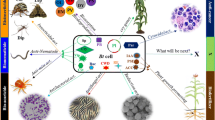
Abstract
Mosquitoes vector many infectious diseases and pose a serious threat to public health in developing countries. A wide array of synthetic insecticides is employed against these vectors. As the mosquitoes have got resistance to most of these chemical insecticides, there is a dire need to explore alternative control strategies such as entomopathogens. This laboratory study was aimed at isolating, purifying and evaluating the insecticidal proteins from indigenous strains of soil-inhabiting pathogenic bacteria against the larvae of southern house mosquito (Culex quinquefasciatus) and to identify the larval midgut receptor of bacterial toxin. One hundred bacterial isolates were isolated and screened. Only two isolates (AA-11 and AA-19) exhibited considerable larval mortality (> 80%) and were further identified by sequencing and blasting their 16 S rDNA gene fragments. Isolate AA-11 was identified as Pseudomonas sp. with 96% similarity, while AA-19 as Lysinibacillus sphaericus with 98% similarity. Microscopic analysis and gram staining of AA-19 confirmed its rod-shape structure and gram positive nature, respectively. It is likely that the bacterial isolate AA-19 was L. sphaericus. Furthermore, protein purification of this bacterial isolate revealed two proteins of approximately 51 and 43 kDa which reduced to 43 and 39 kDa respectively after digestion with trypsin indicating that these were Bin B and Bin A toxin proteins respectively of L. sphaericus. Results of toxicity bioassay revealed that the purified and activated toxin was highly toxic to C. quinquefasciatus larvae with a LC50 value of 191 ppb. Pull-down assays demonstrated that one protein of 66.5 kDa was pulled down by CNBr-beads conjugated with insecticidal proteins (Bin B and A). The pulled down protein was considered as C. quinquefasciatus maltase 1 (Cqm1), a receptor of Bin toxin in the midgut of mosquito larvae. After binding with its receptor, it is probable that Bin toxin induced vaculation, pore formation, autophagy and apoptosis in the midgut cells of C. quinquefasciatus larvae to cause septicemia. Conclusively, the purified and activated insecticidal protein (Bin toxin) can be a key part of integrated vector management and has potential to be developed as commercial microbial formulation against mosquito larvae with a low risk to environment and non-target insects. The present study can be helpful to control the vectors of important diseases by understanding the interaction and binding of toxin and receptor.







Similar content being viewed by others
Data availability
All data generated or analyzed in this work are available in the published manuscript.
References
Achari TS, Barik TK, Acharya UR (2020) Toxins of Bacillus thuringiensis: a novel microbial insecticide for mosquito vector control. Molecular identification of mosquito vectors and their management. Springer, Singapore, pp 89–116. https://doi.org/10.1007/978-981-15-9456-4_5
Alves SB (1998) Controle microbiano de insetos. 2nd edn. Fundação de EstudosAgrários Luiz de Queiroz, Piracicaba
Andreychuk S, Yakob L (2022) Mathematical modelling to assess the feasibility of Wolbachia in malaria vector biocontrol. J Theor Biol 542:111110. https://doi.org/10.1016/j.jtbi.2022.111110
Bartholomew JW, Mittwer T (1952) The gram stain. Bacteriol Rev 16:1–29. https://doi.org/10.1128/br.16.1.1-29.1952
Baumann P, Clark MA, Baumann L, Broadwell AH (1991) Bacillus sphaericus as a mosquito pathogen: properties of the organism and its toxins. Microbiol Rev 55(3):425–436. https://doi.org/10.1128/mr.55.3.425-436.1991
Beegle CC, Rose RI, Ziniu Y (1991) Mass production of Bacillus thuringiensis and B. sphaericus for microbial control of insect pests. In: Maramorosch K (ed) Biotechnology for biological control of pests and vectors. CRC Press, pp 195–216. https://doi.org/10.1201/9781351070300-19
Ben-Dov E (2014) Bacillus thuringiensis subsp. israelensis and its dipteran-specific toxins. Toxins 6(4):1222–1243. https://doi.org/10.3390/toxins6041222
