Abstract
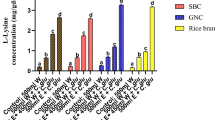
This manuscript deals with cordycepin, an interesting secondary compound produced from entomopathogenic fungus, Cordyceps. It has attracted commercial interest due to its immense pharmacological importance beneficial to human health. In this study, the contents of cordycepin and its derivatives, like adenine and adenosine, were evaluated through solid-state fermentation using combinations of various grains as substrate. Treatment with grain combination numbers 2, 7, 8, and 9 exhibited higher cordycepin content (1.621, 1.929, 1.895, and 1.996 mg/g cordycepin, respectively) than control (rice). The grain combination number 7 exhibited significantly higher adenine content (700 mg/g) than the control and all other combinations. Treatments with grain combination numbers 2, 5, and 7 exhibited higher adenosine content (2.719, 2.938, and 3.392 mg/g, respectively); however, no significant increase in adenosine content was noted in any treatments. The biomass including fresh mycelium and fruit body was found higher in grain combination numbers 7 and 9, leading to enhanced cordycepin content. Overall, the increase in the fresh biomass significantly enhanced cordycepin accumulation. The level of cordycepin was recorded as higher than that of its derivatives, adenosine and adenine. The grain combination of rice, wheat, jowar, bajra, and sugarcane bagasse added to basal medium exhibited the highest cordycepin content and was found suitable for solid-state fermentation of Cordyceps militaris. To our understanding, the present study is the first to use combinations of cereals for the production of cordycepin from C. militaris.





Similar content being viewed by others
Data availability
Data made available on request.
References
Ma L, Song Z, Mei D (2015) Cordycepin from Cordyceps militaris prevents hyperglycemia in alloxan-induced diabetic mice. Nutr Res 35:431–439. https://doi.org/10.1016/j.nutres.2015.04.011
Olatunji OJ, Tang J, Tola A, Auberon F, Oluwaniyi O, Ouyang Z (2018) The genus Cordyceps: an extensive review of its traditional uses, phytochemistry and pharmacology. Fitoter 129:293–316. https://doi.org/10.1016/j.fitote.2018.05.010
Wang M, Meng XY, Le Yang R, Qin T, Wang XY, Zhang KY, Fei CZ, Li Y, Hu YL, Xue FQ (2012) Cordyceps militaris polysaccharides can enhance the immunity and antioxidation activity in immunosuppressed mice. Carbohyd Polym 89:461–466. https://doi.org/10.1016/j.carbpol.2012.03.029
Gregori A (2014) Cordycepin production by C. militaris cultivation on spent brewery grains. Acta Biol Slov 57(2):45–52
Tian X, Li Y, Shen Y, Li Q, Wang Q, Feng L (2015) Apoptosis and inhibition of proliferation of cancer cells induced by cordycepin. Oncol Lett 10:595–599. https://doi.org/10.3892/ol.2015.3273
Cho HJ, Cho JY, Rhee MH, Lim CR, Park HJ (2006) Cordycepin (3′-deoxyadenosine) inhibits human platelet aggregation induced by U46619, a TXA2 analogue. J Pharm Pharmacol 58:1677–1682. https://doi.org/10.1211/jpp.58.12.0016
Guo P, Kai Q, Gao J, Lian ZQ, Wu CM, Wu CA, Zhu HB (2010) Cordycepin prevents hyperlipidemia in hamsters fed a high-fat diet via activation of AMP-activated protein kinase. J Pharmacol Sci 113:395–403. https://doi.org/10.1254/jphs.10041FP
Sugar AM, McCaffrey RP (1998) Antifungal activity of 3’-deoxyadenosine (cordycepin)”. Antimicrob agents chemother 42:61424–61427. https://doi.org/10.1128/AAC.42.6.1424
Cao T, Xu R, Xu Y, Liu Y, Wan Q (2019) The protective effect of Cordycepin on diabetic nephropathy through autophagy induction in vivo and in vitro. Int Urol Nephrol 51:1883–1892. https://doi.org/10.1007/s11255-019-02241-y
Ma L, Zhang S, Du M (2015) Cordycepin from Cordyceps militaris prevents hyperglycemia in alloxan-induced diabetic mice. Nutr Res 35:431–439. https://doi.org/10.1016/j.nutres.2015.04.011
An Y, Li Y, Wang X, Chen Z, Xu H, Wu L, Li S, Wang C, Luan W, Wang X (2018) Cordycepin reduces weight through regulating gut microbiota in high-fat diet-induced obese rats. Lipids Health Dis 17:276. https://doi.org/10.1186/s12944-018-0910-6
Kitakaze M, Hori M (2000) Adenosine therapy: a new approach to chronic heart failure”. Expert Opin Investig Drugs 9(11):2519–2535. https://doi.org/10.1517/13543784.9.11.2519
