Abstract
Purpose
Many works have reported changes in the visual cortex regarding stimulation with different flickering frequencies using various neuroimaging techniques. Also, many phosphorus functional magnetic resonance spectroscopy (31P-fMRS) studies have reported high-energy phosphate (HEP) changes in the visual cortex during visual stimulation at 8 Hz, compared with rest periods. However, there have been no studies attempting to measure HEP changes during visual stimulation with different flickering frequencies. The aim of this work was to use the 31P-fMRS technique to assess possible HEP changes during visual stimulation experiments with stimuli flickering at 4 Hz or 8 Hz, and compare the results obtained with the different frequencies.
Methods
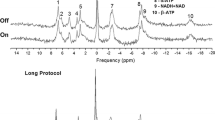
Nineteen subjects participated in the study. Experiments consisted of 31P-fMRS acquisitions in a 3T magnetic resonance scanner while subjects underwent a five blocks Rest/Stimulus protocol, with stimuli flickering at either 4 Hz or 8 Hz. We assessed pH; the alpha and gamma peaks of adenosine tri-phosphate (α-ATP and γ-ATP); the ratios phosphocreatine/inorganic phosphate (Pi), and of the metabolites phosphocreatine, Pi, glycerophosphocholine, phosphoethanolamine, and glycerophosphoethanolamine with respect to α-ATP and γ-ATP; and the linewidth of the phosphocreatine peak.
Results
pH was stable along acquisitions for both 4 Hz and 8 Hz. No significant changes were found for neither α-ATP, γ-ATP, metabolite ratios, nor phosphocreatine linewidth.
Conclusion
This was probably due to the compromise existing between signal-to-noise ratio and temporal/spatial resolutions—the former requires long acquisitions/large voxels which possibly result in missing alterations and partial volume effects regarding changes detected by other techniques such as functional magnetic resonance imaging. These effects may have been amplified by the use of a non-localized sequence.




Similar content being viewed by others
References
Barreto FR, Costa TBS, Landim RCG, Castellano G, Salmon CEG. 31P-MRS using visual stimulation protocols with different durations in healthy young adult subjects. Neurochem Res. 2014;39:2343–50. https://doi.org/10.1007/s11064-014-1433-9.
Cadoux-Hudson T, Blackledge M, Radda G. Imaging of human brain creatine kinase activity in vivo. FASEB J. 1989;3:2660–6.
Chen W, Zhu XH, Adriany G, Ugurbil K. Increase of creatine kinase activity in the visual cortex of human brain during visual stimulation: a 31P magnetization transfer study. Magn Reson Med. 1997;38:551–7. https://doi.org/10.1002/mrm.1910380408.
Chen C, Stephenson MC, Peters A, Morris PG, Francis ST, Gowland PA. 31P magnetization transfer magnetic resonance spectroscopy: assessing the activation induced change in cerebral ATP metabolic rates at 3 T. Magn Reson Med. 2018;79:22–30. https://doi.org/10.1002/mrm.26663.
Emir UE, Bayraktaroglu Z, Ozturk C, Ademoglu A, Demiralp T. Changes in BOLD transients with visual stimuli across 1–44 Hz. Neurosci Lett. 2008;436:185–8. https://doi.org/10.1016/j.neulet.2008.03.021.
Heo HY, Wemmie J, Thedens D, Magnotta VA. Evaluation of activity-dependent functional pH and T1ρ response in the visual cortex. Neuroimage. 2014;95:336–43. https://doi.org/10.1016/j.neuroimage.2014.01.042.
Herrmann CS. Human EEG responses to 1–100 Hz flicker: resonance phenomena in visual cortex and their potential correlation to cognitive phenomena. Exp Brain Res. 2001;137:346–53. https://doi.org/10.1007/s002210100682.
Hetherington HP, Pan JW, Spencer DD. 1H and 31P spectroscopy and bioenergetics in the lateralization of seizures in temporal lobe epilepsy. J Magn Reson Imaging. 2002;16:477–83. https://doi.org/10.1002/jmri.10177.
Jayakumar PN, Gangadhar BN, Venkatasubramanian G, Desai S, Velayudhan L, Subbakrishna D, et al. High energy phosphate abnormalities normalize after antipsychotic treatment in schizophrenia: a longitudinal 31P MRS study of basal ganglia. Psychiatry Res. 2010;181:237–40. https://doi.org/10.1016/j.pscychresns.2009.10.010.
