Abstract
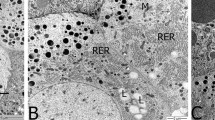
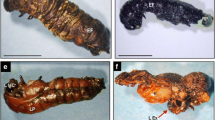
Insect development is disrupted by juvenile hormone (JH) and their mimics in several ways. Many of them have been thoroughly investigated and are already being used to manage pest insects with commercially available agents. This research aims to explore the potentials of juvenile hormone analogue fenoxycarb on morphological, histopathological, and biochemical changes in the ovary of Spodoptera mauritia. Newly emerged female pupae were treated topically with sublethal doses (LD10, LD25) of fenoxycarb to determine their effects on reproduction. The results reveal that this juvenile hormone analogue affects the normal development of the ovary tissue by reducing the number of oocytes and oogonia in the ovaries of S. mauritia. Fenoxycarb treated pupae showed a substantial decrease in the reclaimed adultoids ovaries development and reduction in length of ovarioles, area of basal oocytes and the total number of eggs laid. A microscopic examination exhibited reduced pulsating movements, tumour-like bulbous masses and the germarium region exhibited hypertrophy. Histological investigation of ovaries indicated a degeneration of ovarian follicle cells, deformed oocytes with deteriorated trophocytes, malformed egg chamber, vacuolated ooplasm and defective vitellogenesis in malformed adult female’s ovarioles. The effect of fenoxycarb could be correlated with quantitative depletion of proteins, lipids, and carbohydrates in gonads of the treated groups and there were no significant changes in sodium dodecyl sulphate(SDS)- protein pattern. This study forms baseline data suggesting that fenoxycarb respond considerably for the control of the lepidopteran pest of paddy (S. mauritia) effectively.










Similar content being viewed by others
References
Banu CA, Sathyakala KP, Manogem EM (2019a) Efficacy of JHA, Fenoxycarb on egg hatchability and postembryonic development of Spodoptera mauritia Boisd. (Lepidoptera: Noctuidae). Int J Adv Res 7(6). https://doi.org/10.21474/IJAR01/9198
Banu CA, Sathyakala KP, Manogem EM (2019b) Evaluation and screening of growth disrupting juvenile hormone analogue, fenoxycarb on the development and metamorphosis of Spodoptera mauritia Boisd.(Lepidoptera: Noctuidae). Crop Res 54(5and6):158–164. https://doi.org/10.31830/2454-1761.2019.027
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principleof protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Bruce DH, Gary BQ (1981) Metabolism and mode of action of juvenile hormone, juvenoids and other insect growth regulators John Wiley and sons Ltd. Progress in pesticide Biochemistry 1
Chanbang Y, Arthur FH, Wilde GE, Throne JE, Subramanyam B (2008) Susceptibility of eggs and adult fecundity of the lesser grain borer, Rhyzopertha dominica, exposed to methoprene. J Insect Sci 8(1):48. https://doi.org/10.1673/031.008.4801
Dahm KH, Bhaskaran G, Peter MG, Shirk P D, Seshan KR, Röller H (1976) On the identity of the juvenile hormone in insects. The Juvenile Hormones. Springer, Boston, MA, 19–47. https://doi.org/10.1007/978-1-4684-7947-8_3
Fathpour H, Nouri A, Zeynali B (2007) Effects of a juvenoid pyriproxyfen on reproductive organ development and reproduction in German Cockroach (Dictyoptera: Blattellidae). J Insect Physiol 21:1755–1762. https://doi.org/10.22099/IJSTS.2007.2319
Fotouhi K, Fazel MM, Kavousi A (2015) Effects of pyriproxyfen on bioenergetic resources of Leptinotarsa decemlineata (Say)(Coleoptera: Chrysomelidae). Türk Entomol Derg 39:11–22. https://doi.org/10.16970/ted.14717
