Abstract
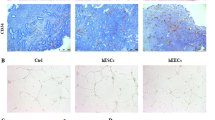
Endometriosis is responsible for pain symptoms with great impact on the patient’s quality of life. Several medication lines have been studied aiming at its definitive treatment. Among them, angiogenesis inhibitor factors may be effective given that angiogenesis has fundamental role in the establishment and growth of endometriotic lesions. In this study, we investigated the influence of bevacizumab, anti-factor drug of endothelial growth (anti-VEGF), used at two different dosages, in experimental endometriosis induced in rats. After the induction of endometriosis lesions in rats, they were divided in 3 groups: control group, no treatment, and two other groups were treated with different dosages of the same medication for 4 weeks. At the end of the treatment, endometriotic lesions were removed and evaluated regarding area of lesions, presence of endometrial tissue in microscopy, positivity for anti-VEGF antibody in immunohistochemistry, and gene expression of Pcna, Mmp9, Tp63, and Vegfa. Bevacizumab acted by reducing the area of lesions in the groups that received medication (p = 0.002) and reducing gene expression to Tp63 in lesions (p = 0.04). There was no significant result in other evaluations. We observed that there was significant reduction of the area of lesions among groups, suggesting that bevacizumab has a positive effect on disease control. The gene expression of Tp63 was significantly lower in the group that received high dose of the drug when compared with the other two groups; therefore, we concluded that bevacizumab acts by reducing cell proliferation and differentiation in lesions, constituting a real option for treating endometriosis.




Similar content being viewed by others
References
Worley MJ, Welch WR, Berkowitz RS, Ng S-W. Endometriosis-associated ovarian cancer: a review of pathogenesis. Int J Mol Sci. 2013;14(3):5367–79.
Farquhar C. Endometriosis. BMJ. 20073;334(7587):249–53.
Catenacci M, Sastry S, Falcone T. Laparoscopic surgery for endometriosis. Clin Obstet Gynecol. 2009;52(3):351–61.
Dunselman GAJ, Vermeulen N, Becker C, Calhaz-Jorge C, D’Hooghe T, De Bie B, et al. ESHRE guideline: management of women with endometriosis. Hum Reprod. 2014;29(3):400–12.
Streuli I, de Ziegler D, Santulli P, Marcellin L, Borghese B, Batteux F, et al. An update on the pharmacological management of endometriosis. Expert Opin Pharmacother. 2013;14(3):291–305.
Practice Committee of the American Society for Reproductive Medicine. Treatment of pelvic pain associated with endometriosis: a committee opinion. Fertil Steril. 2014;101(4):927–35.
Kho RM, Andres MP, Borrelli GM, Neto JS, Zanluchi A, Abrão MS. Surgical treatment of different types of endometriosis: comparison of major society guidelines and preferred clinical algorithms. Best Pract Res Clin Obstet Gynaecol. 2018;51:102–10.
Liu H, Wang J, Wang H, Tang N, Li Y, Zhang Y, et al. Correlation between matrix metalloproteinase-9 and endometriosis. Int J Clin Exp Pathol. 2015;8(10):13399–404.
Groothuis PG, Nap AW, Winterhager E, Grümmer R. Vascular development in endometriosis. Angiogenesis. 2005;8(2):147–56.
Bourlev V, Volkov N, Pavlovitch S, Lets N, Larsson A, Olovsson M. The relationship between microvessel density, proliferative activity and expression of vascular endothelial growth factor-A and its receptors in eutopic endometrium and endometriotic lesions. Reproduction. 2006;132(3):501–9.
Laschke MW, Giebels C, Nickels RM, Scheuer C, Menger MD. Endothelial progenitor cells contribute to the vascularization of endometriotic lesions. Am J Pathol. 2011;178(1):442–50.
Laschke MW, Giebels C, Menger MD. Vasculogenesis: a new piece of the endometriosis puzzle. Hum Reprod Update. 2011;17(5):628–36.
Nisolle M, Casanas-Roux F, Anaf V, Mine JM, Donnez J. Morphometric study of the stromal vascularization in peritoneal endometriosis. Fertil Steril. 1993;59(3):681–4.
Song W-W, Lu H, Hou W-J, Xu G-X, Zhang J-H, Sheng Y-H, et al. Expression of vascular endothelial growth factor C and anti-angiogenesis therapy in endometriosis. Int J Clin Exp Pathol. 2014;7(11):7752–9.
Machado DE, Abrao MS, Berardo PT, Takiya CM, Nasciutti LE. Vascular density and distribution of vascular endothelial growth factor (VEGF) and its receptor VEGFR-2 (Flk-1) are significantly higher in patients with deeply infiltrating endometriosis affecting the rectum. Fertil Steril. 2008;90(1):148–55.
Becker CM, D’Amato RJ. Angiogenesis and antiangiogenic therapy in endometriosis. Microvasc Res. 2007;74(2–3):121–30.