Kato T, Murashita J, Shioiri T, Hamakawa H, Inubushi T. Effect of photic stimulation on energy metabolism in the human brain measured by 31P-MR spectroscopy. J Neuropsychiatry Clin Neurosci. 1996;8:417–22. https://doi.org/10.1176/jnp.8.4.417.
Kato T, Murashita J, Shioiri T, Terada M, Inubushi T, Kato N. Photic stimulation-induced alteration of brain energy metabolism measured by 31P-MR spectroscopy in patients with MELAS. J Neurol Sci. 1998;155:182–5. https://doi.org/10.1016/S0022-510X(97)00308-0.
Lee B-Y, Zhu X-H, Chen W. Elevated ATP synthase and creatine kinase activities in human visual cortex during visual stimulation: a 31P NMR magnetization transfer study at 7T. Proc. 22nd Annu. Meet. ISMRM, vol. 1, Milan; 2014, p. 12.
Lin A-L, Fox PT, Hardies J, Duong TQ, Gao J-H. Nonlinear coupling between cerebral blood flow, oxygen consumption, and ATP production in human visual cortex. Proc Natl Acad Sci. 2010;107:8446–51. https://doi.org/10.1073/pnas.0909711107.
Mochel F, N’Guyen TM, Deelchand D, Rinaldi D, Valabregue R, Wary C, et al. Abnormal response to cortical activation in early stages of Huntington disease. Mov Disord. 2012;27:907–10. https://doi.org/10.1002/mds.25009.
Murashita J, Kato T, Shioiri T, Inubushi T, Kato N. Age-dependent alteration of metabolic response to photic stimulation in the human brain measured by 31P MR-spectroscopy. Brain Res. 1999;818:72–6. https://doi.org/10.1016/S0006-8993(98)01285-2.
Murashita J, Kato T, Shioiri T, Inubushi T, Kato N. Altered brain energy metabolism in lithium-resistant bipolar disorder detected by photic stimulated 31P-MR spectroscopy. Psychol Med. 2000;30:107–15. https://doi.org/10.1017/S0033291799001439.
Nelson DL, David L, Lehninger AL, Cox MM. Lehninger principles of biochemistry. 5th ed. W.H. Freeman and Company; 2008.
Parkes LM, Fries P, Kerskens CM, Norris DG. Reduced BOLD response to periodic visual stimulation. Neuroimage. 2004;21:236–43. https://doi.org/10.1016/j.neuroimage.2003.08.025.
Qiao H, Zhang X, Zhu XH, Du F, Chen W. In vivo31P MRS of human brain at high/ultrahigh fields: a quantitative comparison of NMR detection sensitivity and spectral resolution between 4 T and 7 T. Magn Reson Imaging. 2006;24:1281–6. https://doi.org/10.1016/j.mri.2006.08.002.
Rango M, Castelli A, Scarlato G. Energetics of 3.5 s neural activation in humans: a 31P MR spectroscopy study. Magn Reson Med. 1997;38:878–83.
Rango M, Bozzali M, Prelle A, Scarlato G, Bresolin N. Brain activation in normal subjects and in patients affected by mitochondrial disease without clinical central nervous system involvement: a phosphorus magnetic resonance spectroscopy study. J Cereb Blood Flow Metab. 2001;21:85–91. https://doi.org/10.1097/00004647-200101000-00011.
Rango M, Bonifati C, Bresolin N. Parkinson’s disease and brain mitochondrial dysfunction: a functional phosphorus magnetic resonance spectroscopy study. J Cereb Blood Flow Metab. 2006;26:283–90. https://doi.org/10.1038/sj.jcbfm.9600192.
Rata M, Giles SL, DeSouza NM, Leach MO, Payne GS. Comparison of three reference methods for the measurement of intracellular pH using 31 P MRS in healthy volunteers and patients with lymphoma. NMR Biomed. 2014;27:158–62. https://doi.org/10.1002/nbm.3047.
Sappey-Marinier D, Calabrese G, Fein G, Hugg JW, Biggins C, Weiner MW. Effect of photic stimulation on human visual cortex lactate and phosphates using 1H and 31P magnetic resonance spectroscopy. J Cereb Blood Flow Metab. 1992;12:584–92. https://doi.org/10.1038/jcbfm.1992.82.
