Abstract
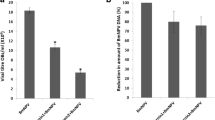
Bombyx mori nucleopolyhedrovirus (BmNPV) is a pathogen causing Bombyx mori larval mortality and is also called grasserie disease (Lepidoptera: Bombycidae). Since this viral disease is not fully under control in sericulture, it is necessary to identify agents that can prevent or reduce the infection/proliferation of BmNPV in B. mori culture boxes. Sericultural activity is similar an agriculture, and the loss of cocoon production leads to economic losses. In this research area, we previously reported that the molecule Bm5 inhibits cathepsin activity from BmNPV (V-CATH) and protects infected B. mori larvae from liquefaction processes, limiting the spread of the disease. Here, we evaluated the efficacy of Bm5 in reducing sericultural loss due to BmNPV infection. Assays were carried out with B. mori fifth-instar hybrids immediately inoculated with BmNPV and Bm5 to prevent viral cathepsin action. The biological and productive variables relevant during larval development and cocoon productivity were analysed to verify the effectiveness of Bm5 treatment. Biological and economic parameter data showed that the larval survival rate of the BmNPV-infected and Bm5-treated groups was 35.6% higher than that of the untreated BmNPV group. In addition, the infected BmNPV and Bm5-treated groups showed surviving pupae that produced good-quality cocoons. The α- and β-esterase analysis from the midgut and fat body did not show a change in isoenzyme expression between Bm5-treated and untreated larvae infected with BmNPV. Therefore, the inhibition of viral cathepsin with the Bm5 was shown to be effective in reducing the loss of silk production from BmNPV-infected B. mori larvae.




Similar content being viewed by others
References
Aramwit P, Siritientong T, Srichana T (2012) Potential applications of silk sericin, a natural protein from textile industry by-products. Waste Manag Res 30:217–224. https://doi.org/10.1177/0734242X11404733
Ardisson-Araújo DMP, Melo FL, de Souza AM et al (2014) Complete genome sequence of the first non-Asian isolate of Bombyx mori nucleopolyhedrovirus. Virus Genes 49:477–484. https://doi.org/10.1007/s11262-014-1112-6
Ashraf H, Qamar A (2023) Silkworm Bombyx mori as a model organism: a review. Physiol Entomol 48:107–121. https://doi.org/10.1111/phen.12421
Bao Y-Y, Lv Z-Y, Liu Z-B et al (2010) Comparative analysis of Bombyx mori nucleopolyhedrovirus responsive genes in fat body and haemocyte of B. mori resistant and susceptible strains. Insect Mol Biol 19:347–358. https://doi.org/10.1111/j.1365-2583.2010.00993.x
Blissard GW, Theilmann DA (2018) Baculovirus entry and egress from insect cells. Annu Rev Virol 5:113–139. https://doi.org/10.1146/annurev-virology-092917-043356
Brancalhão RMC, Torquato EFB, Fernandez MA (2009) Cytopathology of Bombyx mori (Lepidoptera: Bombycidae) silk gland caused by multiple nucleopolyhedrovirus. Genet Mol Res 8:162–172. https://doi.org/10.4238/vol8-1gmr545
Brancalhão RMC, Ribeiro LFC, Torquato EFB et al (2013) Avaliação antiviral do formulado de sódio dicloroisocianurato contra o nucleopoliedrovírus do bicho-da-seda. Arq Inst Biol (sao Paulo) 80:233–235. https://doi.org/10.1590/s1808-16572013000200014
Bueno PSA, Biavatti DC, Chiarello ASG et al (2019) The structure of viral cathepsin from Bombyx mori Nuclear Polyhedrosis Virus as a target against grasserie: docking and molecular dynamics simulations. J Biomol Struct Dyn 37:3607–3615. https://doi.org/10.1080/07391102.2018.1521344
Fujimoto S, Kawamoto M, Shoji K (2020) Whole-genome sequencing and comparative transcriptome analysis of Bombyx mori nucleopolyhedrovirus La strain. Virus Genes 56:249–259. https://doi.org/10.1007/s11262-019-01727-2
Gigliolli AAS, Lapenta AS, Ruvolo-Takasusuki MCC et al (2015) Morpho-functional characterization and esterase patterns of the midgut of Tribolium castaneum Herbst, 1797 (Coleoptera: Tenebrionidae) parasitized by Gregarina cuneata (Apicomplexa: Eugregarinidae). Micron 76:68–78. https://doi.org/10.1016/j.micron.2015.04.008
