Abstract
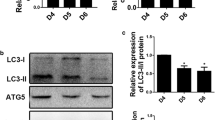
Objective: The purpose of this study was to analyze the autophagy of the human uterine myometrium during the labor. Methods: We collected uterine myometrium strips from term, singleton, nulliparous healthy women undergoing cesarean delivery before labor (nonlabor group, n = 10) or during normal labor (in-labor group, n = 10) without rupturing of membrane. The indications for cesarean delivery were breech presentation or maternal request. Transmission electron microscopy was used to observe autophagosomes. Reverse transcriptase polymerase chain reaction, immunofluorescence, and Western blot were used to quantify the messenger RNA (mRNA) and protein level of the autophagy markers LC3B, P62, and Beclin-1 in the uterine muscle strips. Results: There were no differences between both groups in maternal age, body mass index, gestational week, neonatal weight, operative bleeding, and postpartum bleeding. Transmission electron micrographs showed that autophagosomes existed in myometrial tissue in both groups. There were more autophagosomes in the in-labor group than in the nonlabor group, and the difference had significance. The in-labor group had significantly greater LC3B mRNA expression but significantly lower P62 mRNA expression compared with the nonlabor group. Semiquantitative immunofluorescence in uterine myometrial cells in the in-labor group showed increased LC3B puncta formation and greater Beclin-1 expression but reduced P62 puncta formation compared with the nonlabor group. The ratio of LC3BII/I proteins was significantly higher, but P62 protein was significantly lower in the in-labor group compared with the nonlabor group. The Beclin-1 mRNA and protein expressions were not significantly different between the 2 groups. Conclusion: Autophagy was activated in human uterine myometrium during labor and might play an important role in maintaining uterine contraction function.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arrowsmith S, Kendrick A, Hanley JA, Noble K, Wray S. Myometrial physiology—time to translate? Exp Physiol. 2014;99(3):495–502.
Smith R, Imtiaz M, Banney D, Paul JW, Young RC. Why the heart is like an orchestra and the uterus is like a soccer crowd. Am J Obstet Gynecol. 2015;213(2):181–5.
Chaemsaithong P, Madan I, Romero R, et al. Characterization of the myometrial transcriptome in women with an arrest of dilatation during labor. J Perinat Med. 2013;41(6):665–81.
Cohen WR, Friedman EA. The assessment of labor: a brief history. J Perinat Med. 2018;46(1):1–8.
Mylonas I, Friese K. Indications for and risks of elective cesarean section. Dtsch Arztebl Int. 2015;112(29-30):489–95.
Brar HS, Platt LD, DeVore GR, Horenstein J, Medearis AL. Qualitative assessment of maternal uterine and fetal umbilical artery blood flow and resistance in laboring patients by Doppler velocimetry. Am J Obstet Gynecol. 1988;158(4):952–6.
Fleischer A, Anyaegbunam AA, Schulman H, Farmakides G, Randolph G. Uterine and umbilical artery velocimetry during normal labor. Am J Obstet Gynecol. 1987;157(1):40–3.
Baron J, Shwarzman P, Sheiner E, et al. Blood flow doppler velocimetry measured during active labor. Arch Gynecol Obstet. 2015;291(4):837–40.
Larcombe-McDouall J, Buttell N, Harrison N, Wray S. In vivo pH and metabolite changes during a single contraction in rat uterine smooth muscle. J Physiol. 1999;518(pt 3):783–90.
Delbridge LMD, Mellor KM, Taylor DJ, Gottlieb RA. Myocardial stress and autophagy: mechanisms and potential therapies. Nat Rev Cardiol. 2017;14(7):412–25.
Wolf MS, Bayır H, Kochanek PM, Clark RSB. The role of autophagy in acute brain injury: a state of flux? Neurobiol Dis. 2019;122(2):9–15.
Alotaibi M, Arrowsmith S, Wray S. Hypoxia-induced force increase (HIFI) is a novel mechanism underlying the strengthening of labor contractions, produced by hypoxic stresses. Proc Natl Acad Sci U S A. 2015;112(31):9763–8.
Klionsky DJ, Abdelmohsen K, Abe A, et al. Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy. 2016;12(1):1–222.
Antonioli M, Di Rienzo M, Piacentini M, Fimia GM. Emerging mechanisms in initiating and terminating autophagy. Trends Biochem Sci. 2017;42(1):28–41.
Yan WJ, Dong HL, Xiong LZ. The protective roles of autophagy in ischemic preconditioning. Acta Pharmacol Sin. 2013;34(5):636–43.
