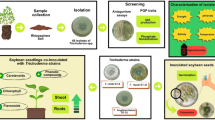
Abstract
In the present study, we explored the disease-control efficacy of a powder preparation of the widely available evergreen medicinal shrub Dodonaea viscosa (L.) Jacquin against the bacterial wilt (BW) of tomato caused by R. solanacearum. Both in-vitro and in-planta experiments were conducted, evaluating different concentrations of D. viscosa dried powder against the BW pathogen at different time interval. Studies revealed that all tested concentrations of the product displayed significantly larger zones of the pathogen inhibition (ZIs) as compared to control treatments. D. viscosa methanol and water extracts at 15% produced 26.66 mm and 26.00 mm ZIs, respectively whereas the same products at 10% produced both 22 mm ZIs. Further, in-vitro inhibition tests were carried out using HPLC fractions. Results evidenced that fraction six produced 23 mm ZI followed by fraction five and eight that displayed 21.66 and 20.00 mm ZIs, respectively. Likewise, the longest degradation time (20 Days Before Transplanting, DBT) coupled with the highest tested dose (30 g powder preparation) produced higher activity respect to the control treatments, improving stem length, root length and fresh plant biomass by 102.3%, 74.3% and 105.8%, respectively. Likewise, the highest dose of D. viscosa powder thoroughly mixed with potted soil 10 days DBT significantly enhanced plant growth parameters such as stem length (+ 88.3%), root length (+ 87.1%) and fresh biomass (+ 96.8%) as compared to control. Our results suggest that 30 g dried powder of D. viscosa mixed well with soil artificially contaminated with the pathogen 20 DBT could be used as an effective tool in integrated disease management against BW of tomato.




Similar content being viewed by others
Data Availability
Data will be made available on request.
Abbreviations
- BW:
-
Bacterial wilt
- ZI:
-
Zone of inhibition
- DBT:
-
Days before transplanting
- IDM:
-
Integrated disease management
- OAs:
-
Organic amendments
- NA:
-
Nutrient agar
- DMSO:
-
Dimethyl sulfoxide
- SDW:
-
Sterilized distilled water
- CRD:
-
Completely randomised design
- AUDPC:
-
Area under disease progress curve
References
Acero JL, Benitez FJ, Real FJ, González M (2008) Chlorination of organophosphorus pesticides in natural waters. J Hazard Mater 153:320–328. https://doi.org/10.1016/j.jhazmat.2007.08.051
Adam OAO, Abadi RSM, Ayoub SMH (2019) The effect of extraction method and solvents on yield and antioxidant activity of certain sudanese medicinal plant extracts. J Phytopharm 8:248–252. https://doi.org/10.31254/phyto.2019.8507
Ahmad F (2009) Food security in Pakistan. Pak J Agri Sci 46:83–89
Ahmed M, Ji M, Qin P, Gu Z, Liu Y, Sikandar A, Iqbal MF, Javeed A (2019) Phytochemical screening, total phenolic and flavonoids contents and antioxidant activities of Citrullus colocynthis L. and Cannabis sativa L. Appl Ecol Environ Res 17:6961–6979. https://www.epa.hu/02500/02583/00059/pdf/EPA02583_applied_ecology_2019_3_69616979.pdf
Al-Shaibani I, Phulan M, Arijo A, Qureshi T (2008) Ovicidal and larvicidal properties of Adhatoda vasica (L.) extracts against gastrointestinal nematodes of sheep in vitro. Pak Vet J 28:79–83. http://pvj.com.pk/pdf-files/28_2/79-83.pdf
Anith KN, Momol MT, Kloepper JW, Marois JJ, Olson SM, B JJ (2004) Efficacy of plant growth-promoting rhizobacteria, acibenzolar-S-methyl, and soil amendment for integrated management of bacterial wilt on tomato. Plant Dis 88:669–673. https://doi.org/10.1094/PDIS.2004.88.6.669
Arumugam T, Premalashmi V, Theradimani M (2010) Effect of biopesticides, organic amendments and chemicals on the incidence of leaf spot (Cercospora rauwolfiae) in sarpagandha. Green Farm 1:633–635