Gerhardt P, Murray RGE, Wood WA, Krieg NR (1994) Methods for General and Molecular Bacteriology. American Society for Microbiology, Washington, DC
Ghasemi A, Sendi J, Ghadamyari M (2010) Physiological and biochemical effect of pyriproxyfen on Indian meal moth Plodia interpunctella (Hübner)(Lepidoptera: Pyralidae). Journal of Plant Protection Research 50:416–422. https://doi.org/10.2478/v10045-010-0070-9
Govind TR (2014) Research article effect of juvenile hormone analogue (fenoxycarb) on ovarian development of Dysdercus similis. Int J Recent Sci Res 5(9):1714–1716. https://recentscientific.com/effect-juvenile-hormone-analogue-fenoxycarb-ovarian-development-dysdercus-similis
Indrasith LS, Sasaki T, Yaginuma T, Yamashita O (1988) The occurrence of a premature form of egg-specific protein in vitellogenic follicles of Bombyx mori. J Comp Physiol B 158(1):1–7. https://doi.org/10.1007/2FBF00692723
Kanost MR, Kawooya JK, Law JH, Ryan RO, Van Heusden MC, Ziegler R (1990) Insect haemolymph proteins. Advances in Insect Physiology 22:299–396. https://doi.org/10.1016/S0065-2806(08)60008-9
Kim DK (2001) Effectof juvenile hormone on metamorphosis and vitellogenesis of the beet armyworm, Spodoptera exigua (Doctoral dissertation, MS thesis, Andong National University, Andong, Korea)
Kunkel JG, Nordin JH (1985) Yolk proteins. Compr Insect Physiol Biochem Pharmacol 1:83-111. http://marlin.bio.umass.edu/biology/kunkel/pub/reprints/kn-1985.pdf
Laemmli UK (1970) Cleavage of structural proteins during assembly of head bacteriophage T. Nature 227:680–685. https://doi.org/10.1038/227680a0
Leonardi MG, Marciani P, Montorfano PG, Cappellozza S, Giordana B, Monticelli G (2001) Effect of fenoxycarb on leucine uptake and lipid composition of midgut brush border membrane in the silkworm, Bombyx mori (Lepidoptera, Bombycidae). Pestic Biochem Physiol 70(1):42–51. https://doi.org/10.1006/pest.2001.2537
Maiza A, Kilani-Morakchi S, Farine JP, Smagghe G, Aribi N, Soltani N (2004) Reproductive effects in german cochroaches by ecdysteroid agonist RH-0345, juvenile hormone analogue methoprene and carbamate benfuracarb. Commun Appl Biol Sci Ghent Uni 69:257–266. https://pubmed.ncbi.nlm.nih.gov/15759422/
Mandal D, Chaudhuri DK (1992) Studies on carbohydrate, protein and lipid levels in normal and stress conditions in fat body and integument as compared to whole body during development of rice moth, Corcyra cephalonica (St.). Int J Trop Insect Sci 13(1):121–128. https://doi.org/10.1017/S1742758400013977
Mathai S, Nair VSK (1990) Histomorphological changes induced in the ovary of Spodoptera mauritia Boisd.(Lepidoptera: Noctuidae) by treatment with a juvenile hormone analogue. Proceedings of the Indian National Science Academy. Part B Biol Scie 56(3):253–258. https://insa.nic.in/writereaddata/UpLoadedFiles/PINSA/Vol56B_1990_3_Art03.pdf
Metwally MM, Landa V (1972) Sterilization of the Khapra beetle, Trogoderma granarium Everts, with juvenile hormone analogues. Z Angew Entomol 72:97–109. https://doi.org/10.1111/j.1439-0418.1972.tb02223.x
Mondal KAMSH, Parween S (2000) Insect growth regulators and their potential in the management of stored-product insect pests. Integr Pest Manag Rev 5(4):255–295. https://doi.org/10.1023/A:1012901832162
Nijhout HF (1994) Reproduction. In: Nijhout HF (ed) Insect Hormone. Princeton University, Princeton, pp 142–159
Qian HY, Zhang X, Zhao GD, Guo HM, Li G, Xu AY (2020) Effects of Pyriproxyfen Exposure on Reproduction and Gene Expressions in Silkworm Bombyx mori. Insects 11(8):467. https://doi.org/10.3390/insects11080467