Hodgson JJ, Arif BM, Krell PJ (2011) Interaction of Autographa californica Multiple Nucleopolyhedrovirus Cathepsin Protease Progenitor (proV-CATH) with Insect Baculovirus Chitinase as a Mechanism for proV-CATH Cellular Retention. J Virol 85:3918–3929. https://doi.org/10.1128/jvi.02165-10
Hodgson JJ, Krell PJ, Passarelli AL (2021) Mature viral cathepsin is required for release of viral occlusion bodies from Autographa californica multiple nucleopolyhedrovirus-infected cells. Virology 556:23–32. https://doi.org/10.1016/j.virol.2021.01.001
Hua X, Zhang Q, Xu W et al (2021) The Antiviral Molecule 5-Pyridoxolactone Identified Post BmNPV Infection of the Silkworm. Bombyx Mori Int J Mol Sci 22:7423. https://doi.org/10.3390/ijms22147423
Karthik T, Rathinamoorthy R (2017) Sustainable silk production in Sustainable Fibres and Textiles. The Textile Institute Book Series. Elsevier Ltd, pp 135–170. https://doi.org/10.1016/B978-0-08-102041-8.00006-8
Katsuma S, Nakanishi T, Shimada T (2008) Bombyx mori nucleopolyhedrovirus FP25K is essential for maintaining a steady-state level of v-cath expression throughout the infection. Virus 140:155–160. https://doi.org/10.1016/j.virusres.2008.11.014
Koh LD, Cheng Y, Teng CP et al (2015) Structures, mechanical properties and applications of silk fibroin materials. Prog Polym Sci 46:86–110. https://doi.org/10.1016/j.progpolymsci.2015.02.001
Kunz RI, Capelassi AN, Alegre-Maller ACP et al (2020) Sericin as treatment of obesity: morphophysiological effects in obese mice fed with high-fat diet. Einstein (São Paulo) 18:eAO4876. https://doi.org/10.31744/einstein_journal/2020AO4876
Latha S, Bhaskar RN, Pallavi C (2011) Effect of botanical treatment to BmNPV polyhedral bodies on cocoon parameters of silkworm B. mori (PM x CSR2). Int J Adv Biol Res 1:45–51
Meng X, Zhu F, Chen K (2017) Silkworm : A Promising Model Organism in Life Science. J Insect Sci 17:1–6. https://doi.org/10.1093/jisesa/iex064
Monesi N, Fernandez MA, Fontes AM et al (1995) Molecular characterization of an 18 kb segment of DNA puff C4 of Bradysia hygida (Diptera, Sciaridae). Chromosoma 103:715–724. https://doi.org/10.1007/BF00344233
Montella IR, Schama R, Valle D (2012) The classification of esterases: an important gene family involved in insecticide resistance - A Review. Memórias Do Instituto Osealdo Cruz 107:437–449. https://doi.org/10.1590/S0074-02762012000400001
Oro AL, Costa RM, Brita EA et al (2021) Cytopathology of Bombyx mori (Lepidoptera: Bombycidae) larva integument infected by Bombyx mori nucleopolyhedrovirus. Genet Mol Biol 20:gmr18758. https://doi.org/10.4238/gmr18758
Pereira EP, Conte H, Ribeiro LFC et al (2008) Cytopathological process by multiple nucleopolyhedrovirus in the testis of Bombyx mori L., 1758 (Lepidoptera: Bombycidae). J Invertebr Pathol 99:1–7. https://doi.org/10.1016/j.jip.2008.04.011
Porto AJ, Okamoto F, Ikuno AA et al (2005) Avaliação biológica e produtiva do bicho da seda (Bombyx mori L.) alimentado com folhas de amoreira pulvrizadas com extrato de Mirabilis jalapa. Arq Inst Biol (sao Paulo) 72:445–453. https://doi.org/10.1590/1808-1657v72p4452005
Ribeiro LFC, Brancalhão RMC, Torquato EFB, Fernandez MA (2009) Susceptibility of the Bombyx mori cardia cells to Nucleopolyhedrovirus, multiple subgroup, BmMNPV. J Invertebr Pathol 100:195–198. https://doi.org/10.1016/j.jip.2009.01.001
Rohrmann GF (2019) Baculovirus Molecular Biology, 4th edn. National Center for Biotechnology Information (US), Betherda, Bethesda. ID:NBK543458P
Ruiz JB, Ronqui L, de Oliveira JS et al (2013) Comparative electrophoretic profile of proteins and esterases in healthy silkworm larvae (Bombyx mori Lineu, 1758) and infected with nucleopolyhedrovirus. Biol Sci 35:77–82. https://doi.org/10.4025/actascibiolsci.v35i1.8612
Sabbag OJ, Nicodemo D, Oliveira JEM (2013) Custos e viabilidade econômica da produção de casulos do bicho-da-seda. Pesqui Agropecu Trop 43:187–194. https://doi.org/10.1590/S1983-40632013000200004
Santorum M, Beu CCL, Ribeiro LFC et al (2017) Ampullae infection of the malpighian tubules of Bombyx mori by Alphabaculovirus. Biosci J 33:703–712. https://doi.org/10.14393/BJ-v33n3-36800