Aslam MN, Mukhtar T, Hussain MA, Raheel M (2017) Assessment of resistance to bacterial wilt incited by Ralstonia solanacearum in tomato germplasm. J Plant Dis Prot 124:585–590. https://doi.org/10.1007/s41348-017-0100-1
Bailey K, Lazarovits G (2003) Suppressing soil-borne diseases with residue management and organic amendments. Soil Tillage Res 72. https://doi.org/10.1016/S0167-1987(03)00086-2. :169 – 80
Balestra G, Heydari A, Ceccarelli D, Ovidi E, A Q (2009) Antibacterial effect of Allium sativum and Ficus carica extracts on tomato bacterial pathogens. Crop Prot 28:807–811. https://doi.org/10.1016/j.cropro.2009.06.004
Bdliya BS, Dahiru B (2006) Efficacy of some plant extracts on the control of potato tuber soft rot caused by Erwinia carotovora ssp. carotovora. J Plant Prot Res 285:94–94. https://doi.org/10.24425/jppr.2018.124634
Begum N, Haque M, Mukhtar T, Naqvi S, Wang J (2012) Status of bacterial wilt caused by Ralstonia solanacearum in Pakistan. Pak J Phytopathol 24:11–20
Berry U S, Geol (1914 Dodonaea viscosoides. Survey Prof. Paper 84: 142
Bonanomi G, Antignani V, Pane C, Scala F (2007) Suppression of soilborne fungal diseases with organic amendments. J Plant Pathol 1:311–324. https://www.jstor.org/stable/41998409
Cardoso SC, Soares ACF, Brito ADS, Laranjeira FF, Ledo CaS, Santos APD (2006) Control of tomato bacterial wilt through the incorporation of aerial part of pigeon pea and crotalaria to soil. Summa Phytopathol 32:27–33. https://doi.org/10.1590/S0100-54052006000100
Chang HJ, Kim YH, Kang YH, Choi MH, Lee JH (2020) Antioxidant and antibacterial effects of medicinal plants and their stick-type medicinal concentrated beverages. Food Sci Biotechnol 29:1413–1423. https://doi.org/10.1007/s10068-020-00793-9
Dasgupta S, Meisner C, Wheeler D, Xuyen K, Lam NT (2007) Pesticide poisoning of farm workers–implications of blood test results from Vietnam. Int J Hyg Environ Health 210:121–132. https://doi.org/10.1016/j.ijheh.2006.08.006
Dhiman A (2018) Gas chromatography-mass spectroscopy analysis of bioactive compounds in the whole plant parts of ethanolic extract of Asclepias curassavica L. Int J Green Pharm 12:107–114. https://doi.org/10.22377/IJGP.V12I02.1840
Din N, Ahmad M, Siddique M, Ali A, Naz I, Ullah N, Ahmad F (2016) Phytobiocidal management of bacterial wilt of tomato caused by Ralstonia solanacearum (Smith) Yabuuchi. Span J Agri Res 14(3):e1006. https://doi.org/10.5424/sjar/2016143-9012
Doran J, Zeiss M (2000) Soil health and sustainability: managing the biotic component of soil quality. App Soil Ecol 15:3–11. https://doi.org/10.1016/S0929-1393(00)00067-6
Elia A, Conversa G (2012) Agronomic and physiological responses of a tomato crop to nitrogen input. Eur J Agron 40:64–74. https://doi.org/10.1016/j.eja.2012.02.001
Elphinstone JG (2005) The current bacterial wilt situation: a global overview. Bacterial wilt disease and the Ralstonia solanacearum species complex. American Phytopathological Society (APS Press, pp 9–28. https://www.cabdirect.org/cabdirect/abstract/20053172657
Fegan M, Prior P (2005) How complex is the Ralstonia solanacearum species complex. Bacterial wilt disease and the Ralstonia solanacearum species complex. J Bacteriol 1:449–461
Floyd J, Michalak P (2007) New pest response guidelines: Ralstonia solanacearum race 3 biovar 2. USDA APHIS-PPQ, Emergency and Domestic Programs
Fock I, Collonnier C, Luisetti J, Purwito A, Souvannavong V, Vedel F, Servaes A, Ambroise A, Kodja H, Ducreux G, Sihachakr D (2001) Use of Solanum stenotomum for introduction of resistance to bacterial wilt in somatic hybrids of potato. Pl Physiol Biochem 39:899–908. https://doi.org/10.1016/S0981-9428(01)01307-9