Jiang Y, Wong JH, Fu M, Ng TB, Liu ZK, Wang CR, Li N, Qiao WT, Wen TY, Liu F (2011) Isolation of adenosine, iso-sinensetin and dimethylguanosine with antioxidant and HIV-1 protease inhibiting activities from fruiting bodies of Cordyceps militaris. Phytomed 15,18(2–3):189–93. https://doi.org/10.1016/j.phymed.2010.04.010
Oh J, Yoon DH, Shrestha B, Choi HK, Sung GH (2019) Metabolomic profiling reveals enrichment of cordycepin in senescence process of Cordyceps militaris fruit bodies. J Microbiol 57:54–63. https://doi.org/10.1007/s12275-019-8486-z
Pang F, Wang L, Jin Y, Guo L, Song L, Liu G, Feng C (2018) Transcriptome analysis of Paecilomyces hepiali at different growth stages and culture additives to reveal putative genes in cordycepin biosynthesis. Genomics 110:162–170. https://doi.org/10.1016/j.ygeno.2017.09.008
Zhou X, Cai G, He Y, Tong G (2016) Separation of cordycepin from Cordyceps militaris fermentation supernatant using preparative HPLC and evaluation of its antibacterial activity as an NAD(+)-dependent DNA ligase inhibitor. Exp Ther Med 12:1812–1816. https://doi.org/10.3892/etm.2016.3536
Lin Q, Long L, Wu L, Zhang F, Wu S, Zhang W, Sun X (2016) Evaluation of different agricultural wastes for the production of fruiting bodies and bioactive compounds by medicinal mushroom Cordyceps militaris. J Sci Food Agric. https://doi.org/10.1002/jsfa.8097
Fan DD, Wang W, Zhong JJ (2012) Enhancement of cordycepin production in submerged cultures of Cordyceps militaris by addition of ferrous sulfate. Biochem Eng J 60:30–35. https://doi.org/10.1016/j.bej.2011.09.014
Yang D, Yaguchi T, Yamamoto H, Nishizaki T (2007) Intracellularly transported adenosine induces apoptosis in HuH-7 human hepatoma cells by downregulating c-FLIP expression causing caspase-3/-8 activation”. Biochem Pharm 73(10):1665–1675. https://doi.org/10.1016/j.bcp.2007.01.020
Nakav S, Chaimovitz C, Sufaro Y (2008) Anti-inflammatory preconditioning by agonists of adenosine A1 receptor. PLoS One 3(5):e2107. https://doi.org/10.1371/journal.pone.0002107
Manfred JP, Sparks Jr HV (1982) Adenosine’s role in coronary vasodilation induced by atrial pacing and norepinephrine. Am J Physiol—Endocrinol Metab 243, 4. H536–H545
Ontyd J, Schrader J (1984) Measurement of adenosine, inosine, and hypoxanthine in human plasma”. J Chromatogr 307(2):404–409
Tsai YJ, Lin LC, Tsai TH (2010) Pharmacokinetics of ade nosine and cordycepin, a bioactive constituent of Cordyceps sinensis in rat”. J Agric Food Chem 58(8):4638–4643. https://doi.org/10.1021/jf100269g
Yang FQ, Li SP, Li P, Wang YT (2007) Optimization of CEC for simultaneous determination of eleven nucleosides and nucleobases in Cordyceps using central composite design”. Electro 28(11):1681–1688. https://doi.org/10.1002/elps.200600416
Yang D, Yaguchi T, Lim CR, Ishizawa Y, Nakano T, Nishizaki T (2010) Tuning of apoptosis-mediator gene transcription in HepG2 human hepatoma cells through an adenosine signal”. Can Lett 291(2):225–229. https://doi.org/10.1016/j.canlet.2009.10.016
Huang SJ, Huang FK, Li YS, Shu-Yao T (2017) The quality improvement of solid-state fermentation with Cordyceps militaris by UVB irradiation. Food Technol Biotechnol 55:445. https://doi.org/10.17113/ftb.55.04.17.5235
Adnan M, Ashraf SA, Khan S, Alshammari E, Awadelkareem AM (2017) Effect of pH, temperature and incubation time on cordycepin production from Cordyceps militaris using solid-state fermentation on various substrates. CyTA-J Food 15:617–621. https://doi.org/10.1080/19476337.2017.1325406
Panjikkaran ST, Mathew D (2013) An environmentally friendly and cost-effective technique for the commercial cultivation of oyster mush room Pleurotus florida (Mont.) Singer]. J Sci Food Agric 93:680–684. https://doi.org/10.1002/jsfa.5827
Yang D, Liang J, Wang Y, Sun F, Tao H, Xu Q (2016) Tea waste: an effective and economic substrate for oyster mushroom cultivation. J Sci Food Agric 96:680–684. https://doi.org/10.1002/jsfa.7140
Huang L, Li Q, Chen Y, Wang X, Zhou X (2009) (2009) Determination and analysis of cordycepin and adenosine in the products of Cordyceps spp. Afr J Microbiol Res 3:957–961
Shrestha B, Zhang W, Zhang Y, Liu X (2012) The medicinal fungus Cordyceps militaris: research and development. Mycol Prog 11:599–614. https://doi.org/10.1007/s11557-012-0825-y
