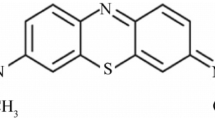
Abstract
The bio-adsorbents can provide an eco-friendly and low-cost solution to treat effluent water from the textile industry. The pH of the textile industrial effluent water varies from 4 to 14. Therefore, it is pertinent to investigate the effect of pH on bio-adsorption efficiency and understand the materialistic reasons driving bio-adsorption. We hypothesise that to develop an understanding of the observed pH dependence on bio-adsorption, knowledge of morphological and chemical features of the adsorbent is critical. This aspect prompted us to study the role of underlying major materialistic factors – functional group and morphology, on bio-adsorption at different pH levels. With this objective, we performed experiments on three different bio-adsorbents – viz. banana peels, orange peels, and neem leaves remove a cationic dye Rhodamine B from water. The dye removal efficiency was quantified using a UV–Vis spectrophotometer. The critical role of functional groups and morphology on the activity of the bio-adsorbents were investigated – utilising Fourier transform infrared spectroscopy and scanning electron microscopy techniques. All the bio-adsorbents exhibited significant removal efficiency. The removal efficiency is understood based on the protonation of functional groups and the dynamics of zwitterion formation. The derived connection between the chemical features of the adsorbent and the pH of the contaminant solution will be helpful in better adoption of bio-adsorbents in treating effluent water from the textile industry.











Similar content being viewed by others
References
H. Xu, S. Ouyang, L. Liu, P. Reunchan, N. Umezawa, J. Ye, Recent advances in TiO 2-based photocatalysis. J. Mater. Chem. A 2(32), 12642 (2014)
A. Lee, J.W. Elamb, S.B. Darling, Membrane materials for water purification: design, development, and application. Environ. Sci. Water Res. Technol. 2, 17–42 (2016)
S.F. Soares, T.R. Simoes, T. Trindade, A.L. Daniel-da-Silva, Highly efficient removal of dye from water using magnetic carrageenan/silica hybrid nano-adsorbents. Water Air Soil Pollut. 87, 228–239 (2017)
L. Liu, Z.Y. Gao, X.P. Su, X. Chen, L. Jiang, J.M. Yao, Adsorption removal of dyes from single and binary solutions using a cellulose-based bio adsorbent. ACS Sustain. Chem. Eng. 3, 432–442 (2015)
C.S.D. Rodrigues, L.M. Madeira, R.A.R. Boaventura, Decontamination of an industrial cotton dyeing wastewater by chemical and biological processes. Ind. Eng. Chem. 53, 2412–2421 (2014)
R.S.D. Castro, L. Caetano, G. Ferreira, P.M. Padilha, M.J. Saeki, L.F. Zara, M.A.U. Martines, G.R. Castro, Banana peel applied to the solid phase extraction of copper and lead from river water: preconcentration of metal ions with a fruit waste. Ind. Eng. Chem. 50, 3446–3451 (2011)
M.I. Khaskheli, S.Q. Memon, A.N. Siyal, M.Y. Khuhawar, Use of orange peel waste for arsenic remediation of drinking water. Waste Biomass Valor. 2, 423–433 (2011)
M.N. Zafar, A. Parveen, R. Nadeem, A pretreated green biosorbent based on neem leaves biomass for the removal of lead from wastewater. Desalin. Water Treat. 51, 4459–4466 (2013)
P. Brown, L.A. Jefcoat, D. Parrish, S. Gill, E. Graham, Evaluation of the adsorptive capacity of peanut hull pellets for heavy metals in solution. Adv. Environ. Res. 4, 19–29 (2000)
P. Ricou-Hoeffer, I. Lecuyer, P.L. Cloirec, Experimental design methodology applied to adsorption of metallic ions onto fly ash. Water Res. 35, 965–976 (2001)
A.C. Texier, Y. Andres, P.L. Cloirec, Selective biosorption of lanthanide (La, Eu, Yb) ions by Pseudomonas aeruginosa. Environ. Sci. Technol. 33, 489–495 (1999)
W.E. Marshall, L.H. Wartelle, D.E. Boler, C.A. Toles, Metal ion adsorption by soybean hulls modified with citric acid: a comparative study. Environ. Technol. 21, 601–607 (2000)
