Abstract
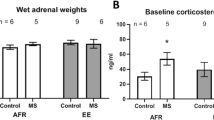
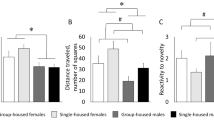
Social stress is associated with depression and anxiety, physiological disruptions, and altered brain morphology in central stress circuitry across development. Environmental enrichment strategies may improve responses to social stress. Socially monogamous prairie voles exhibit analogous social and emotion-related behaviors to humans, with potential translational insight into interactions of social stress, age, and environmental enrichment. This study explored the effects of social isolation and environmental enrichment on behaviors related to depression and anxiety, physiological indicators of stress, and dendritic structural changes in amygdala and hippocampal subregions in young adult and aging prairie voles. Forty-nine male prairie voles were assigned to one of six groups divided by age (young adult vs. aging), social structure (paired vs. isolated), and housing environment (enriched vs. non-enriched). Following 4 weeks of these conditions, behaviors related to depression and anxiety were investigated in the forced swim test and elevated plus maze, body and adrenal weights were evaluated, and dendritic morphology analyses were conducted in hippocampus and amygdala subregions. Environmental enrichment decreased immobility duration in the forced swim test, increased open arm exploration in the elevated plus maze, and reduced adrenal/body weight ratio in aging and young adult prairie voles. Age and social isolation influenced dendritic morphology in the basolateral amygdala. Age, but not social isolation, influenced dendritic morphology in the hippocampal dentate gyrus. Environmental enrichment did not influence dendritic morphology in either brain region. These data may inform interventions to reduce the effects of social stressors and age-related central changes associated with affective behavioral consequences in humans.













Similar content being viewed by others
References
Adams, K. B. (2001). Depressive symptoms, depletion, or developmental change? Withdrawal, apathy, and lack of vigor in the geriatric depression scale. The Gerontologist, 41(6), 768–777. https://doi.org/10.1093/geront/41.6.768
Almaguer, W., Estupiñán, B., Uwe Frey, J., & Bergado, J. A. (2002). Aging impairs amygdala-hippocampus interactions involved in hippocampal LTP. Neurobiology of Aging, 23(2), 319–324. https://doi.org/10.1016/S0197-4580(01)00278-0
Ashokan, A., Hegde, A., & Mitra, R. (2016). Short-term environmental enrichment is sufficient to counter stress-induced anxiety and associated structural and molecular plasticity in basolateral amygdala. Psychoneuroendocrinology, 69, 189–196. https://doi.org/10.1016/j.psyneuen.2016.04.009
Aylett, E., Small, N., & Bower, P. (2018). Exercise in the treatment of clinical anxiety in general practice – A systematic review and meta-analysis. BMC Health Services Research, 18, 559. https://doi.org/10.1186/s12913-018-3313-5
Bartsch, T., & Wulff, P. (2015). The hippocampus in aging and disease: from plasticity to vulnerability. Neuroscience, 309, 1–16. https://doi.org/10.1016/j.neuroscience.2015.07.084
Bland, A. R., Roiser, J. P., Mehta, M. A., Sahakian, B. J., Robbins, T. W., & Elliott, R. (2022). The impact of COVID-19 social isolation on aspects of emotional and social cognition. Cognition and Emotion, 36(1), 49–58. https://doi.org/10.1080/02699931.2021.1892593
Bosch, O. J., Nair, H. P., Ahern, T. H., Neumann, I. D., & Young, L. J. (2009). The CRF system mediates increased passive stress-coping behavior following the loss of a bonded partner in a monogamous rodent. Neuropsychopharmacology, 34(6), 1406–1415. https://doi.org/10.1038/npp.2008.154
Brenes, J. C., & Fornaguera, J. (2008). Effects of environmental enrichment and social isolation on sucrose consumption and preference: Associations with depressive-like behavior and ventral striatum dopamine. Neuroscience Letters, 436(2), 278–282. https://doi.org/10.1016/j.neulet.2008.03.045
Brenes, J. C., Rodríguez, O., & Fornaguera, J. (2008). Differential effect of environment enrichment and social isolation on depressive-like behavior, spontaneous activity and serotonin and norepinephrine concentration in prefrontal cortex and ventral striatum. Pharmacology Biochemistry and Behavior, 89(1), 85–93. https://doi.org/10.1016/j.pbb.2007.11.004
