Abstract
The kinetics and mechanistic study of the electron transfer of tris(1,10-phenanthroline)cobalt(III) complex and iodide ion has been carried out in an aqueous acidic medium at ionic strength(I) = 0.20 mol dm−3 (NaCl), [H+] = 0.02 mol dm−3, T = 28 ± 1.0 °C and λmax of 495 nm. The rate of reaction was found to be third order overall: first order in the oxidant and second order in the reductant, and acid independent. The empirical rate law conforms to the equation:
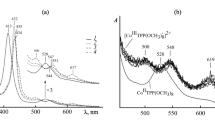
The reaction rate was unaffected by change in ionic strength and dielectric constant of the reaction medium. Added cations and anions catalysed and inhibited the reaction rate respectively. Based on spectroscopic investigation and kinetic pieces of evidence from the Michaelis–Menten plot and effect of added ions, an outer sphere mechanism has been rationalised for the reaction.






Similar content being viewed by others
References
Adman ET (1979) A comparison of the structures of electron transfer proteins. Biochem Biophys Acta 549:107–144
Bennet LE (1972) Metalloprotein redox reactions. Prog Inorg Chem 18:1–8
Prince RC, George GN (1990) Tryptophan radicals. Trends Biochem Sci 15:170–172
Santosh KU (2006) Chemical kinetics and reaction dynamics. Springer, New York with Anamaya publishers, New Delhi, India, pp. 10-110
Asperger S (2003) Chemical kinetics and inorganic reaction mechanism, 2nd edn. Springer Science + Business Media LLC, New York p, p 3
Taube H (1983) Electron transfer between metal complexes—retrospective. Stanford University, Department of Chemistry, Stanford, p 94305
Fierro S, Comninellis C, Einaga Y (2012) Simultaneous detection of iodine and iodide on boron doped diamond electrodes. Talanta 103:33–37
Küpper FC, Carpenter LJ, McFiggans GB (2008) Iodide accumulation provides kelp with an inorganic antioxidant impacting atmospheric chemistry. Proc Natl Acad Sci USA 105:6954–6958
Hubbard CD, Gerhard A, Rudivan E (2001) Electrostriction and counter ion effects on an outer-sphere electron transfer reaction, kinetics of the reduction of hexachloroiridate(IV) by iodide ion. J Chem Soc Dalton Trans. 1069–1075
Nkole IU, Osunkwo CR (2019) Kinetic approach to the reduction of ethylene- diaminetetraacetatoferrate(III) complex by iodide ion in aqueous acidic medium. Asian J Phys Chem Sci 7(2):1–8
Pook NP, Hentrich P, Gjikaj M (2015) Crystal structure of bis[tris(1,10-phenanthroline)cobalt(II)] tetranitrate N, N’–(1,4-phenylenedicarbonyl) diglycine solvate octahydrate. Acta Cryst E71:910–914
Sammes PG, Yahioglu GY (1994) 1,10-Phenanthroline: a versatile ligand. Chem Soc Rev 23(5):327–334
Gaëlle DS, Yufanyi DM, Jagan R, Agwara MO (2016) Synthesis, characterisation and antimicrobial properties of cobalt(II) and cobalt(III) complexes derived from 1,10-phenanthroline with nitrate and azide Co-ligands. Cogent Chemistry 2:125–201
Nkole IU, Onu AD, Osunkwo CR, Idris SO (2018) Kinetic approach to the mechanism of the redox reaction of ethylenediaminetetraacetatoferrate(III) complex and thioglycolic acid in bicarbonate buffer medium. ATBU J Sci Technol Edu (JOSTE) 6(4):6–19
Nkole IU, Osunkwo CR, Onu AD, Idris SO (2018) Kinetics and mechanism of the reduction of n-(2-hydroxyethyl)ethylenediaminetriacetatoiron(III) complex by thioglycol in bicarbonate buffer medium. Int J Adv Chem 6(1):102–107
Hahn RB, Welcher FJ (1963) Inorganic qualitative analysis: a short course for introductory chemistry. J Chem Educ 40(8):442
