Abstract
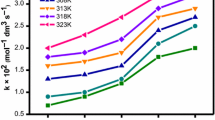
2-(Hydroxyethyl)ethylenediaminetriacetatoiron(III) ion reduction with thiocarbonate (CO2S2−) in the presence and absence of sodium dodecyl sulfate (SDS) and cetyltrimethylammonium bromide (CTAB) micelles at 33 ± 1 °C, µ = 0.1 C2 M and maximum absorption = 490 nm has been investigated spectrophotometrically. The redox reaction follows pseudo-first-order kinetics which resulted to first order with respect to concentration of the both redox species with a correlation coefficient R2 = 0.944 and R2 = 0.9753, respectively for the complex and thiocarbonate ion. The rate of reaction accelerated and decelerated with a change in salt concentration (KNO3) and solvent polarity (water/acetone mixture), respectively. The reaction is catalysed and inhibited by the positive electrostatic charged (CTAB) and the negative electrostatic charged (SDS) surfactants, respectively. Catalysis results are explained following the hydrophobic and electrostatic interactions between micelle aggregates and redox species, the binding constant of the complex and the thiocarbonate ion of the two micelles, and the Berezin procedure. Approximately 1.935 M−1 s−1, 0.9686 M−1 s−1, 54.619 M−1 s−1 and a total of 0.00 M−1 s−1 are obtained for the complex/CTAB, thiocarbonate ion/CTAB, complex/SDS and binding thiocarbonate ion/SDS constants, respectively. The micellar effect on this bimolecular reaction is treated in a way similar to the enzymatic catalysis where in the model assumes association of the reactants (S) to the micellar molecular (Q) as shown below;








Similar content being viewed by others
References
Berezin IV, Martinek K, Yatsimirskii AK (1973) Physicochemical foundations of micellar catalysis. Russ Chem Rev 42(10):787–802
Malik AR, Dileep K (2019) Interaction of ninhydrin with zinc(II) complex of tryptophan in the three dicationic gemini surfactants. Colloid Polym Sci 297(11–12):1519–1527. https://doi.org/10.1007/s00396-019-04569-4
Dileep K, Malik AR (2019) Study of zinc-glycylglycine complex with ninhydrin in aqueous and cationic micellar media: a spectrophotometric technique. Tenside Surfactants Deterg 56(4):312–318
Rub MA, Kumar D (2019) Influence of cationic cetyltrimethylammonium bromide on rate of Zn(II)-histidine complex and ninhydrin. J Oleo Sci 68:1231–1240
Gustavsson H, Lindman B (1975) Nuclear magnetic resonance studies of the interaction between alkali ions and micellar aggregates. J Am Chem 97(14):3923–3930. https://doi.org/10.1021/ja00847a009
Fox KK, Robb ID, Smith R (1972) Solubilisation of p-xylene in sodium dodecy sulphate micelles. J Chem Soc Faraday Transit 1(68):445–449. https://doi.org/10.1039/F19726800445
Oakes J (1973) Magnetic resonance studies in aqueous systems. Part 3. Electron spin and nuclear magnetic relaxation study of interactions between manganese ions and micelles. J Chem Soc Faraday Trans 69:1321–1329. https://doi.org/10.1039/F29736901321
Atanu R, Suman C, Animesh A, Indukamal D, Kuheli D, Satyajit B, Shuvendu SB, Bidyut S (2020) Hetero-aromatic n-base-promoted oxidation of 4-chlorobenzyl alcohol by Cr(VI) in micellar media. Res Chem Intermed 46(2559):1–20. https://doi.org/10.1007/s11164-020-04106-x
Suman C, Atanu R, Animesh A, Kalachand M, Bidyut S (2020) Surface phenomenon in micellar media: an excellent controlling factor for oxidation of fatty aldehyde in aqueous medium. J Mol Liq 310(113224):1–10. https://doi.org/10.1016/j.molliq.2020.113224
Pintu S, Aniruddha G, Alessandro S, Bidyut S (2019) Surfactant for better tomorrow: applied aspect of surfactant aggregates from laboratory to industry. Res Chem Intermed 45(6021):1–21. https://doi.org/10.1007/s11164-019-04017-6
Bunton CA, Carrasco N, Huang SK, Paik CH, Romsted LS (1978) Reagent distribution and micellar catalysis of carbocation reactions. J Am Chem Soc 100:5420–5425
Singh M (2014) Kinetic treatment of the reaction of fructose and n-bromosuccinimide in cationic/anionic/nonionic micelles. J Soft Matter. https://doi.org/10.1155/2014/791563
Pintu S, Bidgut S (2020) Potential application of micellar nanoreactor for electron transfer reactions mediated by a variety of oxidants: a review. Adv Colloid Interface Sci 284(102241):1–14. https://doi.org/10.1016/j.cis.2020.102241