Fujiwara A, Kawasaki T, Usami S, Fujie M, Yamada T (2008) Genomic characterization of Ralstonia solanacearum phage φRSA1 and its related prophage (φRSX) in strain GMI1000. J Bacteriol 190:143–156. https://doi.org/10.1128/JB.01158-07
Gilman EF (1999) Dodonaea viscosa. University of Florida, Cooperative Extension Service. Fact Sheet-181
Gomez KA, Gomez AA (1984) Statistical procedures for agricultural research. John Wiley & Sons
Gop (2010) Agricultural statistics of Pakistan. Ministry of Food, Agriculture and Livestock (Economic Wing), Islamabad, pp 45–49
Gopinath K, Saha S, Mina B, Pande H, Kundu S, Gupta H (2008) Influence of organic amendments on growth, yield and quality of wheat and on soil properties during transition to organic production. Nutr Cycl Agroecosyst 82:51–60. https://doi.org/10.1007/s10705-008-9168-0
Gottlieb OR, De Mb Borin MR, Nrs DB (2002) Integration of ethnobotany and phytochemistry: dream or reality. Phytochemistry 60:45–152. https://doi.org/10.1016/S0031-9422(02)00088-2
Grüter D, Schmid B, Brandl H (2006) Influence of plant diversity and elevated atmospheric carbon dioxide levels on belowground bacterial diversity. BMC Microbiol 6:1–8. https://doi.org/10.1186/1471-2180-6-68
Gupta GL, Rana A (2007) Withania somnifera (Ashwagandha): a review. Pharmacogn Rev 1:129–136
Ha TM (2015) Agronomic requirements and production methods of tomatoes in the Red River Delta of Vietnam. J Trop Crop Sci 2:33–38
Harborne A (1998) Phytochemical methods a guide to modern techniques of plant analysis. Springer Science and Business media. Chapman & Hall. London SE1 8HN, UK
Harrington MG, Gadek PA (2010) Phylogenetics of hopbushes and pepperflowers (Dodonaea, Diplopeltis - Sapindaceae), based on nuclear ribosomal ITS and partial ETS sequences incorporating secondary-structure models. Aust Syst Bot 23:431. https://doi.org/10.1071/sb10002
Hassan M, Bereika M, Abo-Elnaga H, Maa S (2009) Direct antimicrobial activity and induction of systemic resistance in potato plants against bacterial wilt disease by plant extracts. Pl Pathol J 25:352–360. https://doi.org/10.5423/PPJ.2009.25.4.352
Hayward A, El-Nashaar H, Nvdegger U, Lindo LD (1990) Variation in nitrate metabolism in biovars of Pseudomonas solanacearum. J Appl Bacteriol 69:269–280. https://doi.org/10.1111/j.1365-2672.1990.tb01518
Hulebak KL, Schlosser W (2002) Hazard analysis and critical control point (HACCP) history and conceptual overview. Risk Anal 22:547–552. https://doi.org/10.1111/0272-4332.00038
Jawahar N, Manivannan R, Jubie S, Saiganesh E (2004) Pharmacognostical and phytochemical studied on Dodonaea viscose Linn. Anc Sci Life 23:1–3
Jeya S, Santhi V, Borgia V, Devi P (2014) In vitro antibacterial activity, phytochemical screening and FT-IR analysis of Dodonaea viscosa and Adhatoda vasica. Asian J Biochem Pharm Res 2:289–298
Jiang G, Wei Z, Xu J, Chen H, Zhang Y, She X, Macho AP, Ding W, Liao B (2017) Bacterial wilt in China: history, current status, and future perspectives. Front Pl Sci 8:1549. https://doi.org/10.3389/fpls.2017.01549
Kang MJ, Lee MH, Shim JK, Seo ST, Shrestha R, Cho MS, Hahn JH, Park DS (2007) PCR-based specific detection of Ralstonia solanacearum by amplification of cytochrome c1 signal peptide sequences. J Microbiol Biotechnol 17:1765–1771. https://koreascience.kr/article/JAKO200709906361717.pdf
Kareem S, Akpan I, Ojo O (2008) Antimicrobial activities of Calotropis procera on selected pathogenic microorganisms. Afr J Biomed Res 11:105–110. https://doi.org/10.4314/ajbr.v11i1.50674
Khan M, Kihara M, Omoloso A (2001) Antimicrobial activity of Horsfieldia helwigii and Melia azedarach. Fitoterapia 72:423–427. https://doi.org/10.1016/S0367-326X(00)00334-8
