Abstract
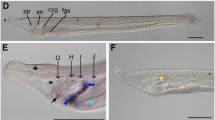
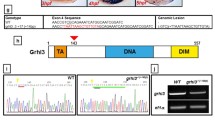
The Gli transcription factors are the primary mediators of Hedgehog (Hh) signaling. Vertebrate genomes contain multiple Gli paralogues with different functions downstream of Hh signal receipt, in part explaining the complexity of cellular responses to Hh that allow concentration-dependent target gene activation. Amphioxus is a chordate that split from the vertebrate lineage early in the evolution of chordates, before the genome duplications that occurred in early vertebrate evolution. It has a single Gli gene whose transcripts can be alternately spliced to yield two protein isoforms called GliS and GliL. We generated two knockout mutations in amphioxus Gli, one that affects the whole gene and a second that only affects GliL. Both knockouts showed major morphological and molecular defects in the development of left–right asymmetry, a phenotype that is similar but not identical to that previously found in Hh mutants. Hh signaling also patterns the amphioxus neural tube. Here, however, knockout of GliL showed no identifiable phenotype, while knockout of the full gene showed only small changes to the expression of one gene family, Olig. Other genes that were prominently affected by Hh knockout were not altered in expression in either knockout. Reasons for the differences between Hh and Gli knockouts in the pharynx and neural tube are discussed in the context of the likely different functions of amphioxus Gli isoforms.





Similar content being viewed by others
Data availability
All data generated during this study are included in this published article.
References
Abbasi AA, Goode DK, Amir S, Grzeschik KH (2009) Evolution and functional diversification of the GLI family of transcription factors in vertebrates. Evol Bioinform Online 5:5–13
Briscoe J, Small S (2015) Morphogen rules: design principles of gradient-mediated embryo patterning. Development 142:3996–4009
Candiani S, Oliveri D, Parodi M, Bertini E, Pestarino M (2006) Expression of AmphiPOU-IV in the developing neural tube and epidermal sensory neural precursors in amphioxus supports a conserved role of class IV POU genes in the sensory cells development. Dev Genes Evol 216:623–633
Candiani S, Holland ND, Oliveri D, Parodi M, Pestarino M (2008) Expression of the amphioxus Pit-1 gene (AmphiPOU1F1/Pit-1) exclusively in the developing preoral organ, a putative homolog of the vertebrate adenohypophysis. Brain Res Bull 75:324–330
Elliott KH, Brugmann SA (2019) Sending mixed signals: Cilia-dependent signaling during development and disease. Dev Biol 447:28–41
Falkenstein KN, Vokes SA (2014) Transcriptional regulation of graded Hedgehog signaling. Semin Cell Dev Biol 33:73–80
Holland LZ, Albalat R, Azumi K, Benito-Gutierrez E, Blow MJ, Bronner-Fraser M, Brunet F, Butts T, Candiani S, Dishaw LJ, Ferrier DE, Garcia-Fernandez J, Gibson-Brown JJ, Gissi C, Godzik A, Hallbook F, Hirose D, Hosomichi K, Ikuta T, Inoko H et al (2008) The amphioxus genome illuminates vertebrate origins and cephalochordate biology. Genome Res 18:1100–1111
Hu G, Li G, Wang H, Wang Y (2017) Hedgehog participates in the establishment of left-right asymmetry during amphioxus development by controlling Cerberus expression. Development 144:4694–4703
Hu GW, Li G, Wang YQ (2021) Hedgehog signaling controls mouth opening in the amphioxus. Zool Lett 7:16
Huangfu D, Anderson KV (2006) Signaling from Smo to Ci/Gli: conservation and divergence of Hedgehog pathways from Drosophila to vertebrates. Development 133:3–14
Hui CC, Angers S (2011) Gli proteins in development and disease. Annu Rev Cell Dev Biol 27:513–537
Jiang J, Hui CC (2008) Hedgehog signaling in development and cancer. Dev Cell 15:801–812
Karlstrom RO, Tyurina OV, Kawakami A, Nishioka N, Talbot WS, Sasaki H, Schier AF (2003) Genetic analysis of zebrafish gli1 and gli2 reveals divergent requirements for gli genes in vertebrate development. Development 130:1549–1564
Ke Z, Kondrichin I, Gong Z, Korzh V (2008) Combined activity of the two Gli2 genes of zebrafish play a major role in Hedgehog signaling during zebrafish neurodevelopment. Mol Cell Neurosci 37:388–401