Brenes, J., Padilla, M., & Fornaguera, J. (2009). A detailed analysis of open-field habituation and behavioral and neurochemical antidepressant-like effects in postweaning enriched rats. Behavioural Brain Research, 197(1), 125–137. https://doi.org/10.1016/j.bbr.2008.08.014
Brenes Sáenz, J. C., Villagra, O. R., & Trías, J. T. (2006). Factor analysis of forced swimming test, sucrose preference test and open field test on enriched, social and isolated reared rats. Behavioural Brain Research, 169, 57–65. https://doi.org/10.1016/j.bbr.2005.12.001
Cacioppo, J. T., Cacioppo, S., Capitanio, J. P., & Cole, S. W. (2015). The neuroendocrinology of social isolation. Annual Review of Psychology, 66(1), 733–767. https://doi.org/10.1146/annurev-psych-010814-015240
Campeau, S., Nyhuis, T. J., Sasse, S. K., Kryakow, E. M., Herlhy, L., Masini, C. V., & Day, H. E. (2010). Hypothalamic pituitary adrenal axis responses to low-intensity stressors are reduced after voluntary wheel running in rats. Journal of Neuroendocrinology, 22(8), 872–888. https://doi.org/10.1111/j.1365-2826.2010.02007.x
Carnevali, L., Mastorci, M., Graiani, G., Razzoli, M., Trombini, M., Pico-Alfonso, M. A., Arban, R., Grippo, A. J., Quaini, F., & Sgoifo, A. (2012). Social defeat and isolation induce clear signs of a depression-like state, but modest cardiac alterations in wild-type rats. Physiology & Behavior, 106, 142–150. https://doi.org/10.1016/j.physbeh.2012.01.022
Carnevali, L., Statello, R., Vacondio, F., Ferlenghi, F., Spadoni, G., Rivara, S., Mor, M., & Sgoifo, A. (2020). Antidepressant-like effects of pharmacological inhibition of FAAH activity in socially isolated female rats. European Journal of Neuropsychopharmacology, 32, 77–87. https://doi.org/10.1016/j.euroneuro.2019.12.119
Carter, C. S., Devries, A. C., & Getz, L. L. (1995). Physiological substrates of mammalian monogamy: The prairie vole model. Neuroscience & Biobehavioral Reviews, 19(2), 303–314. https://doi.org/10.1016/0149-7634(94)00070-H
Casemiro, F. G., Rodrigues, I. A., Dias, J. C., de Sousa Alves, L. C., Inouye, K., & Gratão, A. C. M. (2016). Impact of cognitive stimulation on depression, anxiety, cognition and functional capacity among adults and elderly participants of an open university for senior citizens. Revista Brasileira de Geriatria e Gerontologia, 19(4), 683–694. https://doi.org/10.1590/1809-98232016019.150214
Clemenson, G. D., Deng, W., & Gage, F. H. (2015). Environmental enrichment and neurogenesis: From mice to humans. Current Opinion in Behavioral Sciences, 4, 56–62. https://doi.org/10.1016/j.cobeha.2015.02.005
Clemenson, G. D., Gage, F. H., & Stark, C. E. L. (2018). Environmental enrichment and neuronal plasticity. In M. V. Chao (Ed.), The Oxford Handbook of Developmental Neural Plasticity. Oxford University Press. https://doi.org/10.1093/oxfordhb/9780190635374.013.13
Clemenson, G. D., & Stark, C. E. L. (2015). Virtual Environmental enrichment through video games improves hippocampal-associated memory. Journal of Neuroscience, 35(49), 16116–16125. https://doi.org/10.1523/JNEUROSCI.2580-15.2015
Cohen, J. (1992). A power primer. Psychological Bulletin, 112(1), 155–159. https://doi.org/10.1037//0033-2909.112.1.155
Cohen-Mansfield, J., Hazan, H., Lerman, Y., & Shalom, V. (2016). Correlates and predictors of loneliness in older-adults: A review of quantitative results informed by qualitative insights. International Psychogeriatrics, 28(4), 557–576. https://doi.org/10.1017/S1041610215001532
Cryan, J. F., Valentino, R. J., & Lucki, I. (2005). Assessing substrates underlying the behavioral effect of antidepressants using the modified rat forced swimming test. Neuroscience & Biobehavioral Reviews, 29, 547–569. https://doi.org/10.1016/j.neubiorev.2005.03.008
De Jong, J. G., van der Vegt, B. J., Buwalda, B., & Koolhaas, J. M. (2005). Social environment determines the long-term effects of social defeat. Physiology & Behavior, 84(1), 87–95. https://doi.org/10.1016/j.physbeh.2004.10.013
Dong, B. E., Xue, Y., & Sakata, K. (2018). The effect of enriched environment across ages: a study of anhedonia and BDNF gene induction. Genes, Brain and Behavior, 17(8), e12485, 10/1111/gbb.12485
Donovan, M., Mackey, C. S., Platt, G. N., Rounds, J., Brown, A. N., Trickey, D. J., Liu, Y., Jones, K. M., & Wang, Z. (2020). Social isolation alters behavior, the gut-immune-brain axis, and neurochemical circuits in male and female prairie voles. Neurobiology of Stress, 13, 100278. https://doi.org/10.1016/j.ynstr.2020.100278