Sheng R, Qin ZH. The divergent roles of autophagy in ischemia and preconditioning. Acta Pharmacol Sin. 2015;36(4):411–20.
Oh SY, Choi SJ, Kim KH, Cho EY, Kim JH, Roh CR. Autophagy-related proteins, LC3 and Beclin-1, in placentas from pregnancies complicated by preeclampsia. Reprod Sci. 2008;15(9):912–20.
Li X, Han X, Bao J, et al. Nicotine increases eclampsia-like seizure threshold and attenuates microglial activity in rat hippocampus through the α7 nicotinic acetylcholine receptor. Brain Res. 1642;2016:487–96.
Luo S, Rubinsztein DC. Apoptosis blocks Beclin 1-dependent autophagosome synthesis: an effect rescued by Bcl-xL. Cell Death Differ. 2010;17(2):268–77.
Kanninen TT. de1Andrade Ramos BR, Witkin SS. The role of autophagy in reproduction from gametogenesis to parturition. Eur J Obstet Gynecol Reprod Biol. 2013;171(1):3–8.
Saito S, Nakashima A. Review: the role of autophagy in extravillous trophoblast function under hypoxia. Placenta. 2013;34(suppl):S79–84.
Oh SY, Choi SJ, Kim KH, et al. Autophagy-related proteins, LC3 and Beclin-1, in placentas from pregnancies complicated by preeclampsia. Reprod Sci. 2008;15(9):912–20.
Nakashima A, Yamanaka-Tatematsu M, Fujita N, et al. Impaired autophagy by soluble endoglin, under physiological hypoxia in early pregnant period, is involved in poor placentation in preeclampsia. Autophagy. 2013;9(3):303–16.
Hirota Y, Daikoku T, Tranguch S, Xie H, Bradshaw HB, Dey SK. Uterine-specific p53 deficiency confers premature uterine senescence and promotes preterm birth in mice. J Clin Invest. 2010;120(3):803–15.
Alotaibi M. Brief hypoxic cycles improve uterine contractile function after prolonged hypoxia in term-pregnant but not in non-pregnant rats in vitro. Theriogenology. 2018;113:73–7.
Zhou P, Tan YZ, Wang HJ, Wang GD. Hypoxic preconditioning-induced autophagy enhances survival of engrafted endothelial progenitor cells in ischaemic limb. J Cell Mol Med. 2017;21(10):2452–64.
Gurusamy N, Lekli I, Gorbunov NV, Gherghiceanu M, Popescu LM, Das DK. Cardioprotection by adaptation to ischaemia augments autophagy in association with BAG-1 protein. J Cell Mol Med. 2009;13(2):373–87.
Yan L, Sadoshima J, Vatner DE, Vatner SF. Autophagy in ischemic preconditioning and hibernating myocardium. Autophagy. 2014;5(5):709–12.
Domart MC, Esposti DD, Sebagh M, et al. Concurrent induction of necrosis, apoptosis, and autophagy in ischemic preconditioned human livers formerly treated by chemotherapy. J Hepatol. 2009;51(5):881–9.
Yan L, Vatner DE, Kim SJ, et al. Autophagy in chronically ischemic myocardium. Proc Natl Acad Sci U S A. 2005;102(39):13807–12.
Carloni S, Buonocore G, Balduini W. Protective role of autophagy in neonatal hypoxia-ischemia induced brain injury. Neurobiol Dis. 2008;32(3):329–39.
Carloni S, Buonocore G, Longini M, Proietti F, Balduini W. Inhibition of rapamycin-induced autophagy causes necrotic cell death associated with Bax/Bad mitochondrial translocation. Neuroscience. 2012;203:160–9.
Neel BA, Lin Y, Pessin JE. Skeletal muscle autophagy: a new metabolic regulator. Trends Endocrinol Metab. 2013;24(12):635–43.
Hanley JA, Weeks A, Wray S. Physiological increases in lactate inhibit intracellular calcium transients, acidify myocytes and decrease force in term pregnant rat myometrium. J Physiol. 2015;593(20):4603–14.
Quenby S, Pierce SJ, Brigham S, Wray S. Dysfunctional labor and myometrial lactic acidosis. Obstet Gynecol. 2004;103(4):718–23.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Wang, L., Hu, H., Morse, A.N. et al. Activation of Autophagy in Human Uterine Myometrium During Labor. Reprod. Sci. 27, 1665–1672 (2020). https://doi.org/10.1007/s43032-020-00198-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-020-00198-3