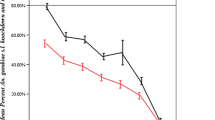
Abstract
Poor domestic waste management in unplanned urban areas have resulted into formation of polluted potential malaria vector larval habitats. Most of these larval habitats are contaminated with domestic pollutants such as hydrogen peroxide and detergents. The present study was designed to evaluate the susceptibility status of Anopheles gambie and Anopheles arabiensis to commonly used insecticides after being exposed to hydrogen peroxide and soap detergents in northern Tanzania. Anopheles gambiae s.s larvae were collected from the laboratory colony whereas those for An. arabiensis were sampled from the rice field in lower Moshi. The larvae were reared in two arms, larval habitats in arm one had water containing soaps while in arm two, the larval habitats contained hydrogen peroxide at different concentrations (0.001%, 0.0001%,0.00001% and 0.000001%). Emerging adult’s female of the two Anopheles species from the two larval habitats were exposed to 0.75% and 0.05% of permethrin and deltamethrin WHO Insecticides impregnated standard papers. Knockdown rates were observed for 1 h and mortality rates within 24 h after exposure. Regression analysis was performed using probit analysis to establish knockdown rates and lethal concentration for 50% and 95%. The wild and laboratory anopheles populations reared in detergent or hydrogen peroxide in different concentrations showed reduced susceptibility to permethrin and deltamethrin. The median knockdown time for 50% of the population (KDT50) ranged between 13.52 min (10.98–19.74) to 30.67 min (28.53–34.34) while the KDT95 raised between 32.01 min (29.47–38.23) to 111.84 min (109.70–115.54) for those reared in detergent and exposed to deltamethrin and permethrin. For those reared in hydrogen peroxide and exposed to deltamethrin and permethrin, KDT50 ranged between 8.24 min (6.92–13.51) to 24.28 min(22.70–27.46) while KDT95 ranged between 25.94 min(24.62–31.21) to 118.68 min(117.10–121.86). Mortality of wild population exposed to standard insectides treated papers after reared in detergent ranged between 40 to 95.3% while for those reared in hydrogen peroxide ranged between 67 to 95.3%. The findings of this study demonstrate that domestic waste pollutants such as hydrogen peroxide and soaps are partly associated with reduced susceptibility to insecticides among potential malaria vectors.

Similar content being viewed by others
References
Antonio-Nkondjio C, Youmsi-Goupeyou M, Kopya E, Tene-Fossog B, Njiokou F, Costantini C, Awono-Ambene P (2014) Exposure to disinfectants (soap or hydrogen peroxide) increases tolerance to permethrin in Anopheles gambiae populations from the city of Yaoundé, Cameroon. Malar J 13(1):296. https://doi.org/10.1186/1475-2875-13-296
Azrag RS, Mohammed BH (2018) Anopheles arabiensis in Sudan: a noticeable tolerance to urban polluted larval habitats associated with resistance to Temephos. Malar J 17(1):204. https://doi.org/10.1186/s12936-018-2350-1
Bigoga JD, Nanfack FM, Awono-Ambene PH, Patchoké S, Atangana J, Otia VS, Fondjo E, Moyou RS, Leke RG (2012) Seasonal prevalence of malaria vectors and entomological inoculation rates in the rubber cultivated area of Niete, south region of Cameroon. Parasit Vectors 5(1):197. https://doi.org/10.1186/1756-3305-5-197
Braimah N, Drakeley C, Kweka E et al (2005) Tests of bednet traps (Mbita traps) for monitoring mosquito populations and time of biting in Tanzania and possible impact of prolonged insecticide treated net use. Int J Trop Insect Sci 25(3):208–213
Chandre F, Darriet F, Manguin S, Brengues C, Carnevale P, Guillet P (1999a) Pyrethroid cross resistance spectrum among populations of Anopheles gambiae ss from cote d'Ivoire. J Am Mosq Control Assoc 15(1):53–59
Chandre F, Manguin S, Brengues C, Dossou Yovo J, Darriet F, Diabate A, Carnevale P, Guillet P (1999b) Current distribution of a pyrethroid resistance gene (kdr) in Anopheles gambiae complex from West Africa and further evidence for reproductive isolation of the Mopti form. Parassitologia 41(1–3):319–322
Fouet C, Gamez S, Kamdem C, White BJ (2017) Pollutants and insecticides drive local adaptation in African malaria mosquitoes. Mol Biol Evol 34(5):1261–1275. https://doi.org/10.1093/molbev/msx087