Driscoll, I., Howard, S. R., Stone, J. C., Monfils, M. H., Tomanek, B., Brooks, W. M., & Sutherland, R. J. (2006). The aging hippocampus: A multi-level analysis in the rat. Neuroscience, 139(4), 1173–1185. https://doi.org/10.1016/j.neuroscience.2006.01.040
Ehninger, D., Wang, L.-P., Klempin, F., Römer, B., Kettenmann, H., & Kempermann, G. (2011). Enriched environment and physical activity reduce microglia and influence the fate of NG2 cells in the amygdala of adult mice. Cell and Tissue Research, 345(1), 69–86. https://doi.org/10.1007/s00441-011-1200-z
Ennaceur, A., Michalikova, S., Vanrensburg, R., & Chazot, P. (2008). Detailed analysis of the behavior and memory performance of middle-aged male and female CD-1 mice in a 3D maze. Behavioural Brain Research, 187(2), 312–326. https://doi.org/10.1016/j.bbr.2007.09.025
Fjell, A. M., McEvoy, L., Holland, D., Dale, A. M., Walhovd, K. B., & for the Alzheimer’s Disease Neuroimaging Initiative. (2014). What is normal aging? Effects of aging, amyloid and Alzheimer’s disease on the cerebral cortex and the hippocampus. Progress in Neurobiology, 117, 20–40. https://doi.org/10.1016/pneurobio.2014.02.004
Flores-Gutiérrez, E., Cabrera-Muñoz, E. A., Vega-Rivera, N. M., Ortiz-López, L., & Ramírez-Rodríguez, G. B. (2018). Exposure to patterned auditory stimuli during acute stress prevents despair-like behavior in adult mice that were previously housed in an enriched environment in combination with auditory stimuli. Neural Plasticity, 2018, 1–14. https://doi.org/10.1155/2018/8205245
Forrest, M. P., Parnell, E., & Penzes, P. (2018). Dendritic structural plasticity and neuropsychiatric disease. Nature Reviews Neuroscience, 19, 215–234. https://doi.org/10.1038/nrn.2018.16
Förster, F., Pabst, A., Stein, J., Röhr, S., Löbner, M., Heser, K., Miebach, L., Stark, A., Hajek, A., Wiese, B., Maier, W., Angermeyer, M. C., Scherer, M., Wagner, M., König, H.-H., & Riedel-Heller, S. G. (2019). Are older men more vulnerable to depression than women after losing their spouse? Evidence from three German old-age cohorts (AgeDifference.de platform). Journal of Affective Disorders, 256, 650–657. https://doi.org/10.1016/j.jad.2019.06.047
Francia, N., Cirulli, F., Chiarotti, F., Antonelli, A., Aloe, L., & Alleva, E. (2006). Spatial memory deficits in middle-aged mice correlate with lower exploratory activity and a subordinate status: Role of hippocampal neurotrophins. European Journal of Neuroscience, 23(3), 711–728. https://doi.org/10.1111/j.1460-9568.2006.04585.x
Galani, R., Berthel, M.-C., Lazarus, C., Majchrzak, M., Barbelivien, A., Kelche, C., & Cassel, J.-C. (2007). The behavioral effects of enriched housing are not altered by serotonin depletion but enrichment alters hippocampal neurochemistry. Neurobiology of Learning and Memory, 88(1), 1–10. https://doi.org/10.1016/j.nlm.2007.03.009
Gibb, R., & Kolb, B. (1998). A method for vibratome sectioning of Golgi–Cox stained whole rat brain. Journal of Neuroscience Methods, 79(1), 1–4. https://doi.org/10.1016/s0165-0270(97)00163-5
Glaser, E. M., & Van der Loos, H. (1981). Analysis of thick brain sections by obverse—Reverse computer microscopy: Application of a new, high clarity Golgi—Nissl stain. Journal of Neuroscience Methods, 4(2), 117–125. https://doi.org/10.1016/0165-0270(81)90045-5
Grippo, A. J., Ihm, E., Wardwell, J., McNeal, N., Scotti, M.-A. L., Moenk, D. A., Chandler, D. L., LaRocca, M. A., & Preihs, K. (2014). The effects of environmental enrichment on depressive and anxiety-relevant behaviors in socially isolated prairie voles. Psychosomatic Medicine, 76(4), 277–284. https://doi.org/10.1097/PSY.0000000000000052
Grippo, A. J., McNeal, N., Normann, M. C., Colburn, W., Dagner, A., & Woodbury, M. (2021). Behavioral and neuroendocrine consequences of disrupting a long-term monogamous social bond in aging prairie voles. Stress, 24(3), 239–250. https://doi.org/10.1080/10253890.2020.1812058
Grippo, A. J., Moffitt, J. A., Sgoifo, A., Jepson, A. J., Bates, S. L., Chandler, D. L., McNeal, N., & Preihs, K. (2012). The integration of depressive behaviors and cardiac dysfunction during an operational measure of depression: Investigating the role of negative social experiences using an animal model. Psychosomatic Medicine, 74, 612–619. https://doi.org/10.1097/PSY.0b013e31825ca8e5
Grippo, A. J., Wu, K. D., Hassan, I., & Carter, C. S. (2008). Social isolation in prairie voles induces behaviors relevant to negative affect: Toward the development of a rodent model focused on co-occurring depression and anxiety. Depression and Anxiety, 25, E17–E26.