Chiang SS, Liang ZC, Wang YC, Liang CH (2017) Effect of lightemitting bits human platelet aggregation induced by U46619, a TXA2 analogue. J Pharm Pharmacol 58:1677–1682. https://doi.org/10.1211/jpp.58.12.0016
Wen TC, Kang C, Meng ZB, Qi YB, Hyde KD, Kang JC (2016) Enhanced production of cordycepin by solid state fermentation of Cordyceps militaris using additives. Chiang Mai J Sci 43(5):972–984. https://doi.org/10.1016/j.enzmictec.2006.09.008
Chen JM, Yu WW, Wu H, Wu DD, Lai FR (2012) Analysis of the nutritional components of corn cobs. Mod Food Sci Technol 28:1073–1075
Gu YX, Wang ZS, Li SX, Yuan QS (2007) Effect of multiple factors on accumulation of nucleosides and bases in Cordyceps militaris. Food Chem 102(4):1304–1309
Jian LR, Li (2017) pin and adenosine production of Cordyceps militaris cultured on wheat solid substrate. Acad J Agric Res 5(10):279–286. https://doi.org/10.15413/ajar.2017.0138
Dong JZ, Lei C, Zheng XJ, Ai XR, Wang Y, Wang Q (2013) Light wavelengths regulate growth and active components of Cordyceps militaris fruit bodies. J Food Biochem 37:578–584. https://doi.org/10.1111/jfbc.12009
Wu CY, Liang ZC, Tseng CY, Hu SH (2016) Effects of illumination pattern during cultivation of fruiting Body and bioactive compound production by the caterpillar medicinal mushroom, Cordyceps militaris (Ascomycetes). Int J Med Mushrooms 18:589–597. https://doi.org/10.1615/intjmedmushrooms.v18.i7.40
Wu CY, Liang CH, Liang ZC (2022) Enhanced production of fruiting bodies and bioactive compounds of Cordyceps militaris with grain substrates and cultivation patterns. J Tai Inst Chem Eng 132:104138
Lim L, Lee C, Chang E (2012) Optimization of solid state culture conditions for the production of adenosine, cordycepin, and D-mannitol in fruiting bodies of medicinal caterpillar fungus Cordyceps militaris (L.:Fr.) Link (Ascomycetes). Int J Med Mushrooms 14:181–187. https://doi.org/10.1615/intjmedmushr.v14.i2.60
Wu F, Chen Y, Chang S, Shih I (2013) Cultivation of medicinal caterpillar fungus, Cordyceps militaris (Ascomycetes), and production of cordycepin using the spent medium from levan fermentation. Int J Med Mushroom 15:393–405. https://doi.org/10.1615/intjmedmushr.v15.i4.70
Cohen N, Cohen J, Asatiani MD, Varshney VK, Yu HT, Yang YC, Li YH, Mau JL, Wasser SP (2014) Chemical composition and nutritional and medicinal value of fruit bodies and submerged cultured mycelia of culinary-medicinal higher Basidiomycetes mushrooms. Int J Med Mushrooms 16(3):273–291. https://doi.org/10.1615/intjmedmushr.v16.i3.80
Yang F, Li D, Feng K, Hu DJ, Li SP (2010) Determination of nucleotides, nucleosides and their transformation products in Cordyceps by ion-pairing reversed-phase liquid chromatography-mass spectrometry. J Chromatogr A 1217:5501–5510. https://doi.org/10.1016/j.chroma.2010.06.062
Chassy BM, Suhadolnik RJ (1969) Nucleoside antibiotics IV. Metabolic fate of adenosine and cordycepin by Cordyceps militaris during cordycepin biosynthesis. Bioch Bioph Acta 182:2:307–315
Kredich NM, Guarino AJ (1961) Studies on the biosynthesis of cordycepin. Biochim Biophys Acta 47:499–535
Suhadolnik RJ, Cory JG (1964) Further evidence for the biosynthesis of cordycepin and proof of the structure of 3-deoxyribose. Biochim Et Biophys Acta 91:661–662
Masuda M, Urabe E, Honda H, Sakurai A, Sakakibara M (2007) Enhanced production of cordycepin by surface culture using the medicinal mushroom Cordyceps militaris. Enzym Microb Technol 39:641–646
Acknowledgements
The authors thank SERB, New Delhi, for the financial support under the SERB-TAR, (TAR/2019/000051) authorities of Savitribai Phule Pune University, and the Director, MACS-Agharkar Research Institute, Pune, for providing all laboratory facilities.
Author information
Authors and Affiliations
Contributions
MB performed the experimental work. SS is responsible for the supervision, data monitoring, and data analysis.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Rosane Freitas Schwan
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Borde, M., Singh, S.K. Enhanced production of cordycepin under solid-state fermentation of Cordyceps militaris by using combinations of grains/substrates. Braz J Microbiol 54, 2765–2772 (2023). https://doi.org/10.1007/s42770-023-01169-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42770-023-01169-x