Bideshi DK, Park HW, Hice RH, Wirth MC, Federici BA (2017) Highly effective broad spectrum chimeric larvicide that targets vector mosquitoes using a lipophilic protein. Sci Rep 7(1):1–7. https://doi.org/10.1038/s41598-017-11717-9
Biswas K, Sinha SN (2022) Microbe-based pesticides for insect pest control and their management. In: Nayak SK, Baliyarsingh B, Singh A, Mannazzu I, Mishra BB (eds) Advances in Agricultural and Industrial Microbiology. Springer, Singapore. https://doi.org/10.1007/978-981-16-9682-4_9
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Charles JF, Silva-Filha MH, Nielsen-LeRoux C, Humphreys MJ, Berry C (1997) Binding of the 51-and 42-kDa individual components from the Bacillus sphaericus crystal toxin to mosquito larval midgut membranes from Culex and Anopheles sp.(Diptera: Culicidae). FEMS Microbiol Lett 156(1):153–159. https://doi.org/10.1111/j.1574-6968.1997.tb12721.x
Cokmus C, Davidson EW, Cooper K (1997) Electrophysiological effects of Bacillus sphaericus binary toxin on cultured mosquito cells. J Invertebr Pathol 69(3):197–204. https://doi.org/10.1006/jipa.1997.4660
Colletier JP, Sawaya MR, Gingery M, Rodriguez JA, Cascio D, Brewster AS, Eisenberg DS (2016) De novo phasing with X-ray laser reveals mosquito larvicide BinAB structure. Nature 539:43–47. https://doi.org/10.1038/nature19825
Colón-González FJ, Sewe MO, Tompkins AM, Sjödin H, Casallas A, Rocklöv J, Lowe R (2021) Projecting the risk of mosquito-borne diseases in a warmer and more populated world: a multi-model, multi-scenario inter comparison modelling study. Lan Planet Health 5(7):404–414. https://doi.org/10.1016/S2542-5196(21)00132-7
Darboux I, Nielsen-LeRoux C, Charles JF, Pauron D (2001) The receptor of Bacillus sphaericus binary toxin in Culex pipiens (Diptera: Culicidae) midgut: molecular cloning and expression. Insect Biochem Mol Biol 31(10):981–990. https://doi.org/10.1016/S0965-1748(01)00046-7
Darboux I, Pauchet Y, Castella C, Silva-Filha MH, Nielsen-LeRoux C, Charles JF, Pauron D (2002) Loss of the membrane anchor of the target receptor is a mechanism of bioinsecticide resistance. Proc Nat Acad Sci 99(9):5830–5835. https://doi.org/10.1073/pnas.092615399
de Barjac H, Lecadet MM (1976) Biochemical determination of B. thuringiensis thermostabile, exotoxin, using the inhibition of bacterial RNA polymerases. CR Acad Sci D Nat 282(23):2119–2122
Derua YA, Kahindi SC, Mosha FW, Kweka EJ, Atieli HE, Wang X, Zhou G, Lee MC, Githeko AK, Yan G (2018) Microbial larvicides for mosquito control: impact of long lasting formulations of Bacillus thuringiensis var. Israelensis and Bacillus sphaericus on non-target organisms in western Kenya highlands. Ecol Evol 8(15):7563–7573. https://doi.org/10.1002/ece3.4250
Enayati A, Hemingway J (2010) Malaria management: past, present, and future. Annu Rev Entomol 55:569–591. https://doi.org/10.1146/annurev-ento-112408-085423
Federici BA, Park HW, Bideshi DK, Wirth MC, Johnson JJ (2003) Recombinant bacteria for mosquito control. J Exp Biol 206(21):3877–3885. https://doi.org/10.1242/jeb.00643
Ferreira LM, Romão TP, de-Melo-Neto OP, Silva-Filha MHNL (2010) The orthologue to the Cpm1/Cqm1 receptor in Aedes aegypti is expressed as a midgut GPI-anchored α-glucosidase, which does not bind to the insecticidal binary toxin. Insect Biochem Mol Biol 40(8):604–610. https://doi.org/10.1016/j.ibmb.2010.05.007
Ferreira LM, Romao TP, Nascimento NA, Costa Mda C, Rezende AM, de-Melo-Neto OP, Silva-Filha MH (2014) Non conserved residues between Cqm1 and Aam1 mosquito alpha-glucosidases are critical for the capacity of Cqm1 to bind the binary toxin from Lysinibacillus sphaericus Insect Biochem Mol Biol 50:34–42. https://doi.org/10.1016/j.ibmb.2014.04.004
Finney DJ (1971) Probit analysis, 3rd edn. Cambridge University Press
Fisher TW, Garczynski SF (2012) Isolation, culture, preservation, and identification of entomopathogenic bacteria of the Bacilli A2 - In: Lacey LA (ed) Manual of techniques in invertebrate pathology (2nd edn). Academic, San Diego, pp 75–98. https://doi.org/10.1016/B978-0-12-386899-2.00003-8