Gualtieri, F., Brégère, C., Laws, G. C., Armstrong, E. A., Wylie, N. J., Moxham, T. T., Guzman, R., Boswell, T., & Smulders, T. V. (2017). Effects of environmental enrichment on doublecortin and BDNF expression along the dorso-ventral axis of the dentate gyrus. Frontiers in Neuroscience, 11, 488. https://doi.org/10.3389/fnins.2017.00488
Gubert, C., & Hannan, A. J. (2019). Environmental enrichment as an experience-dependent modulator of social plasticity and cognition. Brain Research, 1717, 1–14. https://doi.org/10.1016/j.brainres.2019.03.033
Gum, A. M., McDougal, S. J., McIlvane, J. M., & Mingo, C. A. (2010). Older adults are less likely to identify depression without sadness. Journal of Applied Gerontology, 29(5), 603–621. https://doi.org/10.1177/0733464809343106
Harati, H., Barbelivien, A., Herbeaux, K., Muller, M.-A., Engeln, M., Kelche, C., Cassel, J.-C., & Majchrzak, M. (2013). Lifelong environmental enrichment in rats: Impact on emotional behavior, spatial memory vividness, and cholinergic neurons over the lifespan. Age, 35(4), 1027–1043. https://doi.org/10.1007/s11357-012-9424-8
He, S. B., Tang, W. G., Tang, W. J., Kao, X. L., Zhang, C. G., & Wong, X. T. (2012). Exercise intervention may prevent depression. International Journal of Sports Medicine, 7, 525–530. https://doi.org/10.1055/s-0032-1306325
Hercher, C., Canetti, L., Turecki, G., & Mechawar, N. (2010). Anterior cingulate pyramidal neurons display altered dendritic branching in depressed suicides. Journal of Psychiatric Research, 44(5), 286–293. https://doi.org/10.1016/j.jpsychires.2009.08.011
Herman, J. P., Ostrander, M. M., Mueller, N. K., & Figueiredo, H. (2005). Limbic system mechanisms of stress regulation: Hypothalamo-pituitary-adrenocortical axis. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 29, 1201–1213. https://doi.org/10.1016/j.pnpbp.2005.08.006
Hostinar, C. E., Sullivan, R. M., & Gunnar, M. R. (2014). Psychobiological mechanisms underlying the social buffering of the hypothalamic-pituitary-adrenocortical axis: A review of animal models and human studies across development. Psychological Bulletin, 140, 256–282. https://doi.org/10.1037/a0032671
Hüttenrauch, M., Salinas, G., & Wirths, O. (2016). Effects of long-term environmental enrichment on anxiety, memory, hippocampal plasticity and overall brain gene expression in C57BL6 mice. Frontiers in Molecular Neuroscience, 9. https://doi.org/10.3389/fnmol.2016.00062
Hylin, M. J., Watanasriyakul, W. T., Hite, N., McNeal, N., & Grippo, A. J. (2022). Morphological changes in the basolateral amygdala and behavioral disruptions associated with social isolation. Behavioural Brain Research, 416, 113572. https://doi.org/10.1016/j.bbr.2021.113572
Ieraci, A., Mallei, A., & Popoli, M. (2016). Social isolation stress induces anxious-depressive-like behavior and alterations of neuroplasticity-related genes in adult male mice. Neural Plasticity, 2016, 1–13. https://doi.org/10.1155/2016/6212983
Insel, T. R., & Young, L. J. (2001). The neurobiology of attachment. Nature Reviews Neuroscience, 2, 129–136. https://doi.org/10.1038/35053579
Jarcho, M. R., McNeal, N., Colburn, W., Normann, M. C., Watanasriyakul, W. T., & Grippo, A. J. (2019). Wheel access has opposing effects on stress physiology depending on social environment in female prairie voles (Microtus ochrogaster). Stress, 22(2), 265–275. https://doi.org/10.1080/10253890.2018.1553948
Kenkel, W. M., Gustison, M. L., & Beery, A. K. (2021). A neuroscientist’s guide to the vole. Current Protocols, 1, e175. https://doi.org/10.1002/cpz1.175
Kim, S.-E., Ko, I.-G., Kim, B.-K., Shin, M.-S., Cho, S., Kim, C.-J., Kim, S.-H., Baek, S.-S., Lee, E.-K., & Jee, Y.-S. (2010). Treadmill exercise prevents aging-induced failure of memory through an increase in neurogenesis and suppression of apoptosis in rat hippocampus. Experimental Gerontology, 45(5), 357–365. https://doi.org/10.1016/j.exger.2010.02.005
Kobilo, T., Liu, Q. R., Gandhi, K., Mughal, M., Shaham, Y., & van Praag, H. (2011). Running is the neurogenic and neurotrophic stimulus in environmental enrichment. Learning and Memory, 18, 605–609. https://doi.org/10.1101/lm.2283011