Schulz UG, Blamire AM, Davies P, Styles P, Rothwell PM. Normal cortical energy metabolism in migrainous stroke: a 31P-MR spectroscopy study. Stroke. 2009;40:3740–4. https://doi.org/10.1161/STROKEAHA.109.558163.
Shirayama Y, Yano T, Takahashi K, Takahashi S, Ogino T. In vivo31P NMR spectroscopy shows an increase in glycerophosphorylcholine concentration without alterations in mitochondrial function in the prefrontal cortex of medicated schizophrenic patients at rest. Eur J Neurosci. 2004;20:749–56. https://doi.org/10.1111/j.1460-9568.2004.03524.x.
Singh M, Kim S, Kim T-S. Correlation between BOLD-fMRI and EEG signal changes in response to visual stimulus frequency in humans. Magn Reson Med. 2003;49:108–14. https://doi.org/10.1002/mrm.10335.
Solivera J, Cerdán S, Pascual JM, Barrios L, Roda JM. Assessment of 31P-NMR analysis of phospholipid profiles for potential differential diagnosis of human cerebral tumors. NMR Biomed. 2009;22:663–74. https://doi.org/10.1002/nbm.1387.
Srinivasan R, Fornari E, Knyazeva MG, Meuli R, Maeder P. fMRI responses in medial frontal cortex that depend on the temporal frequency of visual input. Exp Brain Res. 2007;180:677–91. https://doi.org/10.1007/s00221-007-0886-3.
Stefan D, Cesare FD, Andrasescu A, Popa E, Lazariev A, Vescovo E, et al. Quantitation of magnetic resonance spectroscopy signals: the jMRUI software package. Meas Sci Technol. 2009;20:104035. https://doi.org/10.1088/0957-0233/20/10/104035.
Vafaee MS, Gjedde A. Model of blood–brain transfer of oxygen explains nonlinear flow-metabolism coupling during stimulation of visual cortex. J Cereb Blood Flow Metab. 2000;20:747–54. https://doi.org/10.1097/00004647-200004000-00012.
Vafaee MS, Meyer E, Marrett S, Paus T, Evans AC, Gjedde A. Frequency-dependent changes in cerebral metabolic rate of oxygen during activation of human visual cortex. J Cereb Blood Flow Metab. 1999;19:272–7. https://doi.org/10.1097/00004647-199903000-00005.
van de Bank BL, Maas MC, Bains LJ, Heerschap A, Scheenen TWJ. Is visual activation associated with changes in cerebral high-energy phosphate levels? Brain Struct Funct. 2018;223:2721–31. https://doi.org/10.1007/s00429-018-1656-7.
Vanhamme L, van den Boogaart A, Van Huffel S. Improved method for accurate and efficient quantification of MRS data with use of prior knowledge. J Magn Reson. 1997;129:35–43.
Wijnen JP, Scheenen TWJ, Klomp DWJ, Heerschap A. 31P magnetic resonance spectroscopic imaging with polarisation transfer of phosphomono- and diesters at 3 T in the human brain: relation with age and spatial differences. NMR Biomed. 2010;23:968–76. https://doi.org/10.1002/nbm.1523.
Zhu X-H, Chen W. Observed BOLD effects on cerebral metabolite resonances in human visual cortex during visual stimulation: a functional 1H MRS study at 4 T. Magn Reson Med. 2001;46:841–7. https://doi.org/10.1002/mrm.1267.
Zuo CS, Lin P, Vitaliano G, Wang K, Villafuerte R, Lukas SE. A preliminary evaluation of the correlation between regional energy phosphates and resting state functional connectivity in depression. NeuroImage Clin. 2015;9:348–54. https://doi.org/10.1016/j.nicl.2015.08.020.
Acknowledgments
This work was supported by São Paulo Research Foundation (FAPESP, Brazil) (grant numbers 2005/56578-4, 2009/00270-2, 2009/10046-2, 2011/01106-1, 2013/07559-3), National Council of Scientific and Technological Development (CNPq, Brazil) (grant number 500148/2011-2), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, Brazil) (Finance Code 001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The project was approved by the Ethics Committee of University of Campinas (CAAE 0568.0.146.000–09), and written informed consent was obtained from all subjects, prior to data acquisition.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Silva Costa, T.B., Landim, R.C.G., Barreto, F.R. et al. Investigation of possible high-energy phosphate concentration changes in the visual cortex for different stimulus frequencies. Res. Biomed. Eng. 35, 203–211 (2019). https://doi.org/10.1007/s42600-019-00023-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42600-019-00023-0