Z. Reddad, C. Gerente, Y. Andres, P.L. Cloirec, Adsorption of several metal ions onto a low-cost biosorbent: kinetic and equilibrium studies. Environ. Sci. Technol. 36, 2067–2073 (2002)
M.H. El-Naas, S.A. Al-Muhtaseb, S. Makhlouf, Biodegradation of phenol by Pseudomonas putida immobilised in polyvinyl alcohol (PVA) gel. J. Hazard. Mater. 164, 720–725 (2009)
Y.C. Sharma, Uma, Optimisation of parameters for adsorption of methylene blue on a low-cost activated carbon. J. Chem. Eng. 55, 435–439 (2010)
H. Li, L. Liu, J. Cui, J. Cui, F. Wang, F. Zhang, High-efficiency adsorption and regeneration of methylene blue and aniline onto activated carbon from waste edible fungus residue and its possible mechanism. RSC Adv. 10, 14262 (2020)
X. Wei, Y. Wang, Y. Feng, X. Xie, X. Li, S. Yang, Different adsorption-degradation behaviour of methylene blue and Congo red in nanoceria/H2O2 system under alkaline conditions. Sci. Rep. 9, 4964 (2019)
S.B. Daffalla, H. Mukhtar, M.S. Shaharun, Characterisation of adsorbent developed from rice husk: effect of surface functional group on phenol adsorption. J. Appl. Sci. 10, 1060–1067 (2010)
R. Mallampati, L. Xuanjun, A. Adin, S. Valiyaveettil, Fruit peels as efficient renewable adsorbents for removal of dissolved heavy metals and dyes from water. ACS Sustain. Chem. Eng. 3(6), 1117–1124 (2015)
M.T. Uddin, M.A. Rahman, M. Rukanuzzaman, M.A. Islam, A potential low cost adsorbent for the removal of cationic dyes from aqueous solutions. Appl. Water Sci. 7, 2831–2842 (2017)
S. Dey, A. Islam, A review on textile wastewater characterization in Bangladesh. Resour. Environ. 5(1), 15–44 (2015)
G. Annadurai, R.S. Juang, D.J. Lee, Adsorption of heavy metals from water using banana and orange peels. Water Sci. Technol. 47, 185–190 (2002)
R.A. Kusumadewi, A. Wijayanti, R. Hadisoebroto, Utilisation of banana peel and water hyacinth leaves as adsorbent for removal of copper from wastewater. Int. J. Sci. Technol. 8, 2529–2534 (2019)
M. Arami, N.Y. Limaee, N.M. Mahmoodi, N.S. Tabrizi, Removal of dyes from colored textile wastewater by orange peel adsorbent: equilibrium and kinetic studies. J. Colloid Interface Sci. 288, 371–376 (2005)
I. Khatod, Removal of methylene blue dye from aqueous solutions by neem leaf and orange peel powder. Int. J. Chem. Tech. Res. 5, 572–577 (2013)
M.S. Mahmoud, Banana peels as an eco-sorbent for manganese ions. Int. J. Agric. Biol. Eng. 8, 1201–1207 (2014)
A. Ahmad, Z.A. Ghazi, M. Saeed, M. Ilyas, R. Ahmad, A.M. Khattak, A. Iqbal, A comparative study of the removal of Cr (VI) from synthetic solution using natural biosorbents. New J. Chem. 41, 10799–10807 (2017)
C.M. Santos, J. Dweck, R.S. Viotto, A.H. Rosa, L.C. de Morais, Application of orange peel waste in the production of solid biofuels and biosorbents. Bioresour. Technol. 196, 469–479 (2015)
M. Thirumavalavan, Y. Lai, J. Lee, Fourier transform infrared spectroscopic analysis of fruit peels before and after the adsorption of heavy metal ions from aqueous solution. J. Chem. Eng. 56, 2249–2255 (2011)
A. Thakur, H. Kaur, Response surface optimisation of Rhodamine B dye removal using paper industry waste as adsorbent. Int. J. Ind. Chem. 8, 175–186 (2017)
Funding
This work was supported by Kerala State Council for Science, Technology & Environment–SRS. Project 107/2016. BT gratefully acknowledges UGC-BSR SAP for fellowship assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Thomas, B., Shilpa, E.P. & Alexander, L.K. Role of functional groups and morphology on the pH-dependent adsorption of a cationic dye using banana peel, orange peel, and neem leaf bio-adsorbents. emergent mater. 4, 1479–1487 (2021). https://doi.org/10.1007/s42247-021-00237-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42247-021-00237-y