Abstract
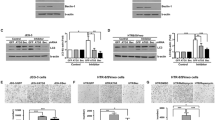
The placenta is important for pregnancy maintenance, and autophagy is documented to be essential for placental development. Autophagy is responsible for degrading and recycling cellular misfolded proteins and damaged organelles. Mitophagy is a selective type of autophagy, where the autophagic machinery engulfs the damaged mitochondria for degradation, and there is reciprocal crosstalk between autophagy and mitochondria. Within these processes, 5’-AMP-activated protein kinase (AMPK) plays an important role. However, the role of AMPK regulation in both autophagy and mitochondria in primary human trophoblasts is unknown. In this study, we address this question by investigating changes in mRNA expression and the abundance of autophagy- and mitochondria-related proteins in isolated human trophoblasts after treatment with AMPK agonists and antagonists. We found that compared to the control group, autophagy was slightly suppressed in the AMPK agonist group and significantly enhanced autophagy in the AMPK antagonist group. However, the expressions of genes related to autophagosome-lysosome fusion were reduced, while genes related to lysosomal function were unchanged in both groups. Furthermore, mitophagy and mitochondrial fusion/fission were both impaired in the AMPK agonist and antagonist groups. Although mitochondrial biogenesis was enhanced in both groups, the function of mitochondrial fatty acid oxidation was increased in the AMPK agonist group but decreased in the AMPK antagonist group. Overall, our study demonstrates that AMPK regulation negatively modulates autophagy and consequently affects mitophagy, mitochondrial fusion/fission, and function in primary human trophoblasts.


Similar content being viewed by others
Data Availability
All the data used to support the findings of this study are included within the article.
References
Woods L, Perez-Garcia V, Hemberger M. Regulation of placental development and its impact on fetal growth-new insights from mouse models. Front Endocrinol. 2018;9:570. https://doi.org/10.3389/fendo.2018.00570.
Kaur J, Debnath J. Autophagy at the crossroads of catabolism and anabolism. Nat Rev Mol Cell Biol. 2015;16(8):461–72. https://doi.org/10.1038/nrm4024.
Gong JS, Kim GJ. The role of autophagy in the placenta as a regulator of cell death. Clin Exp Reprod Med. 2014;41(3):97–107. https://doi.org/10.5653/cerm.2014.41.3.97.
Avagliano L, Massa V, Terraneo L, Samaja M, Doi P, Bulfamante GP, et al. Gestational diabetes affects fetal autophagy. Placenta. 2017;55:90–3. https://doi.org/10.1016/j.placenta.2017.05.002.
Yang Z, Klionsky DJ. Mammalian autophagy: core molecular machinery and signaling regulation. Curr Opin Cell Biol. 2010;22(2):124–31. https://doi.org/10.1016/j.ceb.2009.11.014.
Reggiori F, Klionsky DJ. Autophagy in the eukaryotic cell. Eukaryot Cell. 2002;1(1):11–21. https://doi.org/10.1128/ec.01.1.11-21.2002.
Mizushima N, Levine B. Autophagy in mammalian development and differentiation. Nat Cell Biol. 2010;12(9):823–30. https://doi.org/10.1038/ncb0910-823.
Oh SY, Roh CR. Autophagy Placenta. 2017;60(3):241–59. https://doi.org/10.5468/ogs.2017.60.3.241.
Nixon RA. The role of autophagy in neurodegenerative disease. Nat Med. 2013;19(8):983–97. https://doi.org/10.1038/nm.3232.
Li Q, Liu Y, Sun M. Autophagy and Alzheimer’s disease. Cell Mol Neurobiol. 2017;37(3):377–88. https://doi.org/10.1007/s10571-016-0386-8.
Cerri S, Blandini F. Role of autophagy in Parkinson’s disease. Curr Med Chem. 2019;26(20):3702–18. https://doi.org/10.2174/0929867325666180226094351.
Kroemer G, Mariño G, Levine B. Autophagy and the integrated stress response. Mol Cell. 2010;40(2):280–93. https://doi.org/10.1016/j.molcel.2010.09.023.
Kim J, Guan KL. mTOR as a central hub of nutrient signalling and cell growth. 2019;21(1):63-71. https://doi.org/10.1038/s41556-018-0205-1.
Wang H, Liu Y, Wang D, Xu Y, Dong R, Yang Y, et al. The upstream pathway of mTOR-mediated autophagy in liver diseases. Cells. 2019;8(12). https://doi.org/10.3390/cells8121597.
Mizushima N, Yoshimori T, Levine B. Methods in mammalian autophagy research. Cell. 2010;140(3):313–26. https://doi.org/10.1016/j.cell.2010.01.028.
Han D, Yang B, Olson LK, Greenstein A, Baek SH, Claycombe KJ, et al. Activation of autophagy through modulation of 5'-AMP-activated protein kinase protects pancreatic beta-cells from high glucose. Biochem J. 2010;425(3):541–51. https://doi.org/10.1042/bj20090429.
Schiavi A, Ventura N. The interplay between mitochondria and autophagy and its role in the aging process. Exp Gerontol. 2014;56:147–53. https://doi.org/10.1016/j.exger.2014.02.015.
Holland O, Dekker Nitert M, Gallo LA, Vejzovic M, Fisher JJ, Perkins AV. Review: placental mitochondrial function and structure in gestational disorders. Placenta. 2017;54:2–9. https://doi.org/10.1016/j.placenta.2016.12.012.
Osellame LD, Duchen MR. Quality control gone wrong: mitochondria, lysosomal storage disorders and neurodegeneration. Br J Pharmacol. 2014;171(8):1958–72. https://doi.org/10.1111/bph.12453.
