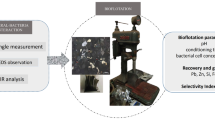
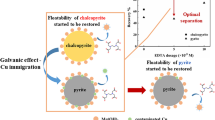
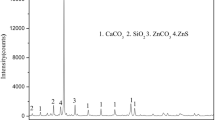
Abstract
Arsenic contamination significantly affects the quality of copper concentrates, presenting challenges during smelting processes. Considering that covellite and enargite have similar surface properties, they are difficult to separate by flotation, resulting in copper concentrates with excessive levels of arsenic. To address this issue, this study systematically investigated the flotation separation of covellite and enargite in microflotation and bench-scale flotation tests using calcium lignosulfonate as a depressant, with the results indicating that calcium lignosulfonate can be used to separate covellite and enargite because it enables the separation of arsenic-bearing copper minerals into high- and low-arsenic (As 0.57%) copper concentrates. According to contact angle and adsorption tests, scanning electron microscopy–energy dispersive spectroscopy, and X-ray photoelectron spectroscopy, the differential adsorption of calcium lignosulfonate onto covellite and enargite is the primary reason for their distinct flotation behaviors in the presence of calcium lignosulfonate. The selective depressive effect of calcium lignosulfonate provides a novel approach for improving the flotation separation of arsenic-bearing minerals from copper sulfide minerals.










Similar content being viewed by others
Data Availability
Date will be made available on request.
References
Filippou D, St-Germain P, Grammatikopoulos T (2013) Recovery of metal values from copper-arsenic minerals and other related resources. Miner Process Extr Metall Rev 28(4):247–298. https://doi.org/10.1080/08827500601013009
Plackowski C, Nguyen AV, Bruckard WJ (2012) A critical review of surface properties and selective flotation of enargite in sulphide systems. Miner Eng 30:1–11. https://doi.org/10.1016/j.mineng.2012.01.014
Tayebi-Khorami M, Manlapig E, Forbes E (2017) Selective flotation of enargite from copper sulphides in Tampakan deposit. Miner Eng 112:1–10. https://doi.org/10.1016/j.mineng.2017.06.021
Long G, Peng YG, Bradshaw D (2012) A review of copper–arsenic mineral removal from copper concentrates. Miner Eng 36:179–186. https://doi.org/10.1016/j.mineng.2012.03.032
Safarzadeh MS, Moats MS, Miller JD (2014) Recent trends in the processing of enargite concentrates. Miner Process Extr Metall 35:283–367. https://doi.org/10.1080/08827508.2012.723651
Ma X, Bruckard WJ (2009) Rejection of arsenic minerals in sulfide flotation – a literature review. Int J Miner Process 93:89–94. https://doi.org/10.1016/j.minpro.2009.07.003
Senior GD, Guy PJ, Bruckard WJ (2006) The selective flotation of enargite from other copper minerals—a single mineral study in relation to beneficiation of the Tampakan deposit in the Philippines. Int J Miner Process 81(1):15–26. https://doi.org/10.1016/j.minpro.2006.06.001
Guo H, Yen WT(2008) Electrochemical study of synthetic and natural enargites. In: Proceedings of [the] 24th International Mineral Processing Congress, Beijing, China, Sept. 24–28, vol. 1, pp. 1138–1145
Plackowski C, Hampton MA, Nguyen AV, Bruckard WJ (2013) Fundamental studies of electrochemically controlled surface oxidation and hydrophobicity of natural enargite. Langmuir 29(7):2371–2386. https://doi.org/10.1021/la3043654
Schwartz DM, Omaynikova VY, Stocker SK(2017) Environmental benefits of the CESL process for the treatment of high-arsenic copper concentrates, in: Hydroprocess-ICMSE In: Presented at the 9th International Seminar on Process Hydrometallury-International Conference on Metal Solvent Extraction, Gecamin Digital Publications, Santiago, Chile
Smith LK, Bruckard WJ (2008) The separation of arsenic from copper in a Northparkes copper-gold ore using controlled-potential flotation. Int J Miner Process 84:15–24. https://doi.org/10.1016/j.minpro.2007.05.002
Pauporté T, Schuhmann D (1996) An electrochemical study of natural enargite under conditions relating to those used in flotation of sulphide minerals. Colloids Surf A 111(1–2):1–19. https://doi.org/10.1016/0927-7757(95)03498-6
Asbjornsson J, Kelsall GH, Pattrick RAD, Vaughan DJ, Wincott PL, Hope GA (2004) Electrochemical and surface analytical studies of enargite in acid solution. J Electrochem Soc 151(7):E250–E256
Guo H, Yen WT (2008) Electrochemical study of synthetic and natural enargites. Proc Int Miner Process Congr 24(1):1138–1145
Yepsen R, Gutierrez L (2020) Effect of Eh and pH on the flotation of enargite using seawater. Miner Eng 159(7):106612. https://doi.org/10.1016/j.mineng.2020.106612
