Abstract
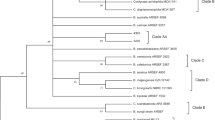
Molecular phylogenetic species recognition is essential in precisely differentiating the morphologically cryptic species of Metarhizium and Beauveria. These fungal entomopathogens can be an alternative to conventional synthetic insecticides and are emerging as effective biocontrol agents against sucking insect pests. In the present study, we identified Metarhizium and Beauveria species in diverse agricultural and semi-natural ecosystems of south Karnataka State of India using phylogenetic markers and evaluated their pathogenicity against Scirtothrips dorsalis. Based on morphological characters, 34 Metarhizium and 15 Beauveria isolates were identified in agricultural and semi-natural ecosystems. Phylogeny of Metarhizium was resolved using markers, TEF1-α, MzIGS3, and ITS rDNA to two species, M. pingshaense and M. guizhouense and Beauveria was resolved using markers, TEF1-α, Bloc, and ITS rDNA to a single species, B. bassiana based on phylogenetic optimality criteria. Isolates of M. pingshaense and B. bassiana resolved three phylogenetic clades each and M. guizhouense resolved one phylogenetic clade. Phylogenetic clades of M. pingshaense and B. bassiana exhibit intraclade variation and are not associated with crop plant or host insect. Laboratory bio-efficacy studies of 49 isolates of Metarhizium and Beauveria against adult S. dorsalis showed that pathogenicity varied significantly among isolates. Greenhouse bioassays revealed the high virulence of B. bassiana isolates over M. pingshaense isolates against S. dorsalis. Low LC50 value of 2.8 × 104 conidia/ml was obtained for B. bassiana GKVK 01_06 isolate against S. dorsalis. Our results indicate that indigenous B. bassiana isolates have strong potential in the management of S. dorsalis.


Similar content being viewed by others
References
Agrawal Y, Mual P, Shenoy BD (2014) Multi-gene genealogies reveal cryptic species Beauveria rudraprayagi sp. nov. from India. Mycosphere 5:719–736. https://doi.org/10.5943/mycosphere/5/6/3
Arthurs SP, Aristizabal LF, Avery PB (2013) Evaluation of entomopathogenic fungi against chilli thrips. Scirtothrips dorsalis. J Insect Sci 13:31. https://doi.org/10.1673/031.013.3101
Bischoff JF, Rehner SA, Humber RA (2009) A multilocus phylogeny of the Metarhizium anisopliae lineage. Mycologia 101:512–530. https://doi.org/10.3852/07-202
Brunner-Mendoza C, Reyes-Montes MR, Moonjely S, Bidochka MJ (2019) A review on the genus Metarhizium as an entomopathogenic microbial biocontrol agent with emphasis on its use and utility in Mexico. Biocontrol Sci Technol 29:83–102. https://doi.org/10.1080/09583157.2018.1531111
Bustamante DE, Oliva M, Leiva S, Mendoza JE, Bobadilla L et al (2019) Phylogeny and species delimitations in the entomopathogenic genus Beauveria (Hypocreales, Ascomycota), including the description of B. peruviensis sp. nov. MycoKeys 58:47–68. https://doi.org/10.3897/mycokeys.58.35764
Campbell LR, Robb KL, Ullman DE (2005) The complete tospovirus resource guide. Kansas State University. http://www.oznet.ksu.edu/tospovirus/tospo_list.htm
Charnley AK (2003) Fungal pathogens of insects: cuticle degrading enzymes and toxins. Adv Bot Res 40:241–321. https://doi.org/10.1016/S0065-2296(05)40006-3
Cullings KW (1992) Design and testing of a plant-specific PCR primer for ecological and evolutionary studies. Mol Ecol 1:233–240. https://doi.org/10.1111/j.1365-294X.1992.tb00182.x
Fisher JJ, Rehner SA, Bruck DJ (2011) Diversity of rhizosphere associated entomopathogenic fungi of perennial herbs, shrubs and coniferous trees. J Invertebr Pathol 106:289–295. https://doi.org/10.1016/j.jip.2010.11.001
Hemalatha S, Ramaraju K, Jeyarani S (2017) Evaluation of Entomopathogenic Fungi and Delivery Methods for Management of Thrips in Chillies. Int J Veg Sci 23:246–259. https://doi.org/10.1080/19315260.2016.1246502
Horticulture Statistics (2020) Department of agriculture, cooperation and farmers welfare. Ministery of agriculture and farmers welfare. Government of India, New Delhi, pp 1–3.