Youle RJ, van der Bliek AM. Mitochondrial fission, fusion, and stress. Science (New York, NY). 2012;337(6098):1062–5. https://doi.org/10.1126/science.1219855.
Muralimanoharan S, Gao X, Weintraub S, Myatt L, Maloyan A. Sexual dimorphism in activation of placental autophagy in obese women with evidence for fetal programming from a placenta-specific mouse model. Autophagy. 2016;12(5):752–69. https://doi.org/10.1080/15548627.2016.1156822.
Mele J, Muralimanoharan S, Maloyan A, Myatt L. Impaired mitochondrial function in human placenta with increased maternal adiposity. Am J Physiol Endocrinol Metab. 2014;307(5):E419–25. https://doi.org/10.1152/ajpendo.00025.2014.
Myatt L, Maloyan A. Obesity and placental function. Semin Reprod Med. 2016;34(1):42–9. https://doi.org/10.1055/s-0035-1570027.
Burton GJ, Yung HW, Murray AJ. Mitochondrial-endoplasmic reticulum interactions in the trophoblast: stress and senescence. Placenta. 2017;52:146–55. https://doi.org/10.1016/j.placenta.2016.04.001.
Chifenti B, Locci MT, Lazzeri G, Guagnozzi M, Dinucci D, Chiellini F, et al. Autophagy-related protein LC3 and Beclin-1 in the first trimester of pregnancy. Clin Exp Reprod Med. 2013;40(1):33–7. https://doi.org/10.5653/cerm.2013.40.1.33.
Shin HJ, Kim H, Oh S, Lee JG, Kee M, Ko HJ, et al. AMPK-SKP2-CARM1 signalling cascade in transcriptional regulation of autophagy. Nature. 2016;534(7608):553–7. https://doi.org/10.1038/nature18014.
Chen M, Liu J, Yang L, Ling W. AMP-activated protein kinase regulates lipid metabolism and the fibrotic phenotype of hepatic stellate cells through inhibition of autophagy. FEBS Open Biol. 2017;7(6):811–20. https://doi.org/10.1002/2211-5463.12221.
Kliman HJ, Nestler JE, Sermasi E, Sanger JM, Strauss JF 3rd. Purification, characterization, and in vitro differentiation of cytotrophoblasts from human term placentae. Endocrinology. 1986;118(4):1567–82. https://doi.org/10.1210/endo-118-4-1567.
Xiao B, Deng X, Zhou W, Tan EK. Flow cytometry-based assessment of mitophagy using MitoTracker. Front Cell Neurosci. 2016;10:76. https://doi.org/10.3389/fncel.2016.00076.
Yang F, Li T, Dong Z, Mi R. MicroRNA-410 is involved in mitophagy after cardiac ischemia/reperfusion injury by targeting high-mobility group box 1 protein. J Cell Biochem. 2018.
Zhang D, Wang W, Sun X, Xu D, Wang C, Zhang Q, et al. AMPK regulates autophagy by phosphorylating BECN1 at threonine 388. Autophagy. 2016;12:1447–59.
Liu X, Chhipa RR, Nakano I, Dasgupta B. The AMPK inhibitor compound C is a potent AMPK-independent antiglioma agent. Mol Cancer Ther. 2014;13(3):596–605. https://doi.org/10.1158/1535-7163.mct-13-0579.
Samari HR, Seglen PO. Inhibition of hepatocytic autophagy by adenosine, aminoimidazole-4-carboxamide riboside, and N6-mercaptopurine riboside. Evidence for involvement of amp-activated protein kinase. J Biol Chem. 1998;273(37):23758–63. https://doi.org/10.1074/jbc.273.37.23758.
Declèves A-E, Sharma K, Satriano J. Beneficial effects of AMPK agonists in kidney ischemia reperfusion: autophagy and cellular stress markers. Nephron Exp Nephrol. 2014;10.
Huang L, Dai K, Chen M, Zhou W, Wang X, Chen J et al. The AMPK agonist PT1 and mTOR inhibitor 3HOI-BA-01 protect cardiomyocytes after ischemia through induction of autophagy. J Cardiovasc Pharmacol Ther. 2015;41–4.
Ji L, Chen Z, Xu Y, Xiong G, Liu R, Wu C, et al. Systematic characterization of autophagy in gestational diabetes mellitus. Endocrinology. 2017;158(8):2522–32. https://doi.org/10.1210/en.2016-1922.
Avagliano L, Terraneo L, Virgili E, Martinelli C, Doi P, Samaja M, et al. Autophagy in normal and abnormal early human pregnancies. Reprod Sci (Thousand Oaks, Calif). 2015;22(7):838–44. https://doi.org/10.1177/1933719114565036.
Code Availability
No custom algorithm, software, or code was used in the present work.
Funding
This study was supported by the National Natural Science Foundation of China (No. 81571455).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest.
Ethics Approval
This study was approved by the institutional review boards of Baylor College of Medicine and Beijing Chaoyang Hospital.
Consent to Participate
Informed consent was obtained from all participants.
Consent for Publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Fig. 1
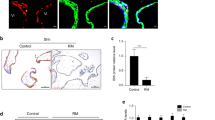
Graph of primary trophoblasts isolated from the human placenta. Purified trophoblasts isolated from human placenta were stained using cell markers CK18 and hPL (PNG 880 kb)
Rights and permissions
About this article
Cite this article
Yang, Hq., Chandra, Y. & Zhang, Zy. Modulation of Autophagy Through Regulation of 5’-AMP-Activated Protein Kinase Affects Mitophagy and Mitochondrial Function in Primary Human Trophoblasts. Reprod. Sci. 28, 2314–2322 (2021). https://doi.org/10.1007/s43032-021-00495-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-021-00495-5