Menacho JM, Aliaga W, Valenzuela R, Ramos V, Olivares I (1993) Selective flotation of enargite and chalcopyrite. Minerales 48:0026-458X
Plackowski C, Hampton MA, Bruckard WJ, Nguyen AV (2014) An XPS investigation of surface species formed by electrochemically induced surface oxidation of enargite in the oxidative potential range. Miner Eng 55(1):60–74. https://doi.org/10.1016/j.mineng.2013.08.010
Plackowski C, Bruckard WJ, Nguyen AV (2014) Surface characterisation, collector adsorption and flotation response of enargite in a redox potential controlled environment. Miner Eng 65:61–73. https://doi.org/10.1016/j.mineng.2014.05.013
Sasaki K, Takatsugi K, Ishikura K, Hirajima T (2010) Spectroscopic study on oxidative dissolution of chalcopyrite, enargite and tennantite at different pH values. Hydrometallurgy 100(3–4):144–151. https://doi.org/10.1016/j.hydromet.2009.11.007
Suyantara GPW, Hirajima T, Miki H, Sasaki K, Kuroiwa S, Aoki Y (2020) Effect of H2O2 and potassium amyl xanthate on separation of enargite and tennantite from chalcopyrite and bornite using flotation. Miner Eng 152:106371. https://doi.org/10.1016/j.mineng.2014.05.013
Suyantara GPW, Berdakh D, Miki H (2023) Effect of hydrogen peroxide on selective flotation of chalcocite and enargite. Int J Min Sci Technol 3(6):703–716. https://doi.org/10.1016/j.ijmst.2023.01.002
Gan YG, Deng RD, Liu QJ (2022) Surface characteristics, collector adsorption, and flotation response of covellite in oxidizing environment. Trans Nonferrous Met Soc China 32(2):657–667. https://doi.org/10.1016/S1003-6326(22)65823-0
Asbjornsson J, Kelsall GH, Pattrick RAD, Vaughan DJ, Wincott PL, Hope GA (2004) Electrochemical and surface analytical studies of enargite in acid solution. J Electrochem Soc 151(7):E250–E256
Suyantara GPW, Hirajima T, Miki H, Sasaki K, Kuroiwa S, Aoki Y (2021) Effect of Na2SO3 on the floatability of chalcopyrite and enargite. Miner Eng 173:107222. https://doi.org/10.1016/j.mineng.2021.107222
Tajadod J (1997) Flotation chemistry of enargite and chalcopyrite using potassium amyl xanthate and depressants (PhD Thesis). Queen’s University, Kingston, Ontario, Canada
Tapley B, Yan D (2003) The selective flotation of arsenopyrite from pyrite. Miner Eng 16(11):1217–1220. https://doi.org/10.1016/j.mineng.2003.07.017
Filippou D, St-Germain P, Grammatikopoulos T (2007) Recovery of metal values from copper—arsenic minerals and other related resources. Miner Process Extr Metall 28:247–298. https://doi.org/10.1080/08827500601013009
Fornasiero D, Fullston D, Li C, Ralston J (2001) Separation of enargite and tennantite from non-arsenic copper sulfide minerals by selective oxidation or dissolution. Int J Miner Process 61(2):109–119. https://doi.org/10.1016/S0301-7516(00)00029-6
Gan YG, Deng RD, Liu QJ (2022) Flotation separation of covellite and enargite via oxidation treatment. Minerals 12(08):970. https://doi.org/10.3390/min12080970
Falconi IBA, Junior ABB, Baltazar MPG, Espinosa DCR, Tenório JAS (2023) An overview of treatment techniques to remove ore flotation reagents from mining wastewater. J Environ Chem Eng 11(6):111270. https://doi.org/10.1016/j.jece.2023.111270
Deng RD, Yang XF, Hu Y, Ku JG, Zuo WR, Ma YQ (2018) Effect of Fe(II) as assistant depressant on flotation separation of scheelite from calcite. Miner Eng 118:133–140. https://doi.org/10.1016/j.mineng.2017.12.017
Nowak P, Laajalehto K (2000) Oxidation of galena surface–an XPS study of the formation of sulfoxy species. Appl Surf Sci 157:101–111. https://doi.org/10.1016/S0169-4332(99)00575-9
Wu DD, Wen SM, Deng JS, Liu J, Mao YB (2015) Study on the sulfidation behavior of smithsonite. Appl Surf Sci 329:315–320. https://doi.org/10.1016/j.apsusc.2014.12.167
Gao WZ (2022) Mechanism of calcium lignosulfonate in apatite and dolomite flotation system. Int J Miner Metall Mater 29(9):1697–1704
McIntyre NS, Zetaruk DG (1977) X-ray photoelectron spectroscopic studies of iron oxides. Anal Chem 49:1521–1529
Funding
This work was financially supported by the National Natural Science Foundation of China (Grant No. 51804080).
Author information
Authors and Affiliations
Contributions
RL: investigation, data curation, validation, visualization, investigation, and writing—original draft. QL: formal analysis, investigation, writing (review and editing), and visualization. RD: formal analysis, writing (review and editing), and funding acquisition.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, R., Liu, Q. & Deng, R. Calcium Lignosulfonate as a Depressant for Enhancing Flotation Separation of Covellite and Enargite. Mining, Metallurgy & Exploration 41, 1135–1144 (2024). https://doi.org/10.1007/s42461-024-00936-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42461-024-00936-0