Kabula B, Kisinza W, Tungu P et al (2014) Co-occurrence and distribution of east (L1014S) and west (L1014F) African knock-down resistance in Anopheles gambiae sensu lato population of Tanzania. Tropical Med Int Health 19(3):331–341. https://doi.org/10.1111/tmi.12248
Kisinza WN, Nkya TE, Kabula B, Overgaard HJ, Massue DJ, Mageni Z, Greer G, Kaspar N, Mohamed M, Reithinger R, Moore S, Lorenz LM, Magesa S (2017) Multiple insecticide resistance in Anopheles gambiae from Tanzania: a major concern for malaria vector control. Malar J 16(1):439. https://doi.org/10.1186/s12936-017-2087-2
Kivuyo HS, Mbazi PH, Kisika DS, Munga S, Rumisha SF, Urasa FM, Kweka EJ (2014) Performance of five food regimes on Anopheles gambiae Senso Stricto larval rearing to adult emergence in insectary. PLoS One 9(10):e110671. https://doi.org/10.1371/journal.pone.0110671
Komalamisra N, Srisawa R, Phanbhuwong T, Oatwanees S (2011) Insecticide susceptibility of the dengue vector Aedes Aegypti (L) in Metropolitanian Bankok. SE Asian J Trop Med Public Health 42:814–823
Kudom AA (2015) Larval ecology of Anopheles coluzzii in Cape Coast, Ghana: water quality, nature of habitat and implication for larval control. Malar J 14(1):447. https://doi.org/10.1186/s12936-015-0989-4
Kweka EJ, Nkya WM, Mahande AM et al (2008) Mosquito abundance, bed net coverage and other factors associated with variations in sporozoite infectivity rates in four villages of rural Tanzania. Malar J 7(1):59
Kweka EJ, Nyindo M, Mosha F, Silva AG (2011) Insecticidal activity of the essential oil from fruits and seeds of Schinus terebinthifolia Raddi against African malaria vectors. Parasit Vectors 4(1):129. https://doi.org/10.1186/1756-3305-4-129
Kweka EJ, Mausa EA, Venter N, Derua YA, Kimaro EE, Coetzee M (2018) Application of hydrolysis probe analysis to identify clade types of the malaria vector mosquito Anopheles funestus sensu stricto from Muheza, northeastern Tanzania. Med Vet Entomol 32(1):125–128. https://doi.org/10.1111/mve.12274
Lindsay S, Shenton F, Snow R, Greenwood B (1989) Responses of Anopheles gambiae complex mosquitoes to the use of untreated bednets in the Gambia. Med Vet Entomol 3(3):253–262
Mahande A, Dusfour I, Matias J, Kweka E (2012) Knockdown resistance, Rdl alleles, and the annual entomological inoculation rate of wild mosquito populations from lower Moshi, northern Tanzania. J Global Infect Dis 4(2):114–119. https://doi.org/10.4103/0974-777x.96776
Matowo J, Kulkarni MA, Mosha FW, Oxborough RM, Kitau JA, Tenu F, Rowland M (2010) Biochemical basis of permethrin resistance in Anopheles arabiensis from lower Moshi, North-Eastern Tanzania. Malar J 9(1):193. https://doi.org/10.1186/1475-2875-9-193
Mattah PAD, Futagbi G, Amekudzi LK, Mattah MM, de Souza DK, Kartey-Attipoe WD, Bimi L, Wilson MD (2017) Diversity in breeding sites and distribution of Anopheles mosquitoes in selected urban areas of southern Ghana. Parasit Vectors 10(1):25. https://doi.org/10.1186/s13071-016-1941-3
Mbepera S, Nkwengulila G, Peter R, Mausa EA, Mahande AM, Coetzee M, Kweka EJ (2017) The influence of age on insecticide susceptibility of Anopheles arabiensis during dry and rainy seasons in rice irrigation schemes of northern Tanzania. Malar J 16(1):364. https://doi.org/10.1186/s12936-017-2022-6
Mdoe FP, Nkwengulila G, Chobu M et al (2014) Larvicidal effect of disinfectant soap on Anopheles gambiae s.s (Diptera: Culicidae) in laboratory and semifield environs. Parasit Vectors 7(1):211. https://doi.org/10.1186/1756-3305-7-211
Mireji PO, Keating J, Hassanali A, Mbogo CM, Nyambaka H, Kahindi S, Beier JC (2008) Heavy metals in mosquito larval habitats in urban Kisumu and Malindi, Kenya, and their impact. Ecotoxicol Environ Saf 70(1):147–153. https://doi.org/10.1016/j.ecoenv.2007.03.012
Mireji P, Keating J, Hassanali A, Impoinvil DE, Mbogo CM, Muturi MN, Nyambaka H, Kenya EU, Githure JI, Beier JC (2010a) Expression of metallothionein and α-tubulin in heavy metal-tolerant Anopheles gambiae sensu stricto (Diptera: Culicidae). Ecotoxicol Environ Saf 73(1):46–50. https://doi.org/10.1016/j.ecoenv.2009.08.004
