Abstract
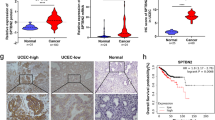
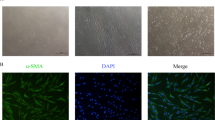
Abnormal expression of SPARC (osteonectin), cwcv and kazal-like domains proteoglycan 2 (SPOCK2) plays a significant role in the development and progression of various human cancers, yet a relationship between SPOCK2 and endometrial cancer (EC) has not been reported. Here, we assessed the potential role and mechanism by which SPOCK2 acts in the pathogenesis and progression of EC. First, protein expression of SPOCK2 in EC tissue from patients was detected by immunohistochemistry and associated clinical data were analyzed. Then, HEC-1A and Ishikawa cells were transfected with an adenoviral vector containing an SPOCK2 recombinant fragment and the biological behavior of transfected cells was observed. Finally, the expression of membrane type 1 matrix metalloproteinase (MT1-MMP) and MMP2 in the transfected cells was detected by Western blot and zymography gel assay to analyze the effect of SPOCK2 on the regulation of the MT1-MMP/MMP2 pathway. We found that there was significantly less SPOCK2 protein expression in the EC tissue than in the normal endometrium tissue, and lack of SPOCK2 protein expression in EC tissue was associated with distant metastasis and myometrial invasion. Upregulation of SPOCK2 in HEC-1A and Ishikawa cells inhibited cell proliferation, invasion, adhesion, and apoptosis. Upregulation of SPOCK2 inhibited the expression of MT1-MMP and MMP2 and activation of MMP2 in HEC-1A and Ishikawa cells. Collectively, our data indicated that SPOCK2 contributed to the progression of EC by regulating the biological behavior of cancer cells, which is achieved partly through regulating protein expression of MT1-MMP and MMP2 and activation of MMP2.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Oki S, Sone K, Oda K, et al. Oncogenic histone methyltransferase EZH2: a novel prognostic marker with therapeutic potential in endometrial cancer. Oncotarget. 2017;8(25):40402–11.
Bokhman JV. Two pathogenetic types of endometrial carcinoma. Gynecol Oncol. 1983;15(1):10–7.
Noer MC, Antonsen SL, Ottesen B, Christensen IJ, Høgdall C. Type I versus type II endometrial cancer: differential impact of comorbidity. Int J Gynecol Cancer. 2018;28(3):586–93.
Nordgard SH, Johansen FE, Alnaes GI, et al. Genome-wide analysis identifies 16q deletion associated with survival, molecular subtypes, mRNA expression, and germline haplotypes in breast cancer patients. Genes Chromosomes Cancer. 2008;47(8):680–96.
Chung W, Kwabi-Addo B, Ittmann M, et al. Identification of novel tumor markers in prostate, colon and breast cancer by unbiased methylation profiling. PLoS One. 2008;3(4):e2079.
Ren F, Wang DB, Li T, Chen YH, Li Y. Identification of differentially methylated genes in the malignant transformation of ovarian endometriosis. J Ovarian Res. 2014;7:73.
Wang CT, Wang DB, Liu KR, et al. Inducing malignant transformation of endometriosis in rats by long-term sustaining hyperestrogenemia and type II diabetes. Cancer Sci. 2015;106(1):43–50.
Rodolakis A, Thomakos N, Akrivos N, et al. Clinicopathologic insight of simultaneously detected primary endometrial and ovarian carcinomas. Arch Gynecol Obstet. 2012;285(3):817–21.
Alliel PM, Perin JP, Jolles P, Bonnet F. Testican, a multidomain testicular proteoglycan resembling modulators of cell social behaviour. Eur J Biochem. 1993;214(1):347–50.
Sambuudash O, Kim HS, Cho MY. Lack of aberrant methylation in an adjacent area of left-sided colorectal cancer. Yonsei Med J. 2017;58(4):749–55.
Ren F, Wang DB, Li T. Epigenetic inactivation of SPOCK2 in the malignant transformation of ovarian endometriosis. Zhonghua Fu Chan Ke Za Zhi. 2011;46(11):822–5.
Song H, Pan D, Sun W, et al. SiRNA directed against annexin II receptor inhibits angiogenesis via suppressing MMP2 and MMP9 expression. Cell Physiol Biochem. 2015;35(3):875–84.
Wang HY, Tu YS, Long J, et al. SRF-miR 29b-MMP2 axis inhibits NSCLC invasion and metastasis. Int J Oncol. 2015;47(2):641–9.
Yi SA, Ryu HW, Lee DH, Han JW, Kwon SH. HP1β suppresses metastasis of human cancer cells by decreasing the expression and activation of MMP2. Int J Oncol. 2014;45(6):2541–8.
Sato H, Takino T. Coordinate action of membrane-type matrix metalloproteinase-1 (MT1-MMP) and MMP-2 enhances pericellular proteolysis and invasion. Cancer Sci. 2010;101(4):843–7.
Al-Raawi D, Abu-El-Zahab H, El-Shinawi M, Mohamed MM. Membrane type-1 matrix metalloproteinase (MT1-MMP) correlates with the expression and activation of matrix metalloproteinase-2 (MMP-2) in inflammatory breast cancer. Int J Clin Exp Med. 2011;4(4):265–75.
Koshikawa N, Giannelli G, Cirulli V, Miyazaki K, Quaranta V. Role of cell surface metalloprotease MT1-MMP in epithelial cell migration over laminin-5. J Cell Biol. 2000;148(3):615–24.
Lehti K, Lohi J, Juntunen MM, Pei D, Keski-Oja J. Oligomerization through hemopexin and cytoplasmic domains regulates the activity and turnover of membrane-type 1 matrix metalloproteinase. J Biol Chem. 2002;277(10):8440–8.
Nakada M, Yamada A, Takino T, et al. Suppression of membrane-type 1 matrix metalloproteinase (MMP)-mediated MMP-2 activation and tumor invasion by testican 3 and its splicing variant gene product. N-Tes. Cancer Res. 2001;61(24):8896–902.
Nakada M, Miyamori H, Yamashita J, Sato H. Testican 2 abrogates inhibition of membrane-type matrix metalloproteinases by other testican family proteins. Cancer Res. 2003;63(12):3364–9.
Berger EA, McClellan SA, Barrett RP, Hazlett LD. Testican-1 promotes resistance against Pseudomonas aeruginosa-induced keratitis through regulation of MMP-2 expression and activation. Invest Ophthalmol Vis Sci. 2011;52(8):5339-5346.
Kudo T, Takino T, Miyamori H, Thompson EW, Sato H. Substrate choice of membrane-type matrix metalloproteinase is dictated by tissue inhibitor of metalloproteinase-2 levels. Cancer Sci. 2007;98(4):563–8.
Yang J, Yang Q, Yu J, Li X, Yu S, Zhang X. SPOCK1 promotes the proliferation, migration and invasion of glioma through PI3K/AKT and Wnt/β-catenin signaling pathways. Oncol Rep. 2016;35(6):3566–76.
Chen D, Zhou H, Liu G, Zhao Y, Cao G, Liu Q. SPOCK1 promotes the invasion and metastasis of gastric cancer through Slug-induced epithelial–mesenchymal transition. J Cell Mol Med. 2018;22(2):797–807.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Ren, F., Wang, D., Wang, Y. et al. SPOCK2 Affects the Biological Behavior of Endometrial Cancer Cells by Regulation of MT1-MMP and MMP2. Reprod. Sci. 27, 1391–1399 (2020). https://doi.org/10.1007/s43032-020-00197-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-020-00197-4