Abstract
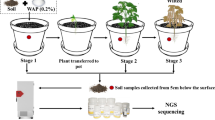
Soils have become an important sink for antibiotic resistance genes (ARGs). To better understand the impacts of ARGs on the soil ecosystem, the transport of ARGs is a basic question. So far, however, the role of soil animals in the dispersal of ARGs is not understood. Here, two treatments (without collembolans and with collembolans) were established, each treatment included unamended and manure-amended soil, and soil samples were collected at 14, 28 and 56 days after incubation. The effects of the collembolan Folsomia Candida on dispersal of ARGs in the soil ecosystem were explored using high-throughput qPCR combined with Illumina sequencing. As the culture time increased, more shared ARGs and OTUs were detected between the unamended and manured soil, especially in the treatment with collembolans. Vancomycin, aminoglycoside and MLSB genes may have been more readily transported by the collembolan. On the 28th day after incubation, a high abundance of mobile genetic elements (MGEs) was found in the treatment with collembolans. These results clearly reveal that collembolans can accelerate the dispersal of ARGs in the soil ecosystem. Procrustes analysis and the Mantel test both indicate that soil bacterial communities were significantly correlated with ARG profiles. Furthermore, partial redundancy analysis indicates that soil bacterial communities can explain 41.28% of the variation in ARGs. These results suggest that the change of soil microbial community have an important contribution to the dispersal of ARGs by the collembolan.
Article PDF
Similar content being viewed by others
References
Ashbolt, N.J., Amézquita, A., Backhaus, T., Borriello, P., Brandt, K.K., Collignon, P., Coors, A., Finley, R., Gaze, W.H., Heberer, T., Lawrence, J.R., Larsson, D.G., McEwen, S.A., Ryan, J.J., Schönfeld, J., Silley, P., Snape, J.R., Van den Eede, C., Topp, E., 2013. Human Health Risk Assessment (HHRA) for environmental development and transfer of antibiotic resistance. Environmental Health Perspectives 121, 993–1001.
Bengtsson-Palme, J., Angelin, M., Huss, M., Kjellqvist, S., Kristiansson, E., Palmgren, H., Larsson, D.G., Johansson, A., 2015. The human gut microbiome as a transporter of antibiotic resistance genes between continents. Antimicrobial Agents and Chemotherapy 59, 6551–6560.
Bengtsson-Palme, J., Kristiansson, E., Larsson, D.G.J., 2018. Environmental factors influencing the development and spread of antibiotic resistance. FEMS Microbiology Reviews 42, 68–80.
Caporaso, J.G., Bittinger, K., Bushman, F.D., DeSantis, T.Z., Andersen, G.L., Knight, R., 2010a. PyNAST: a flexible tool for aligning sequences to a template alignment. Bioinformatics (Oxford, England) 26, 266–267.
Caporaso, J.G., Kuczynski, J., Stombaugh, J., Bittinger, K., Bushman, F.D., Costello, E.K., Fierer, N., Peña, A.G., Goodrich, J.K., Gordon, J.I., Huttley, G.A., Kelley, S.T., Knights, D., Koenig, J.E., Ley, R.E., Lozupone, C.A., McDonald, D., Muegge, B.D., Pirrung, M., Reeder, J., Sevinsky, J.R., Turnbaugh, P.J., Walters, W.A., Widmann, J., Yatsunenko, T., Zaneveld, J., Knight, R., 2010b. QIIME allows analysis of high-throughput community sequencing data. Nature Methods 7, 335–336.
Chen, Q., An, X., Li, H., Su, J., Ma, Y., Zhu, Y.G., 2016. Long-term field application of sewage sludge increases the abundance of antibiotic resistance genes in soil. Environment International 92–93, 1–10.
Chu, B.T.T., Petrovich, M.L., Chaudhary, A., Wright, D., Murphy, B., Wells, G., Poretsky, R., 2018. Metagenomics reveals the impact of wastewater treatment plants on the dispersal of microorganisms and genes in aquatic sediments. Applied and Environmental Microbiology 84, e02168–e17.
Davidson, D.A., Grieve, I.C., 2006. The influence of soil fauna on soil structural attributes under a limed and untreated upland grassland. Land Degradation & Development 17, 393–400.
Duranti, S., Lugli, G.A., Mancabelli, L., Turroni, F., Milani, C., Mangifesta, M., Ferrario, C., Anzalone, R., Viappiani, A., van Sinderen, D., Ventura, M., 2017. Prevalence of antibiotic resistance genes among human gut-derived bifidobacteria. Applied and Environmental Microbiology 83, e02894–e16.
Edgar, R.C., 2010. Search and clustering orders of magnitude faster than BLAST. Bioinformatics (Oxford, England) 26, 2460–2461.
Forsberg, K.J., Patel, S., Gibson, M.K., Lauber, C.L., Knight, R., Fierer, N., Dantas, G., 2014. Bacterial phylogeny structures soil resistomes across habitats. Nature 509, 612–616.
Fountain, M.T., Hopkin, S.P., 2005. Folsomia Candida (Collembola): a “standard” soil arthropod. Annual Review of Entomology 50, 201, 201–222.
Garofalo, C., Vignaroli, C., Zandri, G., Aquilanti, L., Bordoni, D., Osimani, A., Clementi, F., Biavasco, F., 2007. Direct detection of antibiotic resistance genes in specimens of chicken and pork meat. International Journal of Food Microbiology 113, 75–83.