Richardson ML, Lagos DM (2007) Effects of a juvenile hormone analogue, pyriproxyfen, on the apterous form of soybean aphid (Aphis glycines). J Appl Entomol 131(5):297–302. https://doi.org/10.1111/j.1439-0418.2007.01163.x
Rohdendorf EB, Sehnal F (1972) The induction of ovarian dysfunctions in Thermobia domestica by the Cecropia juvenile hormones. Experientia 28(9):1099–1101. https://doi.org/10.1007/BF01918699
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Tinevez JY (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9(7):676–682. https://doi.org/10.1038/nmeth.2019
Sturm MDL (1978) Aspects of the biology of Scomberomorus maculatus (Mitchill) in Trinidad. J Fish Biol 13(2):155–172. https://doi.org/10.1111/j.1095-8649.1978.tb03423.x
Telfer WH, Woodruff RI, Huebner E (1981) Electrical polarity and cellular differentiation in meroistic ovaries. Am Zool 21(3):675–686. https://doi.org/10.1093/icb/21.3.675
Tobe SS, Stay B (1985) Structure and regulation of the corpus allatum. Adv In Insect Phys 18:305–432. https://doi.org/10.1016/S0065-2806(08)60043-0
van Handel E (1985) Rapid determination of total lipids in mosquitoes. J Am Mosq Control Assoc 1:302–304. https://www.biodiversitylibrary.org/content/part/JAMCA/JAMCA_V01_N3_P302-304.pdf
Verma V (1990) Effect of JH : Ecdysone and other factors on the regulation of reproduction in insects. Ph. D. Thesis, Dr. H. S. Gour University, Sagar (M.P). http://hdl.handle.net/10603/38190
Wheeler D (1996) The role of nourishment in oogenesis. Annu Rev Entomol 41(1):407–431
Wigglesworth VB (1964) The hormonal regulation of growth and reproduction in insects. Adv Insect Physiol 247–336.https://doi.org/10.1146/annurev.en.41.010196.002203
Xu Q, Tang B, Zou Q, Zheng H, Liu X, Wang S (2015) Effects of pyriproxyfen on female reproduction in the common cutworm, Spodoptera litura (F.)(Lepidoptera: Noctuidae). PloS One 10(10):e0138171. https://doi.org/10.1371/journal.pone.0138171
Zibaee A, Zibaee I, Sendi JJ (2011) A juvenile hormone analog, pyriproxifen, affects some biochemical components in the hemolymph and fat bodies of Eurygaster integriceps Puton (Hemiptera: Scutelleridae). Pestic Biochem Physiol 100(3):289–298. https://doi.org/10.1016/j.pestbp.2011.05.002
Acknowledgements
We gratefully acknowledge the Department of Zoology, University of Calicut, Kerala, India, for supporting and providing the essential infrastructure facility needed for this study. Individual research funding provided as major research project DST-SERB, New Delhi (SB/FT/LS-402/2012) for the corresponding author is gratefully acknowledged. Thanks are also extended to the special assistance program (SAP) of the Department of Zoology, University of Calicut.
Funding
The first author was funded by the University Grant Commission (UGC), New Delhi, in the form of Maulana Azad National Fellowship for 2015–16 F1-17.1/2015–16/MANF-2015–17-KER-51524 /(SA-III/Website).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors of this study announce no conflict of interest about the publication and dissemination of information.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Banu, C.A., Praseeja, C. & Manogem, E. Evaluation of the histopathological and biochemical effects of fenoxycarb in the ovaries of Spodoptera mauritia (Lepidoptera: Noctuidae). Int J Trop Insect Sci 42, 2413–2424 (2022). https://doi.org/10.1007/s42690-022-00769-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-022-00769-8