Inorganic Chemistry/Qualitative Analysis/Tests for anions. (2020). Wikibooks, The Free Textbook Project. Retrieved 16:55, September 25, 2020 from https://en.wikibooks.org/w/index.php?title=Inorganic_Chemistry/Qualitative_Analysis/Tests_for_anions&oldid=3692292
Osunkwo CR, Nkole IU, Onu AD, Idris SO (2018a) Electron transfer reaction of tris-(1,10-phenanthroline)cobalt(III) complex [Co(phen)3]3+ and thiosulphate ion (S2O32−) in an aqueous acidic medium. Int J Adv Chem 6(1):121–126
Osunkwo CR, Nkole IU, Onu AD, Idris SO (2018b) Kinetics and the mechanism of the reduction of tris-(1,10-Phenanthroline)cobalt(III) complex by n-methylthiourea in an aqueous acidic medium. Nigerian Res J Chem Sci 5:82–97
Nord G, Pedersen B, Farver O (1978) Outer-sphere oxidation of iodide and thiocynate by tris(2,2’-bipyridyl)- and tris(1, 10-phenanthroline) osmium(III) in aqueous solutions. Inorg Chem 17:2233–2238
Gordon FG (1997) The kinetics and mechanism of the chlorine dioxide – iodide ion reaction. Inorg Chem 36:2494–2497
Hubbard CD, Gerhard A, Rudivan E (2001) Electrostriction and counter ion effects on an outer-sphere electron transfer reaction, kinetics of the reduction of hexachloroiridate(IV) by iodide ion. J Chem Soc Dalton Trans 2001:1069–1075
Bauer J, Tomisic V, Vrldjan PB (2008) Kinetics and mechanism of iodide oxidation by iron(III): a clock reaction approach. J Chem Educ 85(8):1123–1125
Srinivas B, Reddy RM, Chandra AV (2007) The anation kinetics of cis[Co(en)2(H2O)2](NO3)3 by anthranilic acid in aqueous medium. Asian J Chem 19(4):2586–2594
Sasikala K, Arunachalam S (2010) Studies on outer-sphere electron transfer reactions of some surfactant-cobalt(III) complexes with ferrocyanide anion. Colloid J 72(4):530–537
Ige J, Ojo FJ, Olubuyide O (1979) Kinetics and mechanism of tris-(1,10-phenanthroline)iron(II) by chlorine and bromine and of the reduction of tris-(1,10-phenanthroline)iron(III) by iodide ions. Can J Chem 57(15):2065–2070
Sharpe AG (1981) Inorganic chemistry, 1st edn. Longman, London, p 518
Babatunde OA, Ajayi JO (2013) Kinetic approach to the reduction of toluidine blue by dithionate ion in aqueous acidic medium. Global J Sci Front Res Chem 13(8):20–30
Onu AD, Iyun JF, Idris SO (2016) Oxidation of ethylenediaminetetraacetato-cobaltate(II) complex by hydrogen peroxide in aqueous acidic medium: a kinetic study. J Chem Soc Nigeria 41(2):81–86
Osunlaja AA, Idris SO, Iyun JF (2013) Kinetics and mechanism of thiourea oxidation by oxygenated [Co2(O2)(NH3)10]5+ complex. J Chem Pharm Res 5(2):328–336
Ukoha PO, Atiga S, Ujam TO, Asegbeloyin JN, Okpareke CO, Okereke SO (2015) Kinetics and mechanism of electron transfer reaction of an adipato bridged iron(III)-Salen complex with dithionite ion in perchloric acid medium. Croat Chem Acta 88(3):259–266
Housecroft CE, Sharpe AG (2005) Inorganic chemistry third edition. Pearson Education Limited England, 802–803
Ludovic TG, Wesley BS, Gerald JM (2019) Iodide photoredox and bond formation chemistry. Acc Chem Res 52:170–179
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arthur, D.E., Nkole, I.U. & Osunkwo, C.R. Electron Transfer Reaction of Tris(1,10-phenanthroline)cobalt(III) Complex and Iodide Ion in An Aqueous Acidic Medium. Chemistry Africa 4, 63–69 (2021). https://doi.org/10.1007/s42250-020-00201-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42250-020-00201-z