Koike, H., Ibi, D., Mizoguchi, H., Nagai, T., Nitta, A., Takuma, K., Nabeshima, T., Yoneda, Y., & Yamada, K. (2009). Behavioral abnormality and pharmacologic response in social isolation-reared mice. Behavioural Brain Research, 202(1), 114–121. https://doi.org/10.1016/j.bbr.2009.03.028
Kolb, B., Forgie, M., Gibb, R., Gorny, G., & Rowntree, S. (1998). Age, experience and the changing brain. Neuroscience & Biobehavioral Reviews, 22(2), 143–159. https://doi.org/10.1016/s0149-7634(97)00008-0
Kolb, B., Gibb, R., & Gomy, G. (2003). Experience-dependent changes in dendritic arbor and spine density in neocortex vary qualitatively with age and sex. Neurobiology of Learning and Memory, 79(1), 1–10. https://doi.org/10.1016/s1074-7427(02)00021-7
Li, L., & Tang, B. L. (2005). Environmental enrichment and neurodegenerative diseases. Biochemical and Biophysical Research Communications, 334(2), 293–297. https://doi.org/10.1016/j.bbrc.2005.05.162
Lieberwirth, C., Liu, Y., Jia, X., & Wang, Z. (2012). Social isolation impairs adult neurogenesis in the limbic system and alters behaviors in female prairie voles. Hormones and Behavior, 62(4), 357–366. https://doi.org/10.1016/j.yhbeh.2012.03.005
Luchetti, M., Lee, J. H., Aschwanden, D., Sesker, A., Strickhouser, J. E., Terracciano, A., & Sutin, A. R. (2020). The trajectory of loneliness in response to COVID-19. American Psychologist, 75(7), 897–908.
Malatynska, E., Steinbusch, H. W. M., Redkozubova, O., Bolkunov, A., Kubatiev, A., Yeritsyan, N. B., Vignisse, J., Bachurin, S., & Strekalova, T. (2012). Anhedonic-like traits and lack of affective deficits in 18-month-old C57BL/6 mice: Implications for modeling elderly depression. Experimental Gerontology, 47(8), 552–564. https://doi.org/10.1016/j.exger.2012.04.010
Malykhin, N. V., Bouchard, T. P., Camicioli, R., & Coupland, N. J. (2008). Aging hippocampus and amygdala. NeuroReport, 19(5), 543–547. https://doi.org/10.1097/WNR.0b013e3282f8b18c
McEwen, B. S. (2002). Sex, stress and the hippocampus: allostasis, allostatic load and the aging process. Neurobiology of Aging, 23(5), 921–939. https://doi.org/10.1016/s0197-4580(02)00027-1
McEwen, B. S. (2017). Neurobiological and systemic effects of chronic stress. Chronic Stress, 1, 1–11. https://doi.org/10.1177/2470547017692328
McKlveen, J. M., Myers, B., & Herman, J. P. (2015). The medial prefrontal cortex: Coordinator of autonomic, neuroendocrine and behavioral responses to stress. Journal of Neuroendocrinology, 27, 446–456. https://doi.org/10.1111/jne.12272
McNeal, N., Anderson, E. M., Moenk, D., Tranahas, D., Matuszewich, L., & Grippo, A. J. (2018). Social isolation alters central nervous system monoamine content in prairie voles following acute restraint. Social Neuroscience, 13(2), 173–183. https://doi.org/10.1080/17470919.2016.1276473
McQuaid, R. J., Audet, M. C., Jacobson-Pick, S., & Anisman, H. (2016). The differential impact of social defeat on mice living in isolation or groups in an enriched environment: Plasma corticosterone and monoamine variations. International Journal of Neuropsychopharmacology, 16, 351–363. https://doi.org/10.1017/S1461145712000120
Normann, M. C., Cox, M., Akinbo, O. I., Watanasriyakul, W. T., Kovalev, D., Ciosek, S., Miller, T., & Grippo, A. J. (2021). Differential paraventricular nucleus activation and behavioral responses to social isolation in prairie voles following environmental enrichment with and without physical exercise. Social Neuroscience, 16(4), 375–390. https://doi.org/10.1080/17470919.2021.1926320
Normann, M. C., McNeal, N., Dagner, A., Ihm, E., Woodbury, M., & Grippo, A. J. (2018). The influence of environmental enrichment on cardiovascular and behavioral responses to social stress. Psychosomatic Medicine, 80(3), 271–277. https://doi.org/10.1097/PSY.0000000000000558
Novaes, L. S., dos Santos, N. B., Batalhote, R. F. P., Malta, M. B., Camarini, R., Scavone, C., & Munhoz, C. D. (2017). Environmental enrichment protects against stress-induced anxiety: Role of glucocorticoid receptor, ERK, and CREB signaling in the basolateral amygdala. Neuropharmacology, 113, 457–466. https://doi.org/10.1016/j.neuropharm.2016.10.026
O’Súilleabháin, P. S., Gallagher, S., & Steptoe, A. (2019). Loneliness, living alone, and all-cause mortality: The role of emotional and social loneliness in the elderly during 19 years of follow-up. Psychosomatic Medicine, 81, 521–526. https://doi.org/10.1097/PSY.000000000000071