Santos BH, Lorini I, Lazzari SMN (2006) Efeito do sinergista butóxido de piperonila na resistência de Oryzaephilus surinamensis (L.) (Coleoptera, Silvanidae) a deltametrina e fenitrotiom. Rev Bras Entomol 50:110–114. https://doi.org/10.1590/S0085-56262006000100016
Santos SA, Silva JL, Balani VA et al (2010) Conserved baculoviral ORFs 10 and 14 from Bombyx mori multiple nucleopolyhedrovirus. Genet Mol Res 9:457–470. https://doi.org/10.4238/vol9-1gmr723
Senem JV, Torquato EFB, Ribeiro LFC et al (2016) Cytopathology of the trachea of Bombyx mori (Lepidoptera: Bombycidae) to Bombyx mori nucleopolyhedrovirus. Micron 80:39–44. https://doi.org/10.1016/j.micron.2015.09.005
Sharma K, Kapoor B (2020) Sericulture as a Profit-Based Industry-A Review. Indian J Pure Appl Biosci 8:550–562. https://doi.org/10.18782/2582-2845.8210
Shen R, Lü D, Chen G et al (2022) SUMOylation Regulates BmNPV Replication by Moderating PKIP Intracellular Localization. Processes 10:261. https://doi.org/10.3390/pr10020261
Shobahah J, Xue S, Hu D et al (2017) Quantitative phosphoproteosome on the silkworm (Bombyx mori) cells infected with baculovirud. Virol J 14:117. https://doi.org/10.1186/s12985-017-0783-8
Su Z, Zhao C, Huang X et al (2023) Bombyx mori Ecdysone Receptor B1 May Inhibit BmNPV Infection by Triggering Apoptosis. InSects 14:505. https://doi.org/10.3390/insects14060505
Suman D, Ajit KS, Atul K (2021) Studies on synergistic effects of different chemicals on nuclear polyhedrosis (grasserie disease) of silkworm, Bombyx mori Linnaeus. Afr J Biol Sci 3:104–113. https://doi.org/10.33472/AFJBS.3.4.2021.104-113
Thiem SM, Cheng X-W (2009) Baculovirus Host-Range Virol Sin 24:436–457. https://doi.org/10.1007/s12250-009-3058-8
Torquato EFB, Miranda Neto MH, Brancalhão RMC (2006) Nucleopolyhedrovirus infected central nervous system cells of Bombyx mori (L.) (Lepidoptera: Bombycidae). Neotrop Entomol 35:70–74. https://doi.org/10.1590/S1519-566X2006000100010
Toufeeq S, Wang J, Shang-Zhi Z et al (2019) Bmserpin2 Is Involved in BmNPV Infection by Suppressing Melanization in Bombyx mori. InSects 10:399. https://doi.org/10.3390/insects10110399
Vessaro-Silva SA, Miranda Neto MH, Brancalhão RMC et al (2019) Antioxidant Systems as a Response to Midgut Cellular of Bombyx mori Lineu, 1758 (Lepidoptera: Bombycidae) Infection for Baculoviruses. J Econ Entomol 112:1089–1097. https://doi.org/10.1093/jee/toz009
Wang F, Zhang C-X, Kumar VS et al (2005) Influences of chitinase gene deletion from BmNPV on the cell lysis and host liquefaction. Arch Virol 150:981–990. https://doi.org/10.1007/s00705-004-0458-9
Wang X-y, Shao Z-m, Chen Q-y et al (2019) Knockdown of BmTCP-1β Delays BmNPV Infection in vitro. Front Microbiol 10:578.https://doi.org/10.3389/fmicb.2019.00578
Xu Y, Xiaoqian Z, Yujia L et al (2021) Transgenic genome editing-derived antiviral therapy to nucleopolyhedrovirus infection in the industrial strain of the silkworm. Insect Biochem Mol Biol 139:103672. https://doi.org/10.1016/j.ibmb.2021.103672
Zhou W, Zeng C, Liu R et al (2016) Antiviral activity and specific modes of action of bacterial prodigiosin against Bombyx mori nucleopolyhedrovirus in vitro. Appl Microbiol Biotechnol 100:3979–3988. https://doi.org/10.1007/s00253-015-7242-5
Acknowledgements
The authors thank COMCAP/UEM and UNIOESTE - Cascavel/Paraná – Brazil for the equipment facility and BRATAC S.A. for the cooperation and supply of commercial silkworm hybrids.
Funding
This work was supported by Fundação Araucária (40/16), Brazil; Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, CAPES (code 001), Brazil; Conselho Nacional de Desenvolvimento Científico e Tecnológico, CNPq (311077/2018-8; 409985/2018-0), Brazil, and the European Union (Proyecto SEDA, LA/2016/378-553). The contents of this publication are the sole responsibility of Proyecto Seda and do not reflect the views of the European Union.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
De La Cruz Cisneros, J.J., Cabral, L.C., Caligari, D. et al. A novel molecule reduces cocoon production loss in BmNPV-infected Bombyx mori larvae. Int J Trop Insect Sci (2024). https://doi.org/10.1007/s42690-024-01216-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42690-024-01216-6