Abstract
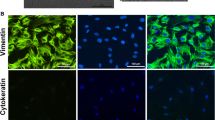
Thoracic endometriosis (TE) syndrome is a clinical condition known as an extrapelvic form of endometriosis with the presence of functioning endometrial tissue involving lung parenchyma, pleura, chest wall, or diaphragm. In an effort to obtain an endometriosis ex vivo model, we established the spontaneously growing TH-EM1 cell line from endometriotic implants in lung parenchyma from a woman with TE. Maintained in long-term culture, the cells grew as large mesenchymal-like cells with a doubling time between 5 and 6 days. Treatment with medroxyprogesterone acetate (10–7 mol/L) inhibited the TH-EM1 cells growth and induced morphological changes to an epithelial-like cells. Strong expression of the nuclear estrogen receptors, progesterone receptors, and erytropoietin receptors were found in both the pulmonary implant and the TH-EM1 cells by immunohistochemical analysis. Consistent immunoreactivity of TH-EM1 cells for CD9, CD13, CD73, CD90, CD105, and CD157 was revealed by flow cytometry. Likewise, the embryonic markers, SRY-box 2 (SOX-2) and the Nanog molecules, were detected in 76% and 52% of the cells, while fetal hemoglobin and a-globin were detected in 76% and 65% of TH-EM1 cells, respectively. By RHG banding, normal metaphases were observed, while the microarray chromosomal analysis showed gains of DNA sequences located on the segments 8p23.1, 11p15.5, and 12p11.23. The described in vitro cellular model can serve as a useful tool to study the pathogenesis of endometriosis and to improve the knowledge of molecular mechanisms controlling the endometriotic cell dissemination potential.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alifano M. Catamenial pneumothorax. Curr Opin Pulm Med. 2010;16(4):381–6.
Channabasavaiah AD, Joseph JV. Thoracic endometriosis: revisiting the association between clinical presentation and thoracic pathology based on thoracoscopic findings in 110 patients. Medicine (Baltimore). 2010;89(3):183–8.
Honore GM. Extrapelvic endometriosis. Clin Obstet Gynecol. 1999;42(3):699–711.
Nair SS, Nayar J. Thoracic endometriosis syndrome: a veritable Pandora’s box. J Clin Diagn Res. 2016;10(4):QR04–8.
Joseph J, Sahn SA. Thoracic endometriosis syndrome: new observations from an analysis of 110 cases. Am J Med. 1996;100(2):164–70.
Augoulea A, Lambrinoudaki I, Christodoulakos G. Thoracic endometriosis syndrome. Respiration. 2008;75(1):113–9.
Soriano D, Schonman R, Gat I, et al. Thoracic endometriosis syndrome is strongly associated with severe pelvic endometriosis and infertility. J Minim Invasive Gynecol. 2012;19(6):742–8.
Markham SM, Carpenter SE, Rock JA. Extrapelvic endometriosis. Obstet Gynecol Clin North Am. 1989;16(1):193–219.
Korom S, Canyurt H, Missbach A, et al. Catamenial pneumothorax revisited: clinical approach and systematic review of the literature. J Thorac Cardiovasc Surg. 2004;128(4):502–8.
Li F, Alderman MH. 3 rd, Tal A, et al. Hematogenous dissemination of mesenchymal stem cells from endometriosis. Stem Cells. 2018;36(6):881–90.
Maruyama T, Yoshimura Y. Stem cell theory for the pathogenesis of endometriosis. Front Biosci (Elite Ed). 2012;4:2754-2763.
Suginami H. A reappraisal of the coelomic metaplasia theory by reviewing endometriosis occurring in unusual sites and instances. Am J Obstet Gynecol. 1991;165(1):214–8.
Bulun SE, Monsivais D, Kakinuma T, et al. Molecular biology of endometriosis: from aromatase to genomic abnormalities. Semin Reprod Med. 2015;33(3):220–4.
Giudice LC. Clinical practice. Endometriosis. N Engl J Med. 2010;362(25):2389–98.
Jiang Y, Jahagirdar BN, Reinhardt RL, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002;418(6893):41–9.
Hayrabedyan S, Kyurkchiev S, Kehayov I. Endoglin (cd105) and S100A13 as markers of active angiogenesis in endometriosis. Reprod Biol. 2005;5(1):51–67.
Ortolan E, Arisio R, Morone S, et al. Functional role and prognostic significance of CD157 in ovarian carcinoma. J Natl Cancer Inst. 2010;102(15):1160–77.
Riekstina U, Cakstina I, Parfejevs V, et al. Embryonic stem cell marker expression pattern in human mesenchymal stem cells derived from bone marrow, adipose tissue, heart and dermis. Stem Cell Rev. 2009;5(4):378–86.