Palgi, Y., Shrira, A., Ring, L., Bodner, E., Avidor, S., Bergman, Y., Cohen-Fridel, S., Keisari, S., & Hoffman, Y. (2020). The loneliness pandemic: Loneliness and other concomitants of depression, anxiety and their comorbidity during the COVID-19 outbreak. Journal of Affective Disorders, 275, 109–111. https://doi.org/10.1016/j.jad.2020.06.036
Pan, Y., Liu, Y., Young, K. A., Zhang, Z., & Wang, Z. (2009). Post-weaning social isolation alters anxiety-related behavior and neurochemical gene expression in the brain of male prairie voles. Neuroscience Letters, 454(1), 67–71. https://doi.org/10.1016/j.neulet.2009.02.064
Panossian, A., Cave, M. W., Patel, B. A., Brooks, E. L., Flint, M. S., & Yeoman, M. S. (2020). Effects of age and social isolation on murine hippocampal biochemistry and behavior. Mechanisms of Ageing and Development, 191, 111337. https://doi.org/10.1016/j.mad.2020.111337
Park, C. L., Finkelstein-Fox, L., Russell, B. S., Fendrich, M., Hutchison, M., & Becker, J. (2021). Psychological resilience early in the COVID-19 pandemic: Stressors, resources, and coping strategies in a national sample of Americans. American Psychologist, 76(5), 715–728. https://doi.org/10.1037/amp000813
Park, J.-M., Kim, M.-W., & Shim, H.-Y. (2019). Effects of a multicomponent cognitive stimulation program on cognitive function improvement among elderly women. Asian Nursing Research, 13(5), 306–312. https://doi.org/10.1016/j.anr.2019.11.001
Paxinos, G., & Watson, C. (2006). The rat brain in stereotaxic coordinates (6th ed). Elsevier.
Pellow, S., Chopin, P., File, S. E., & Briley, M. (1985). Validation of open: Closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. Journal of Neuroscience Methods, 14, 149–167. https://doi.org/10.1016/0165-0270(85)90031-7
Peña, Y., Prunell, M., Rotllant, D., Armario, A., & Escorihuela, R. M. (2009). Enduring effects of environmental enrichment from weaning to adulthood on pituitary-adrenal function, pre-pulse inhibition and learning in male and female rats. Psychoneuroendocrinology, 34(9), 1390–1404. https://doi.org/10.1016/j.psyneuen.2009.04.019
Prenderville, J. A., Kennedy, P. J., Dinan, T. G., & Cryan, J. F. (2015). Adding fuel to the fire: The impact of stress on the ageing brain. Trends in Neurosciences, 38(1), 13–25. https://doi.org/10.1016/j.tins.2014.11.001
Prounis, G. S., Thomas, K., & Ophir, A. G. (2018). Developmental trajectories and influences of environmental complexity on oxytocin receptor and vasopressin 1a receptor expression in male and female prairie voles. Journal of Comparative Neurology, 526(11), 1820–1842. https://doi.org/10.1002/cne.24450
Qiao, H., Li, M.-X., Xu, C., Chen, H.-B., An, S.-C., & Ma, Z.-M. (2016). Dendritic spines in depression: What we learned from animal models. Neural Plasticity, 2016, 8056370. https://doi.org/10.1155/2016/8056370
Rapley, S. A., Prickett, T. C. R., Dalrymple-Alford, J. C., & Espiner, E. A. (2018). Environmental enrichment elicits a transient rise of bioactive C-type natriuretic peptide in young but not aged rats. Frontiers in Behavioral Neuroscience, 12, 142. https://doi.org/10.3389/fnbeh.2018.00142
Reguilón, M. D., Ferrer-Pérez, C., Ballestín, R., Miñarro, J., & Rodríguez-Arias, M. (2020). Voluntary wheel running protects against the increase in ethanol consumption induced by social stress in mice. Drug and Alcohol Dependence, 212, 108004. https://doi.org/10.1016/j.drugalcdep.2020.108004
Reinhart, C. J., McIntyre, D. C., Pellis, S. M., & Kolb, B. E. (2021). Prefrontal neuronal morphology in kindling-prone (FAST) and kindling-resistant (SLOW) rats. Synapse, 75(9), e22217. https://doi.org/10.1002/syn.22217
Rubinow, M. J., Drogos, L. L., & Juraska, J. M. (2009). Age-related dendritic hypertrophy and sexual dimorphism in rat basolateral amygdala. Neurobiology of Aging, 30(1), 137–146. https://doi.org/10.1016/j.neurobiolaging.2007.05.006
Segovia, G., Yagüe, A. G., García-Verdugo, J. M., & Mora, F. (2006). Environmental enrichment promotes neurogenesis and changes the extracellular concentrations of glutamate and GABA in the hippocampus of aged rats. Brain Research Bulletin, 70(1), 8–14. https://doi.org/10.1016/j.brainresbull.2005.11.005