Soriyan OO, Owoyomi O (2008) Periodate oxidation of dicyano-bis-(1,10-phenanthroline) iron(II) dihydrate in aqueous sodium dodecyl sulphate (SDS). Transit Met Chem 33:121–126. https://doi.org/10.1007/s11243-007-9036-9
Raducan A, Olteanu A, Puiu M, Oancea D (2008) Influence of surfactants on the fading of malachite green. Cent Eur J Chem 6(1):89–92. https://doi.org/10.2478/s11532-007-0066-0
Balakumar S, Thanasekaram P, Rajkumar E, Adaikalasamy KJ, Rajagopal S, Ramaraj R (2006) Micellar catalysis on the electron transfer reactions of iron(III)-polypyridyl complexes with organic sulfides—importance of hydrophobic interactions. J R Soc Chem 2:352–358
Laguta AN, Eltsov SV, Mchedlov-Petrossyan NO (2018) Kinetics of alkaline fading of methyl violet in micellar solutions of surfactants: comparing Piszkiewicz’s, Berezin’s, and pseudophase ion-exchange models. Int J Chem Kinet. https://doi.org/10.1002/kin.21231
Esan OS (2014) Effect of micellar aggregate on the kinetics and mechanism of the reaction between ethylene glycol and periodate. Int Sch Res Not 2014:1–3
Ogunlusi GO, Oyetunji OA, Owoyomi O, Ige J (2016) Effects of alkyltrimethylammonium bromide surfactants on the kinetics of the oxidation of tris(1,10-phenanthroline)iron(II) by azidopentacyanocobaltate(III) complex. J Dispers Sci Technol. https://doi.org/10.1080/01932691.2016.1225218
Schneppensieper T, Seibig S, Zahl A, Tregloan P, Eldik RV (2001) Influence of chelate effects on the water-exchange mechanism of polyaminecarboxylate complexes of iron(III). Inorg Chem 40(15):3670–3676
Wubs HJ, Beenackers AACM (1994) Kinetics of H2S absorption into aqueous ferric solutions of EDTA and HEDTA. AlChE J 40(3):433–444
Nkole IU, Osunkwo CR, Onu AD, Idris SO (2018) Kinetics and mechanism of the reduction of n-(2-hydroxyethyl)ethylenediaminetriacetateiron(III) complex by thioglycol in bicarbonate buffer medium. Int J Adv Chem 6(1):102–107. https://doi.org/10.14419/ijac.v6i1.10902
Abdulsalam S, Idirs SO, Shallangwa GA, Onu AO (2020) Reaction of n,n1-phenylenebis(salicyalideneiminato)cobalt(III) and l-cysteine in mixed aqueous medium: kinetics and mechanism. Heliyon 6(e3050):1–8. https://doi.org/10.1016/j.heliyon.2020.e03850
Arthur DE, Nkole IU, Osunkwo CR (2020) Electron transfer reaction of tris-(1,10-phenanthroline)cobalt(III) complex and iodide ion in an aqueous acidic medium. Chem Afr. https://doi.org/10.1007/s42250-020-00201-z
Feigl F (1966) Spot tests in organic analysis. Elsevier Publishing Company, London, pp 540–545
Nkole IU, Onu AD, Osunkwo CR, Idris SO (2018) Kinetic approach to the mechanism of the redox reaction of ethylenediaminetetraacetatoferrate(III) complex and thioglycolic acid in bicarbonate buffer medium. ATBU J Sci Technol Educ (JOSTE) 6(4):6–19
Ghosh GK, Misra K, Baskim M, Linert W, Moi SC (2013) Kinetics and mechanism of the interaction of di-μ-hydroxo-bis(1,10-phenanthroline)dipalladium(II) perchlorate with thioglycolic acid and glutathione in aqueous solution. J Solut Chem 42:526–543
Astray G, Cid A, Manso JA, Mejuto JC, Moldes O, Morales J (2011) Influence of anionic and nonionic micelles upon hydrolysis of 3-hydroxy-carbofuran. Int J Chem Kinet 43:402–408
Sar P, Ghosh A, Susanta M, Dhiman R, Bijan D, Saha B (2016) Selective heteroaromatic nitrogen base promoted chromium(VI) oxidation of isomeric pentanols in aqueous micellar media at room temperature. J Ind Eng Chem 42:53–62. https://doi.org/10.1016/j.jiec.2016.07.028
Sar P, Ghosh A, Saha R, Saha B (2014) Micellar effect on pentavalent vanadium oxidation of methanol to formaldehyde to formic acid in aqueous acid media at room temperature. Res Chem Intermed. https://doi.org/10.1007/s11164-014-1635-4
Sar P, Saha B (2020) Potential application of micellar nanoreactor for electron transfer reactions mediated by a variety of oxidants: a review. Adv Colloid Interface Sci 284(102241):1–14. https://doi.org/10.1016/j.cis.2020.102241
Funding
This study did not receive any specific grant from funding agencies in the public, commercial or private sector.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Nkole, I.U., Abdulsalam, S., Ibrahim, I. et al. Micellar Effect on Electron Transfer Reaction of 2-(hydroxyethyl)ethylenediaminetriacetatoiron(III) Complex with Thiocarbonate Ion: Kinetic Model. Chemistry Africa 4, 525–533 (2021). https://doi.org/10.1007/s42250-021-00241-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42250-021-00241-z