Alcayaga-Miranda F, Cuenca J, Luz-Crawford P, et al. Characterization of menstrual stem cells: angiogenic effect, migration and hematopoietic stem cell support in comparison with bone marrow mesenchymal stem cells. Stem Cell Res Ther. 2015;6:32.
Taylor HS. Endometrial cells derived from donor stem cells in bone marrow transplant recipients. JAMA. 2004;292(1):81–5.
Matsuzaki S, Canis M, Yokomizo R, Yaegashi N, Bruhat MA, Okamura K. Expression of erythropoietin and erythropoietin receptor in peritoneal endometriosis. Hum Reprod. 2003;18(1):152–6.
Yokomizo R, Matsuzaki S, Uehara S, Murakami T, Yaegashi N, Okamura K. Erythropoietin and erythropoietin receptor expression in human endometrium throughout the menstrual cycle. Mol Hum Reprod. 2002;8(5):441–6.
Song Y, Xiao L, Fu J, et al. Increased expression of the pluripotency markers sex-determining region Y-box 2 and Nanog homeobox in ovarian endometriosis. Reprod Biol Endocrinol. 2014;12:42.
Gotte M, Wolf M, Staebler A, Buchweitz O, Kiesel L, Schüring AN. Aberrant expression of the pluripotency marker SOX-2 in endometriosis. Fertil Steril. 2011;95(1):338–41.
Zheng C, Yang S, Guo Z, et al. Human multipotent mesenchymal stromal cells from fetal lung expressing pluripotent markers and differentiating into cell types of three germ layers. Cell Transplant. 2009;18(10):1093–109.
Haga T, Kurihara M, Kataoka H, Ebana H. Clinical-pathological findings of catamenial pneumothorax: comparison between recurrent cases and non-recurrent cases. Ann Thorac Cardiovasc Surg. 2014;20(3):202–6.
Haga T, Kumasaka T, Kurihara M, Kataoka H, Miura M. Immunohistochemical analysis of thoracic endometriosis. Pathol Int. 2013;63(9):429–34.
Martin JD Jr, Hauck AE. Endometriosis in the male. Am Surg. 1985;51(7):426–30.
Pinkert TC, Catlow CE, Straus R. Endometriosis of the urinary bladder in a man with prostatic carcinoma. Cancer. 1979;43(4):1562–7.
Hobbs JE, Bortnick AR. Endometriosis of the Lungs. Am J Obstetric Gynecol. 1940;40(5):832–43.
Sarma D, Iyengar P. Marotta TR, terBrugge KG, Gentili F, Halliday W. Cerebellar endometriosis. AJR Am J Roentgenol. 2004;182(6):1543–6.
von Stemm AM, Meigel WN, Scheidel P, Gocht A. Umbilical endometriosis. J Eur Acad Dermatol Venereol. 1999;12(1):30–2.
Cheng Y, Li L, Wang D, et al. Characteristics of human endometrium-derived mesenchymal stem cells and their tropism to endometriosis. Stem Cells Int. 2017;2017:4794827.
Gargett CE, Schwab KE, Deane JA. Endometrial stem/progenitor cells: the first 10 years. Hum Reprod Update. 2016;22(2):137–63.
Rolfing JH, Baatrup A, Stiehler M, Jensen J, Lysdahl H, Bünger C. The osteogenic effect of erythropoietin on human mesenchymal stromal cells is dose-dependent and involves non-hematopoietic receptors and multiple intracellular signaling pathways. Stem Cell Rev. 2014;10(1):69–78.
Zwezdaryk KJ, Coffelt SB, Figueroa YG, et al. Erythropoietin, a hypoxia-regulated factor, elicits a pro-angiogenic program in human mesenchymal stem cells. Exp Hematol. 2007;35(4):640–52.
Buhring HJ, Battula VL, Treml S, Schewe B, Kanz L, Vogel W. Novel markers for the prospective isolation of human MSC. Ann N Y Acad Sci. 2007;1106:262–71.
Dominici M, Le Blanc K, Mueller I, et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8(4):315–7.
Le Blanc K, Davies LC. MSCs-cells with many sides. Cytotherapy. 2018;20(3):273–8.
MacDonald JR, Ziman R, Yuen RK, Feuk L, Scherer SW. The database of genomic variants: a curated collection of structural variation in the human genome. Nucleic Acids Res. 2014;42(Database issue):D986–92.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Gogusev, J., Lepelletier, Y., El Khattabi, L. et al. Establishment and Characterization of a Stromal Cell Line Derived From a Patient With Thoracic Endometriosis. Reprod. Sci. 27, 1627–1636 (2020). https://doi.org/10.1007/s43032-020-00193-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-020-00193-8