Serra, M., Pisu, M. G., Floris, I., & Biggio, G. (2005). Social isolation-induced changes in the hypothalamic-pituitary-adrenal axis in the rat. Stress, 8(4), 259–264. https://doi.org/10.1080/10253890500495244
Seong, H.-H., Park, J.-M., & Kim, Y.-J. (2018). Antidepressive effects of environmental enrichment in chronic stress–induced depression in rats. Biological Research For Nursing, 20(1), 40–48. https://doi.org/10.1177/1099800417730400
Simpson, J., & Kelly, J. P. (2011). The impact of environmental enrichment in laboratory rats—Behavioural and neurochemical aspects. Behavioural Brain Research, 222(1), 246–264. https://doi.org/10.1016/j.bbr.2011.04.002
Singhal, G., Morgan, J., Jawahar, M. C., Corrigan, F., Jaehne, E. J., Toben, C., Breen, J., Pederson, S. M., Hannan, A. J., & Baune, B. T. (2019). Short-term environmental enrichment, and not physical exercise, alleviate cognitive decline and anxiety from middle age onwards without affecting hippocampal gene expression. Cognitive, Affective, & Behavioral Neuroscience, 19(5), 1143–1169. https://doi.org/10.3758/s13415-019-00743-x
Smith, B. L., Morano, R. L., Ulrich-Lai, Y. M., Myers, B., Solomon, M. B., & Herman, J. P. (2018). Adolescent environmental enrichment prevents behavioral and physiological sequelae of adolescent chronic stress in female (but not male) rats. Stress, 21(5), 464–473. https://doi.org/10.1080/10253890.2017.1402883
Sotoudeh, N., Namavar, M. R., Zarifkar, A., & Heidarzadegan, A. R. (2020). Age-dependent changes in the medial prefrontal cortex and medial amygdala structure, and elevated plus-maze performance in the healthy male Wistar rats. IBRO Reports, 9, 183–194. https://doi.org/10.1016/j.ibror.2020.08.002
Steptoe, A., Shankar, A., Demakakos, P., & Wardle, J. (2013). Social isolation, loneliness, and all-cause mortality in older men and women. Proceedings of the National Academy of Science, 110(15), 5797–5801. https://doi.org/10.1073/pnas.1219686110
Stowe, J. R., Liu, Y., Curtis, J. T., Freeman, M. E., & Wang, Z. (2005). Species differences in anxiety-related responses in male prairie and meadow voles: The effects of social isolation. Physiology & Behavior, 86, 369–378. https://doi.org/10.1016/j.physbeh.2005.08.007
Sun, P., Smith, A. S., Lei, K., Liu, Y., & Wang, Z. (2014). Breaking bonds in male prairie vole: Long-term effects on emotional and social behavior, physiology, and neurochemistry. Behavioural Brain Research, 265, 22–31. 10. 1016/j.bbr.2014.02.016
Sztainberg, Y., Kuperman, Y., Tsoory, M., Lebow, M., & Chen, A. (2010). The anxiolytic effect of environmental enrichment is mediated via amygdalar CRF receptor type 1. Molecular Psychiatry, 15(9), 905–917. https://doi.org/10.1038/mp.2009.151
Torres-Lista, V., & Giménez-Llort, L. (2015). Early postnatal handling and environmental enrichment improve the behavioral responses of 17-month-old 3xTg-AD and non-transgenic mice in the forced swim test in a gender-dependent manner. Behavioural Processes, 120, 120–127. https://doi.org/10.1016/j.beproc.2015.09.011
Tsai, S.-F., Huang, T.-Y., Chang, C.-Y., Hsu, Y.-C., Chen, S.-J., Yu, L., Kuo, Y.-M., & Jen, C. J. (2014). Social instability stress differentially affects amygdalar neuron adaptations and memory performance in adolescent and adult rats. Frontiers in Behavioral Neuroscience, 8, 27. https://doi.org/10.3389/fnbeh.2014.00027
van Praag, H., Kempermann, G., & Gage, F. H. (2000). Neural consequences of environmental enrichment. Nature Reviews Neuroscience, 1, 191–198. https://doi.org/10.1038/35044558
Volkert, J., Schulz, H., Härter, M., Wlodarczyk, O., & Andreas, S. (2013). The prevalence of mental disorders in older people in Western countries – A meta-analysis. Ageing Research Reviews, 12(1), 339–353. https://doi.org/10.1016/j.arr.2012.09.004
Watanasriyakul, W. T., Normann, M. C., Akinbo, O. I., Colburn, W., Dagner, A., & Grippo, A. J. (2019). Protective neuroendocrine effects of environmental enrichment and voluntary exercise against social isolation: Evidence for mediation by limbic structures. Stress, 22(5), 603–618. https://doi.org/10.1080/10253890.2019.1617691