Mireji P, Keating J, Hassanali A et al (2010b) Biological cost of tolerance to heavy metals in the mosquito Anopheles gambiae. Med Vet Entomol 24(2):101–107. https://doi.org/10.1111/j.1365-2915.2010.00863.x
Nkya TE, Akhouayri I, Poupardin R, Batengana B, Mosha F, Magesa S, Kisinza W, David JP (2014a) Insecticide resistance mechanisms associated with different environments in the malaria vector Anopheles gambiae: a case study in Tanzania. Malar J 13(1):28. https://doi.org/10.1186/1475-2875-13-28
Nkya TE, Mosha FW, Magesa SM, Kisinza WN (2014b) Increased tolerance of Anopheles gambiae ss to chemical insecticides after exposure to agrochemical mixture. Tanzania J Health Res 16(4):329–32
Nkya TE, Poupardin R, Laporte F, Akhouayri I, Mosha F, Magesa S, Kisinza W, David JP (2014c) Impact of agriculture on the selection of insecticide resistance in the malaria vector Anopheles gambiae: a multigenerational study in controlled conditions. Parasit Vectors 7(1):480. https://doi.org/10.1186/s13071-014-0480-z
Nnko EJ, Kihamia C, Tenu F, Premji Z, Kweka EJ (2017) Insecticide use pattern and phenotypic susceptibility of Anopheles gambiae sensu lato to commonly used insecticides in lower Moshi, northern Tanzania. BMC Res Notes 10(1):443. https://doi.org/10.1186/s13104-017-2793-4
Oduola AO, Idowu ET, Oyebola MK et al (2012) Evidence of carbamate resistance in urban populations of Anopheles gambiae s.s. mosquitoes resistant to DDT and deltamethrin insecticides in Lagos, South-Western Nigeria. Parasit Vectors 5(1):116. https://doi.org/10.1186/1756-3305-5-116
Perez MH, Noriega FG (2012) Aedes aegypti pharate 1st instar quiescence affects larval fitness and metal tolerance. J Insect Physiol 58(6):824–829. https://doi.org/10.1016/j.jinsphys.2012.03.005
Temu E, Minjas J, Coetzee M, Hunt R, Shiff CJ, Hygiene (1998) The role of four anopheline species (Diptera: Culicidae) in malaria transmission in coastal Tanzania. Trans R Soc Trop Med 92(2):152–158
WHO (1975) Manual on practical entomology in malaria. Part II. Methods and techniques. WHO, Geneva
WHO (2000) WHO expert committee on malaria: twentieth report. World Health Organization
WHO (2005) Guidelines for laboratory and field testing of mosquito larvicides. World Health Organization, Geneva
WHO (2016) Test procedures for insecticide resistance monitoring in malaria vector mosquitoes. WHO, Geneva
WHO (2019) World malaria report 2019. World Health Organisation, Geneva
Yadouleton AWM, Asidi A, Djouaka RF, Braïma J, Agossou CD, Akogbeto MC (2009) Development of vegetable farming: a cause of the emergence of insecticide resistance in populations of Anopheles gambiae in urban areas of Benin. Malar J 8(1):103. https://doi.org/10.1186/1475-2875-8-103
Acknowledgements
Authors would like to extend our appreciation to Insectary staff, Ibrahim Sungi and Adrian Massawe, for rearing An. gambiae larvae. Authors are in debt appreciating the help rendered by Dr. Flora Magige and Dr. Hendry Ndangarasi in organizing procedures of conducting this research. Prof. Karl Martin is appreciated for the assistance gave in data analysis. The authors are thankful to Mabogini field station field assistants who did collection of An. arabiensis wild larvae and rearing. TPRI is acknowledge for granting her premises to be used for these research activities.
Funding
This study had no financial support from any source. It used available resources from TPRI insectary and Mabogini field station.
Author information
Authors and Affiliations
Contributions
FDS and EJK conceived and designed the study. AT, FDS and AMM coordinated experiments. FDS undertook laboratory work. FDS, WK and EJK analysed and interpreted results. FDS, WK, HDM, AT, AMM and EJK drafted the manuscript and revised to final version. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
The protocol of this study was reviewed and approved by the ethical review board of University of Dar-es-salaaam.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shayo, F.D., Kidima, W., Thomas, A. et al. Exposure of malaria vector larval habitats to domestic pollutants escalate insecticides resistance: experimental proof. Int J Trop Insect Sci 40, 729–740 (2020). https://doi.org/10.1007/s42690-020-00123-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-020-00123-w