Fontes G, Leite AB, Vasconcelos de Lima AR, Freitas H, Ehrenberg JP, da Rocha EMM (2012) Lymphatic filariasis in Brazil: epidemiological situation and outlook for elimination. Parasites Vectors 5(1):1–11. https://doi.org/10.1186/1756-3305-5-272
Humphreys MJ, Berry C (1998) Variants of the Bacillus sphaericus binary toxins: implications for differential toxicity of strains. J Invertebr Pathol 71(2):184–185. https://doi.org/10.1006/jipa.1997.4711
Kauffman E, Payne A, Franke MA, Schmid MA, Harris E, Kramer LD (2017) Rearing of Culex spp. and Aedes spp. Mosquitoes. Bio Protoc 7(17). https://doi.org/10.21769/BioProtoc.2542
Li Y, Zhou G, Zhong D, Wang X, Hemming-Schroeder E, David RE, Yan G (2021) Widespread multiple insecticide resistance in the major dengue vector Aedes albopictus in Hainan Province, China. Pest Manag Sci 77(4):1945–1953. https://doi.org/10.1002/ps.6222
Lopes RP, Lima JBP, Martins AJ (2019) Insecticide resistance in Culex quinquefasciatus say, 1823 in Brazil: a review. Parasites Vectors 12:591. https://doi.org/10.1186/s13071-019-3850-8
Messina JP, Brady OJ, Golding N, Kraemer MU, Wint GR, Ray SE, Pigott DM, Shearer FM, Johnson K, Earl L, Marczak LB (2019) The current and future global distribution and population at risk of dengue. Nat Microbiol 4(9):1508–1515. https://doi.org/10.1038/s41564-019-0476-8
Nauen R (2007) Insecticide resistance in disease vectors of public health importance. Pest Manag Sci 63(7):628–633. https://doi.org/10.1002/ps.1406
Navarro-Payá D, Flis I, Anderson MA, Hawes P, Li M, Akbari OS, Alphey L (2020) Targeting female flight for genetic control of mosquitoes. PLoS Negl Trop Dis 14(12):e0008876. https://doi.org/10.1371/journal.pntd.0008876
Nicolas L, Dossou-Yovo J, Hougard JM (1987) Persistence and recycling of Bacillus sphaericus 2362 spores in Culex quinquefasciatus breeding sites in West Africa. Appl Microbiol Biotechnol 25(4):341–345. https://doi.org/10.1007/BF00252544
Opota O, Charles JF, Warot S, Pauron D, Darboux I (2008) Identification and characterization of the receptor for the Bacillus sphaericus binary toxin in the malaria vector mosquito, Anopheles gambiae Comp Biochem Physiol Biochem Molec Biol 149(3):419–427. https://doi.org/10.1016/j.cbpb.2007.11.002
Palma L (2015) Protocol for the fast isolation and identification of insecticidal Bacillus thuringiensis strains from soil (6). https://doi.org/10.5376/bt.2015.06.0007
Paris M, Tetreau G, Laurent F, Lelu M, Despres L, David JP (2011) Persistence of Bacillus thuringiensis israelensis (bti) in the environment induces resistance to multiple bti toxins in mosquitoes. Pest Manag Sci 67(1):122–128. https://doi.org/10.1002/ps.2046
Prabakaran G, Hoti SL, Paily KP (2009) Development of cost-effective medium for the large-scale production of a mosquito pupicidal metabolite from Pseudomonas fluorescens Migula. Biol Control 48(3):264–266. https://doi.org/10.1016/j.biocontrol.2008.10.011
Qadri M, Short S, Gast K, Hernandez J, Wong ACN (2020) Microbiome innovation in agriculture: development of microbial based tools for insect pest management. Front Sustain Food Syst 4:547751. https://doi.org/10.3389/fsufs.2020.547751
Rezende TMT, Romão TP, Batista M, Berry C, Adang MJ, Silva-Filha MHNL (2017) Identification of Cry48Aa/Cry49Aa toxin ligands in the midgut of Culex quinquefasciatus larvae. Insect Biochem Mol Biol 88:63–70. https://doi.org/10.1016/j.ibmb.2017.08.001
Riaz MA, Adang MJ, Hua G, Rezende TMT, Rezende AM, Shen GM (2020) Identification of Lysinibacillus sphaericus binary toxin binding proteins in a malarial mosquito cell line by proteomics: a novel approach towards improving mosquito control. J Proteom 227:103918. https://doi.org/10.1016/j.jprot.2020.103918
Rojas-Pinzon PA, Dussan J (2017) Efficacy of the vegetative cells of Lysinibacillus sphaericus for biological control of insecticide-resistant Aedes aegypti. Parasites Vectors 10(1):1–7. https://doi.org/10.1186/s13071-017-2171-z
Romao TP, de MeloChalegre KD, Key S, Ayres CF, Fontes de Oliveira CM, de-Melo-Neto OP, Silva-Filha MH (2006) A second independent resistance mechanism to Bacillus sphaericus binary toxin targets its alpha-glucosidase receptor in Culex quinquefasciatus FEBS J 273(7):1556–1568. https://doi.org/10.1111/j.1742-4658.2006.05177.x