Hull ML, Charnock-Jones DS, Chan CLK, Bruner-Tran KL, Osteen KG, Tom BDM, et al. Antiangiogenic agents are effective inhibitors of endometriosis. J Clin Endocrinol Metab. 2003;88(6):2889–99.
Laschke MW, Menger MD. Anti-angiogenic treatment strategies for the therapy of endometriosis. Hum Reprod Update. 2012;18(6):682–702.
Nap AW, Griffioen AW, Dunselman GAJ, Bouma-Ter Steege JCA, Thijssen VLJL, Evers JLH, et al. Antiangiogenesis therapy for endometriosis. J Clin Endocrinol Metab. 2004;89(3):1089–95.
Taylor RN, Lebovic DI, Mueller MD. Angiogenic factors in endometriosis. Ann N Y Acad Sci. 2002;955:89–100 discussion 118, 396–406.
Shih T, Lindley C. Bevacizumab: an angiogenesis inhibitor for the treatment of solid malignancies. Clin Ther. 2006;28(11):1779–802.
Ollendorf DA, Colby JA, Pearson SD. Comparative effectiveness of anti-VEGF agents for diabetic macular edema. Int J Technol Assess Health Care. 2013;29(4):392–401.
Vernon MW, Wilson EA. Studies on the surgical induction of endometriosis in the rat. Fertil Steril. 1985;44(5):684–94.
Abbott FV, Hellemans KG. Phenacetin, acetaminophen and dipyrone: analgesic and rewarding effects. Behav Brain Res. 2000;112(1–2):177–86.
Ozer H, Boztosun A, Açmaz G, Atilgan R, Akkar OB, Kosar MI. The efficacy of bevacizumab, sorafenib, and retinoic acid on rat endometriosis model. Reprod Sci. 2013;20(1):26–32.
Novella-Maestre E, Carda C, Noguera I, Ruiz-Saurí A, García-Velasco JA, Simón C, et al. Dopamine agonist administration causes a reduction in endometrial implants through modulation of angiogenesis in experimentally induced endometriosis. Hum Reprod. 2009;24(5):1025–35.
Rosa-e-Silva JC, Garcia SB, de Sá Rosa-e-Silva ACJ, Candido-dos-Reis FJ, Poli-Neto OB, Ferriani RA, et al. Increased cell proliferation in experimentally induced endometriosis in rabbits. Fertil Steril. 2010;93(5):1637–42.
Remmele W, Stegner HE. Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe. 1987;8(3):138–40.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25(4):402–8.
Liu S, Xin X, Hua T, Shi R, Chi S, Jin Z, et al. Efficacy of anti-VEGF/VEGFR agents on animal models of endometriosis: a systematic review and meta-analysis. PLoS One. 2016;11(11):e0166658.
Soysal D, Kızıldağ S, Saatlı B, Posacı C, Soysal S, Koyuncuoğlu M, et al. A novel angiogenesis inhibitor bevacizumab induces apoptosis in the rat endometriosis model. Balkan J Med Genet. 2014;17(2):73–80.
Ricci AG, Olivares CN, Bilotas MA, Meresman GF, Barañao RI. Effect of vascular endothelial growth factor inhibition on endometrial implant development in a murine model of endometriosis. Reprod Sci. 2011;18(7):614–22.
Bulletti C, Coccia ME, Battistoni S, Borini A. Endometriosis and infertility. J Assist Reprod Genet. 2010;27(8):441–7.
Graesslin O, Cortez A, Uzan C, Birembaut P, Quereux C, Daraï E. Endometrial tumor invasiveness is related to metalloproteinase 2 and tissue inhibitor of metalloproteinase 2 expressions. Int J Gynecol Cancer. 2006;16(5):1911–7.
Weigel MT, Krämer J, Schem C, Wenners A, Alkatout I, Jonat W, et al. Differential expression of MMP-2, MMP-9 and PCNA in endometriosis and endometrial carcinoma. Eur J Obstet Gynecol Reprod Biol. 2012;160(1):74–8.
Wren JD, Wu Y, Guo S-W. A system-wide analysis of differentially expressed genes in ectopic and eutopic endometrium. Hum Reprod. 2007;22(8):2093–102.
Di Como CJ, Urist MJ, Babayan I, Drobnjak M, Hedvat CV, Teruya-Feldstein J, et al. p63 expression profiles in human normal and tumor tissues. Clin Cancer Res. 2002;8(2):494–501.
Poli Neto OB, Ferreira HM, Ramalho LNZ, Rosa e Silva JC, Candido dos Reis FJ, Nogueira AA. Expression of p63 differs in peritoneal endometriosis, endometriomas, adenomyosis, rectovaginal septum endometriosis, and abdominal wall endometriosis. Arch Pathol Lab Med. 2007;131(7):1099–102.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zani, A.C.T., Valerio, F.P., Meola, J. et al. Impact of Bevacizumab on Experimentally Induced Endometriotic Lesions: Angiogenesis, Invasion, Apoptosis, and Cell Proliferation. Reprod. Sci. 27, 1943–1950 (2020). https://doi.org/10.1007/s43032-020-00213-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-020-00213-7