Watanasriyakul, W. T., Wardwell, J., McNeal, N., Schultz, R., Woodbury, M., Dagner, A., Cox, M., & Grippo, A. J. (2018). Voluntary physical exercise protects against behavioral and endocrine reactivity to social and environmental stressors in the prairie vole. Social Neuroscience, 13(5), 602–615. https://doi.org/10.1080/17470919.2017.1365761
Woo, C. C., & Leon, M. (2013). Environmental enrichment as an effective treatment for autism: A randomized controlled trial. Behavioral Neuroscience, 127(4), 487–497. https://doi.org/10.1037/a0033010
Wuthrich, V. M., Johnco, C. J., & Wetherell, J. L. (2015). Differences in anxiety and depression symptoms: Comparison between older and younger clinical samples. International Psychogeriatrics, 27(9), 1523–1532. https://doi.org/10.1017/S1041610215000526
Yagi, S., & Galea, L. A. M. (2019). Sex differences in hippocampal cognition and neurogenesis. Neuropsychopharmacology Reviews, 44, 200–213. https://doi.org/10.1038/s41386-018-0208-4
Yagi, S., Splinter, J. E. J., Tai, D., Wong, S., Wen, Y., & Galea, L. A. M. (2020). Sex differences in maturation and attrition of adult neurogenesis in the hippocampus. eNeuro, 7(4), ENEURO.0468-19-2020 1-14., 7. https://doi.org/10.1523/ENEURO.0468-19.2020
Young, K. A., Gobrogge, K. L., Liu, Y., & Wang, Z. (2011). The neurobiology of pair bonding: Insights from a socially monogamous rodent. Frontiers in Neuroendocrinology, 32, 53-69. 10.1016.j.yfrne.2010.07.006
Xia, N., & Li, H. (2018). Loneliness, social isolation, and cardiovascular health. Antioxidants & Redox Signaling, 28(9), 837–851. https://doi.org/10.1089/ars.2017.7312
Zanier-Gomes, P. H., de Abreu Silva, T. E., Zanetti, G. C., Benati, É. R., Pinheiro, N. M., Murta, B. M. T., & Crema, V. O. (2015). Depressive behavior induced by social isolation of predisposed female rats. Physiology & Behavior, 151, 292–297. https://doi.org/10.1016/j.physbeh.2015.07.026
Zhong, F., Liu, L., Wei, J.-L., & Dai, R.-P. (2019). Step by step Golgi-Cox staining for cryosection. Frontiers in Neuroanatomy, 13, 62. https://doi.org/10.3389/fnana.2019.00062
Acknowledgements
The authors would like to thank the following individuals for valuable assistance: Sarah Ciosek, William Colburn, Miranda Cox, Nicole Holzapfel, Blessy Johnson, Meredith McCormick, Marigny Normann, Cassidy Padal, Samantha Sujet, and Cynthia Sanchez-Vazquez. Thank you to the Northern Illinois University College of Liberal Arts and Sciences Instrumentation Shop for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding Information
This research was funded in part by National Institutes of Health grants HL112350 and HL147179. The funder had no role in the study design, data collection, data analysis and interpretation, writing of the report, or decision to submit the article for publication.
Conflicts of Interest
The authors declare no competing interests.
Data Availability
Data for the project can be obtained here: https://osf.io/d29zg/.
Author Contributions
All authors contributed significantly to the study conceptualization, study design, data collection, analyses, interpretation, and/or writing of the manuscript. All authors have approved the final version of the submitted manuscript.
Ethical Approval
All procedures described in this manuscript were approved by the Northern Illinois University Institutional Animal Care and Use Committee, and are in compliance with all federal guidelines. The methods conform to the Guide for the Care and Use of Laboratory Animals from the Institute of Laboratory Animal Research. All efforts were made to ensure that procedures were conducted in a responsible and ethical manner in the context of the Three Rs (replacement, reduction, and refinement).
Informed Consent
Not applicable.
Additional information
Handling Editor: Forrest Dylan Rogers
Open Access
(1) Data and/or materials are not available per the Level 2 TOP guidelines.
(2) These experiments were not preregistered.
Rights and permissions
About this article
Cite this article
Akinbo, O.I., McNeal, N., Hylin, M. et al. The Influence of Environmental Enrichment on Affective and Neural Consequences of Social Isolation Across Development. Affec Sci 3, 713–733 (2022). https://doi.org/10.1007/s42761-022-00131-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42761-022-00131-8