Humber RA (2012) Identification of entomopathogenic fungi. In: Lacey LA (ed) Manual of techniques in insect pathology, 2nd edn. San Diego, CA: Academic Press. pp 151–187. https://doi.org/10.1016/B978-0-12-386899-2.00006-3
Imoulan A, Hussain M, Kirk PM, Meziane AE, Yao YJ (2017) Entomopathogenic fungus Beauveria: Host specificity, ecology and significance of morpho-molecular characterization in accurate taxonomic classification. J Asia Pac Entomol 20:1204–1212. https://doi.org/10.1016/j.aspen.2017.08.015
Jacobsen CS, Hjelmsø MH (2014) Agricultural soils, pesticides, and microbial diversity. Curr Opin Biotech 27:15–20. https://doi.org/10.1016/j.copbio.2013.09.003
Jaronski ST (2010) Ecological factors in the inundative use of fungal entomopathogens. BioControl 55:159–185. https://doi.org/10.1007/s10526-009-9248-3
Kepler RM, Rehner SA (2013) Genome-assisted development of nuclear intergenic sequence markers for entomopathogenic fungi of the Metarhizium anisopliae species complex. Mol Ecol Resour 13:210–217. https://doi.org/10.1111/1755-0998.12058
Kepler RM, Ugine TA, Maul JE, Cavigelli MA (2015) Community composition and population genetics of insect pathogenic fungi in the genus Metarhizium from soils of a long-term agricultural research system. Environ Microbiol 17:2791–2804. https://doi.org/10.1111/1462-2920.12778
Kryukova V, Yaroslavtsevaa O, Tyurina M, Akhanaeva Y (2017) Ecological preferences of Metarhizium spp. from Russia and neighboring territories and their activity against Colorado potato beetle larvae. J Invertebr Pathol 149:1–7. https://doi.org/10.1016/j.jip.2017.07.001
LeOra Software (2002) Polo Plus – a user’s guide to Probit and Logit analysis. LeOra Software, Berkeley, CA
Litwin A, Nowak M, Rozalska S (2020) Entomopathogenic fungi: unconventional applications. Rev Environ Sci Biotechnol 19:23–42. https://doi.org/10.1007/s11157-020-09525-1
Lopes RB, Mesquita ALM, Tigano MS, Souza DA, Martins I, Faria M (2013) Diversity of indigenous Beauveria and Metarhizium spp. in a commercial banana field and their virulence toward Cosmopolites sordidus (Coleoptera: Curculionidae). Fungal Ecol 6:356–364. https://doi.org/10.1016/j.funeco.2013.06.007
Meyling NV, Lübeck M, Buckley EP, Eilenberg J (2009) Community composition, host range and genetic structure of the fungal entomopathogen Beauveria in adjoining agricultural and semi-natural habitats. Mol Ecol 18:1282–1293. https://doi.org/10.1111/j.1365-294X.2009.04095.x
Meyling NV, Pilz C, Keller S, Widmer F, Enkerli J (2012) Diversity of Beauveria spp. isolates from pollen beetles Meligethes aeneus in Switzerland. J Invertebr Pathol 109:76–82. https://doi.org/10.1016/j.jip.2011.10.001
Minitab 19 Statistical Software (2019) State College, PA: Minitab, Inc.
