/S0482-50042005000300018.
Artal R, O’Toole M. Guidelines of the American College of Obstetricians and Gynecologists for exercise during pregnancy and the postpartum period. Br J Sports Med. 2003;37:6–12. https://doi.org/10.1136/bjsm.37.1.6.
Spinillo A, Capuzzo E, Baltaro F, Piazzi G, Incola S, Iasci A. The effect of work activity in pregnancy on the risk of fetal growth retardation. Acta Obstet Gynecol Scand. 1996;75:531–5. https://doi.org/10.3109/00016349609054666.
Corvino SB, Damasceno DC, Sinzato YK, Netto AO, Macedo NCD, Zambrano E, et al. Comparative analysis of two different models of swimming applied to pregnant rats born small for pregnant age. An Acad Bras Cienc. 2017;89(1):223–30. https://doi.org/10.1590/0001-3765201720160285.
Volpato GT, Damasceno DC, Kempinas WG, Rudge MV, Calderon IM. Effect of exercise on the reproductive outcome and fetal development of diabetic rats. Reprod BioMed Online. 2009;19:852–8. https://doi.org/10.1016/j.rbmo.2009.09.027.
Jones MT, Norton KI, Black DM, Graham RE, Armstrong RB. Effect of regular voluntary exercise on resting cardiovascular responses in SHR and WKY pregnant rats. Am J Phys. 1992;73:713–20. https://doi.org/10.1152/jappl.1992.73.2.713.
Bellizzi S, Ali MM, Abalos E, Betran AP, Kapila J, Pileggi-Castro C, et al. Are hypertensive disorders in pregnancy associated with congenital malformations in offspring? Evidence from the WHO Multicountry cross sectional survey on maternal and newborn health. BMC Pregnancy Childbirth. 2016;16:198. https://doi.org/10.1186/s12884-016-0987-8.
Soares TS, Moraes-Souza RQ, Carneiro TB, Araujo-Silva VC, Schavinski AZ, Gratão TB, et al. Maternal-fetal outcomes of exercise applied in rats with mild hyperglycemia after embryonic implantation. Birth Defects Res. 2021;113:287–98. https://doi.org/10.1002/bdr2.1818.
Umpierre D, Stein R. Hemodynamic and vascular effects of resistance training: implications for cardiovascular disease. Arq Bras Cardiol. 2007;89:256–62. https://doi.org/10.1590/s0066-782x2007001600008.
Tatro DL, Dudley GA, Convertino VA. Carotid-cardiac baroreflex response and LBNP tolerance following resistance training. Med Sci Sports Exerc. 1992;24:789–96.
Acknowledgment
The authors are grateful to Mr. Claudio Benedito Silva and Carlos Roberto G. Lima, Academic Support Assistants (ASA) of the Experimental Research Unit (UNIPEX) of the Botucatu Medical School, UNESP, for the excellent technical assistance; Dr. José Eduardo Corrente, Office of Research Support, for statistical analysis; and the financial support provided by CAPES (Coordination of Superior Level Staff Improvement)—financial code 001.
Author information
Authors and Affiliations
Contributions
JCP, RR, and GTV are the project leaders for the study. DCD, GTV, and LAS wrote the first draft and made revisions. DCD, GTV, and LAS conducted the data analysis. DCD, RR, and GTV directed the work. DCD, GTV, ILI, and LAS contributed to the interpretation of the results and critically commented and provided revisions to the manuscript. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sene, L.d.B., Damasceno, D.C., Rocha, R. et al. Influence of Swimming Program on the Blood Pressure of Pregnant Hypertensive Rats and Their Fetuses. Reprod. Sci. 28, 3440–3447 (2021). https://doi.org/10.1007/s43032-021-00617-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-021-00617-z