Silva-Filha MH, Nielsen-Leroux C, Charles JF (1997) Binding kinetics of Bacillus sphaericus binary toxin to midgut brush-border membranes of Anopheles and Culex sp. mosquito larvae. Eur J Biochem 247(3):754–761. https://doi.org/10.1111/j.1432-1033.1997.00754.x
Silva-Filha MH, Nielsen-LeRoux C, Charles JF (1999) Identification of the receptor for Bacillus sphaericus crystal toxin in the brush border membrane of the mosquito Culex pipiens (Diptera: Culicidae). Insect Biochem Mol Biol 29(8):711–72. https://doi.org/10.1016/S0965-1748(99)00047-8
Silva-Filha MHNL, Berry C, Regis L (2014) Lysinibacillus sphaericus: toxins and mode of action, applications for mosquito control and resistance management. Adv Insect Phys 47:89–176. https://doi.org/10.1016/B978-0-12-800197-4.00003-8
Silva-Filha MHNL, Romão TP, Rezende TMT, Carvalho KDS, Gouveia de Menezes HS, Alexandre do Nascimento N, Bravo A (2021) Bacterial toxins active against mosquitoes: Mode of action and resistance. Toxins 13(8):523. https://doi.org/10.3390/toxins13080523
Tangsongcharoen C, Chomanee N, Promdonkoy B, Boonserm P (2015) Lysinibacillus sphaericus binary toxin induces apoptosis in susceptible Culex quinquefasciatus larvae. J Invertebr Pathol 128:57–63. https://doi.org/10.1016/j.jip.2015.04.008
Tangsongcharoen C, Jupatanakul N, Promdonkoy B, Dimopoulos G, Boonserm P (2017) Molecular analysis of Culex quinquefasciatus larvae responses to Lysinibacillus sphaericus bin toxin. PLoS ONE 12(4):e0175473. https://doi.org/10.1371/journal.pone.0175473
Wang GH, Du J, Chu CY, Madhav M, Hughes GL, Champer J (2022) Symbionts and gene drive: two strategies to combat vector-borne disease. Trends Genet. https://doi.org/10.1016/j.tig.2022.02.013
Weisburg WG, Barns SM, Pelletier DA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173(2):697–703. https://doi.org/10.1128/jb.173.2.697-703.1991
Wilson K (2001) Preparation of genomic DNA from bacteria. Curr Protoc Mol Biol 56(1):2–4. https://doi.org/10.1002/0471142727.mb0204s56
Woradulayapinij W, Audtho M, Wiwat C (2015) Suppression of α-glucosidase gene by RNA interference can increase resistance of Culex quinquefasciatus to Bacillus sphaericus binary toxin. Biocontrol Sci Technol 25(12):1453–1466. https://doi.org/10.1080/09583157.2015.1066310
World Health Organization (2020) WHO malaria policy advisory committee (MPAC) meeting: meeting report, May 2020
Zhang Q, Hua G, Bayyareddy K, Adang MJ (2013) Analyses of α-amylase and α-glucosidase in the malaria vector mosquito, Anopheles gambiae, as receptors of Cry11Ba toxin of Bacillus thuringiensis subsp. jegathesan. Insect Biochem Mol Biol 43(10):907–915. https://doi.org/10.1016/j.ibmb.2013.07.003
Zhang Z, Schwartz S, Wagner L, Miller W (2000) A greedy algorithm for aligning DNA sequences. J Comput Biol 7(1–2):203–214. https://doi.org/10.1089/10665270050081478
Acknowledgements
Authors are thankful to Dr. Salman Ahmed (Department of Plant Pathology, College of Agriculture, University of Sargodha, Sargodha, Pakistan) for the technical assistance during the study.
Funding
This research work was financially supported by the Higher Education Commission of Pakistan (HEC) under its NRPU Program (Project No. NRPU-11326).
Author information
Authors and Affiliations
Contributions
MAR and SA conceived the idea and designed the study. SI, AA and ZA performed experimentation. MAR and SI analyzed the data and prepared results. MAR and MZM wrote the first draft of manuscript. MZM and SA technically proofread the manuscript. MAR and MS supervised the research work. MA and MAR provided technical and financial assistance to the study. All authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 38.2 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Iftikhar, S., Riaz, M.A., Majeed, M.Z. et al. Isolation, characterization and larvicidal potential of indigenous soil inhabiting bacteria against larvae of southern house mosquito (Culex quinquefasciatus Say). Int J Trop Insect Sci 43, 781–791 (2023). https://doi.org/10.1007/s42690-023-00992-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-023-00992-x