Nishi O, Sato H (2019) Isolation of Metarhizium spp. from rhizosphere soils of wild plants reflects fungal diversity in soil but not plant specificity. Mycology 58:1–11. https://doi.org/10.1080/21501203.2018.1524799
Patel BH, Koshiya DJ, Korat DM (2009) Population dynamics of chilli thrips, Scirtothrips dorsalis Hood in relation to weather parameters. Karnataka J Agric Sci 22:108–110
Rehner SA, Kepler RM (2017) Species limits, phylogeography and reproductive mode in the Metarhizium anisopliae complex. J Invertebr Pathol 48:60–66. https://doi.org/10.1016/j.jip.2017.05.008
Rehner SA, Minnis AM, Sung GH, Luangsa-ard JJ (2011) Phylogeny and systematics of the anamorphic, entomopathogenic genus Beauveria. Mycologia 103:1055–107. https://doi.org/10.3852/10-302
Rehner SA, Posada F, Buckley EP, Infante F (2006) Phylogenetic origins of African and Neotropical Beauveria bassiana pathogens of the coffee berry borer, Hypothenemus hampei. J Invertebr Pathol 93:11–21. https://doi.org/10.1016/j.jip.2006.04.005
Rezende JM, Botelho AB, Lopes MS, Delalibera I, Rehner SA (2015) Phylogenetic diversity of Brazilian Metarhizium associated with sugarcane agriculture. BioControl 60:495–505. https://doi.org/10.1007/s10526-015-9656-5
Seal DR, Kumar V (2010) Biological response of chilli thrips, Scirtothrips dorsalis Hood (Thysanoptera: Thripidae), to various regimes of chemical and biorational insecticides. Crop Prot 29:1241–1247. https://doi.org/10.1016/j.cropro.2010.07.011
Silvestro D, Michalak I (2012) raxmlGUI: a graphical front-end for RAxML. Org Diver Evol 12:335–337. https://doi.org/10.1007/s13127-011-0056-0
Sridevi KM, Jeyarani S, Ramaraju K (2018) Evaluation of oil-based formulation of Beauveria bassiana (Bb 112) (Bals.) Vuill. and delivery methods for the management of chilli thrips, Scirtothrips dorsalis Hood. J Biol Control 32:62–67. https://doi.org/10.18311/jbc/2018/16438
Steinwender BM, Enkerli J, Widmer F, Eilenberg J, Kristensen HL, Bidochka MJ, Meyling NV (2015) Root isolations of Metarhizium spp. from crops reflect diversity in the soil and indicate no plant specificity. J Invertebr Pathol 132:142–148. https://doi.org/10.1016/j.jip.2015.09.007
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA 5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739. https://doi.org/10.1093/molbev/msr121
Valero-Jiménez CA, Wiegers H, Zwaan BJ, Koenraadt CJ, van Kan JA (2016) Genes involved in virulence of the entomopathogenic fungus Beauveria bassiana. J Invertebr Pathol 133:41–49. https://doi.org/10.1016/j.jip.2015.11.011
Velavan V, Sasidharan TO, Patel A, Sundararaj R, Dharmarajan P et al (2016) Some observations on the diversity of Metarhizium fungi from forest soils of south India using molecular methods. National conference on frontiers in eco-biological sciences and its applications. p 72
Venette RC, Davis EE (2004) Chilli thrips/ yellow thrips, Scirtothrips dorsalis Hood (Thysanoptera: Thripidae) mini risk assessment. University of Minnesota, St. Paul, MN, p 31
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR Protocols: A Guide to Methods and Applications. Academic Press, San Diego, CA, pp 315–322
Zhang Z, Lu Y, Xu W, Sui L, Du Q et al (2020) Influence of genetic diversity of seventeen Beauveria bassiana isolates from different hosts on virulence by comparative genomics. BMC Genomics 21:451. https://doi.org/10.1186/s12864-020-06791-9
Zimmermann G (1986) The ‘Galleria bait method’ for detection of entomopathogenic fungi in soil. J Appl Entomol 102:213–215. https://doi.org/10.1111/j.1439-0418.1986.tb00912.x
Acknowledgements
JRF thank Start-up grant for Young Scientists’ Scheme (Grant number: SR/FT/LS-156/2010) of Science and Engineering Research Board (SERB), Govt. of India for providing grant and late Professor C. K. Suresh for his ideas.
Author information
Authors and Affiliations
Contributions
JRF designed and performed experiments, analyzed data, and wrote the manuscript. HKM revised the manuscript and both authors approved the final manuscript.
Corresponding author
Ethics declarations
Research involving human participants and/or animals
This article does not contain any studies with human participants or animals (vertebrates) performed by any of the authors. All applicable international, national, and institutional guidelines for the care and use of animals were followed.
Conflict of interest
The authors have declared no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Francis, J.R., Manchegowda, H.K. Molecular phylogenetic identification of Metarhizium and Beauveria and their bio-efficacy against chilli thrips, Scirtothrips dorsalis. Int J Trop Insect Sci 43, 909–918 (2023). https://doi.org/10.1007/s42690-023-01002-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-023-01002-w