Gaze, W.H., Zhang, L., Abdouslam, N.A., Hawkey, P.M., Calvo-Bado, L., Royle, J., Brown, H., Davis, S., Kay, P., Boxall, A.B.A., Wellington, E.M., 2011. Impacts of anthropogenic activity on the ecology of class 1 integrons and integron-associated genes in the environment. ISME Journal 5, 1253–1261.
Hopkin, S.P., 1997. Biology of the Springtails (Insecta: Collembola). Oxford University Press.
Johnson, D.L., Wellington, W.G., 1983. Dispersal of the collembolan Folsomia Candida Willem, as a function of age. Canadian Journal of Zoology 61, 2535–2538.
Lee, Q., Widden, P., 1996. Folsomia candida, a “fungivorous” collembolan, feeds preferentially on nematodes rather than soil fungi. Soil Biology & Biochemistry 28, 689–690.
Maaß, S., Daphi, D., Lehmann, A., Rillig, M.C., 2017. Transport of microplastics by two collembolan species. Environ Pollut 225, 456–459.
McDonald, D., Price, M.N., Goodrich, J., Nawrocki, E.P., DeSantis, T. Z., Probst, A., Andersen, G.L., Knight, R., Hugenholtz, P., 2012. An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. ISME Journal 6, 610–618.
Nakamori, T., Suzuki, A., 2005. Spore-breaking capabilities of collembolans and their feeding habitat within sporocarps. Pedobiologia 49, 261–267.
Oksanen, J., Blanchet, F.G., Kindt, R., Legendre, P., Minchin, P.R., O’Hara, R.B., Simpson, G.L., Solymos, P., Stevens, M.H.H., Wagner, H., 2015. vegan: Community Ecology Package. R package version 2.3–1. Lorenzo Cachón Rodríguez 48, págs. 103–132.
Organization, W.H., 2014. Antimicrobial resistance: global report on surveillance. Australasian Medical Journal 7, 237.
Paradelo, R., Barral, M.T., 2009. Effect of moisture and disaggregation on the microbial activity of soil. Soil & Tillage Research 104, 317–319.
Price, M.N., Dehal, P.S., Arkin, A.P., 2010. FastTree 2—approximately maximum-likelihood trees for large alignments. PLoS One 5, e9490.
RCoreTeam, 2017. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria; http://www.R-project.org.
Roberts, D.W., 2016. labdsv: Ordination and Multivariate Analysis for Ecology; http://th.archive.ubuntu.com/cran/web/packages/labdsv/.
Rusek, J., 1998. Biodiversity of Collembola and their functional role in the ecosystem. Biodiversity and Conservation 7, 1207–1219.
Siddiky, M.R.K., Schaller, J., Caruso, T., Rillig, M.C., 2012. Arbuscular mycorrhizal fungi and Collembola non-additively increase soil aggregation. Soil Biology & Biochemistry 47, 93–99.
Thiele-Bruhn, S., Bloem, J., de Vries, F.T., Kalbitz, K., Wagg, C., 2012. Linking soil biodiversity and agricultural soil management. Current Opinion in Environmental Sustainability 4, 523–528.
Zhou, H., Wang, X., Li, Z., Kuang, Y., Mao, D., Luo, Y., 2018. Occurrence and distribution of urban dust-associated bacterial antibiotic resistance in northern China. Environmental Science & Technology Letters 5, 50–55.
Zhu, B., Chen, Q., Chen, S., Zhu, Y.G., 2017a. Does organically produced lettuce harbor higher abundance of antibiotic resistance genes than conventionally produced? Environment International 98, 152–159.
Zhu, D., An, X.L., Chen, Q.L., Yang, X.R., Christie, P., Ke, X., Wu, L.H., Zhu, Y.G., 2018a. Antibiotics disturb the microbiome and increase the incidence of resistance genes in the gut of a common soil collembolan. Environmental Science & Technology 52, 3081–3090.
Zhu, D., Bi, Q.F., Xiang, Q., Chen, Q.L., Christie, P., Ke, X., Wu, L.H., Zhu, Y.G., 2018b. Trophic predator-prey relationships promote transport of microplastics compared with the single Hypoaspis aculeifer and Folsomia candida. Environ Pollut 235, 150–154.
Zhu, D., Ke, X., Wu, L., Christie, P., Luo, Y., 2016. Biological transfer of dietary cadmium in relation to nitrogen transfer and 15N fractionation in a soil collembolan-predatory mite food chain. Soil Biology & Biochemistry 101, 207–216.
Zhu, P., Kong, T., Tang, X., Wang, L., 2017b. Well-defined porous membranes for robust omniphobic surfaces via microfluidic emulsion templating. Nature Communications 8, 15823.
Zhu, Y.G., Johnson, T.A., Su, J.Q., Qiao, M., Guo, G.X., Stedtfeld, R. D., Hashsham, S.A., Tiedje, J.M., 2013. Diverse and abundant antibiotic resistance genes in Chinese swine farms. Proceedings of the National Academy of Sciences of the United States of America 110, 3435–3440.
Acknowledgments
This work was funded by the National Natural Science Foundation of China (41571130063), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB15020302 and XDB15020402) and the National Key Research and Development Program of China-International collaborative project from Ministry of Science and Technology (Grant No. 2017YFE0107300).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, D., Wang, HT., Zheng, F. et al. Collembolans accelerate the dispersal of antibiotic resistance genes in the soil ecosystem. Soil Ecol. Lett. 1, 14–21 (2019). https://doi.org/10.1007/s42832-019-0002-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42832-019-0002-1