Knight RD, Panopoulou GD, Holland PW, Shimeld SM (2000) An amphioxus Krox gene: insights into vertebrate hindbrain evolution. Dev Genes Evol 210:518–521
Leung B, Shimeld SM (2019) Evolution of vertebrate spinal cord patterning. Dev Dyn 248:1028–1043
Li G, Shu Z, Wang Y (2013) Year-round reproduction and induced spawning of Chinese amphioxus, Branchiostoma belcheri, in laboratory. PLoS ONE 8:e75461
Li G, Feng J, Lei Y, Wang J, Wang H, Shang LK, Liu DT, Zhao H, Zhu Y, Wang YQ (2014) Mutagenesis at specific genomic loci of amphioxus Branchiostoma belcheri using TALEN method. J Genet Genom 41:215–219
Li G, Liu X, Xing C, Zhang H, Shimeld SM, Wang Y (2017) Cerberus-Nodal-Lefty-Pitx signaling cascade controls left-right asymmetry in amphioxus. Proc Natl Acad Sci USA 114:3684–3689
Litingtung Y, Chiang C (2000) Specification of ventral neuron types is mediated by an antagonistic interaction between Shh and Gli3. Nat Neurosci 3:979–985
Liu X, Li G, Feng J, Yang X, Wang YQ (2013) An efficient microinjection method for unfertilized eggs of Asian amphioxus Branchiostoma belcheri. Dev Genes Evol 223:269–278
Park HL, Bai C, Platt KA, Matise MP, Beeghly A, Hui CC, Nakashima M, Joyner AL (2000) Mouse Gli1 mutants are viable but have defects in SHH signaling in combination with a Gli2 mutation. Development 127:1593–1605
Putnam NH, Butts T, Ferrier DE, Furlong RF, Hellsten U, Kawashima T, Robinson-Rechavi M, Shoguchi E, Terry A, Yu JK, Benito-Gutierrez EL, Dubchak I, Garcia-Fernandez J, Gibson-Brown JJ, Grigoriev IV, Horton AC, de Jong PJ, Jurka J, Kapitonov VV, Kohara Y et al (2008) The amphioxus genome and the evolution of the chordate karyotype. Nature 453:1064–1071
Ren Q, Zhong Y, Huang X, Leung B, Xing C, Wang H, Hu G, Wang Y, Shimeld SM, Li G (2020) Step-wise evolution of neural patterning by Hedgehog signalling in chordates. Nat Ecol Evol 4:1247–1255
Sagner A, Briscoe J (2019) Establishing neuronal diversity in the spinal cord: a time and a place. Development 146:182154
Shimeld SM (2008) C2H2 zinc finger genes of the Gli, Zic, KLF, SP, Wilms’ tumour, Huckebein, Snail, Ovo, Spalt, Odd, Blimp-1, Fez and related gene families from Branchiostoma floridae. Dev Genes Evol 218:639–649
Shimeld SM, van den Heuvel M, Dawber R, Briscoe J (2007) An amphioxus Gli gene reveals conservation of midline patterning and the evolution of hedgehog signalling diversity in chordates. PLoS ONE 2:e864
Venkatesh TV, Holland ND, Holland LZ, Su MT, Bodmer R (1999) Sequence and developmental expression of amphioxus AmphiNk2-1: insights into the evolutionary origin of the vertebrate thyroid gland and forebrain. Dev Genes Evol 209:254–259
von Mering C, Basler K (1999) Distinct and regulated activities of human Gli proteins in Drosophila. Curr Biol 9:1319–1322
Wang H, Li G, Wang YQ (2015) Generating amphioxus Hedgehog knockout mutants and phenotype analysis. Hereditas 37:1036–1043 (in Chinese with English abstract)
Yu JK, Holland LZ (2009) Amphioxus whole-mount in situ hybridization. Cold Spring Harb Protoc 2009:pdb.prot5286.
Yuan L, Wang Y, Li G (2020) Differential expression pattern of two Brachyury genes in amphioxus embryos. Gene Expr Patterns 38:119152
Acknowledgements
We thank the two anonymous reviewers for their critical and helpful comments. This work was supported by grants from the National Natural Science Foundation of China (Nos. 32070815, 32070458, 31872186 and 32061160471) and from the Youth Innovation Fund Project of Xiamen (3502Z20206032).
Author information
Authors and Affiliations
Contributions
GL, SS and YW designed the experiments. QR generated the mutants, and XH performed all other experiments. QR and XH prepared the figures. All authors interpreted the data. GL and SS wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Animal and human rights statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Special Topic: EvoDevo.
Edited by Jiamei Li.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, X., Ren, Q., Wang, Y. et al. Amphioxus Gli knockout disrupts the development of left–right asymmetry but has limited impact on neural patterning. Mar Life Sci Technol 5, 492–499 (2023). https://doi.org/10.1007/s42995-023-00195-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42995-023-00195-w