Khan RAA, Ahmad B, Ahmad M, Ali A, Naz I, Fahim M (2019) Management of Ralstonia solanacearum (Smith) Yabuuchi wilt in tomato (Solanum lycopersicum L.) with dried powder of the medicinal plant Withania somnifera (L.) Dunal. Pak J Bot 51:297–306. https://doi.org/10.30848/PJB2019
Khan RAA, Tang Y, Naz I, Alam SS, Wang W, Ahmad M, Najeeb S, Rao C, Li Y, Xie B, Li Y (2021) Management of Ralstonia solanacearum in tomato using ZnO nanoparticles synthesized through Matricaria chamomilla. Pl Dis 105:3224–3230. https://doi.org/10.1094/PDIS-08-20-1763-RE
Khurram M, Khan MA, Hameed A, Abbas N, Qayum A, Inayat H (2009) Antibacterial activities of Dodonaea viscosa using contact bioautography technique. Molecules 14:1332–1341. https://doi.org/10.3390/molecules14031332
Lin CH, Tsai KC, Prior P, Wang JF (2014) Phylogenetic relationships and population structure of Ralstonia solanacearum isolated from diverse origins in T aiwan. Plant Pathol 63:1395–1403. https://doi.org/10.1111/ppa.12209
Little E, Skolmen R (1989) Common forest trees of Hawaii. USDA Forest Serv. U.S. Department of Agriculture, Washington DC. Agric. Handbook, p 321
Madden LV, Hughes G, Van Den Bosch F, Madden LV, Hughes G, Van Den Bosch F (2007) 2007. Temporal Analysis I Quantifying and Comparing Epidemics. in: The Study of Plant Disease Epidemics American Phytopathological Society (APS). pp. 63–116 https://doi.org/10.1094/9780890545058.004
Mamphogoro T, Babalola O, Aiyegoro O (2020) Sustainable management strategies for bacterial wilt of sweet peppers (Capsicum annuum) and other Solanaceous crops. J Appl Microbiol 129:496–508. https://doi.org/10.1111/jam.14653
Mathalaimuthu B, Shanmugam D, Kovendan K, Kadarkarai M, Jayapal G, Benelli G (2017) Coleus aromaticus leaf extract fractions: a source of novel ovicides, larvicides and repellents against Anopheles, Aedes and Culex mosquito vectors? Process Saf Environ Prot 106:23–33. https://doi.org/10.1016/j.psep.2016.12.003
Minfal (2009) Ministry of Food, Agric. and Livest. Econ. Wing, Islamabad, Pakistan. Agricultural Statistics of Pakistan. Fruit, vegetables and condiments statistics of Pakistan. Islamabad. pp. 11–22
Mostafa AA, Al-Askar AA, Almaary KS, Dawoud TM, Sholkamy EN, Bakri MM (2018) Antimicrobial activity of some plant extracts against bacterial strains causing food poisoning diseases. Saudi J Bio Sci 25:361–366. https://doi.org/10.1016/j.sjbs.2017.02.004
Najeeb S, Ahmad M, Khan R, Naz IAA, Alam S (2019) Management of bacterial wilt in tomato using dried powder of Withania coagulan (L) Dunal. Australas Plant Pathol 48:183–192. https://doi.org/10.1007/s13313-019-0618-8
Nakato G, Akinbade S, Kumar PL, Bandyopadhyay R, Beed F (2013) 11 development of ELISA for the detection of Xanthomonas campestris. Banana systems in the humid highlands of sub-saharan Africa: enhancing resilience and productivity. CABI, Wallingford UK, pp 93–100. https://doi.org/10.1079/9781780642314.0093
Naz I, Khan M (2013) Nematicidal activity of nonacosane-10-ol and 23a-homostigmast-5-en-3β-ol isolated from the roots of Fumaria parviflora (Fumariaceae). J Agri Food Chem 61:5689–5695. https://doi.org/10.1021/jf401309r
Naz I, Palomares-Rius JE, Khan SM, Ali S, Ahmad M, Ali A, Khan A (2015) Control of Southern root knot nematode Meloidogyne incognita (Kofoid and White) Chitwood on tomato using green manure of Fumaria parviflora Lam (Fumariaceae). Crop Prot 67:121–129. https://doi.org/10.1016/j.cropro.2014.10.005
Nazar S, Hussain M, Khan A, Muhammad G, Bukhari S (2020) Alkaloid-rich plant Tylophora indica; current trends in isolation strategies, chemical profiling and medicinal applications. Arab J Chem 13:6348–6365. https://doi.org/10.1016/j.arabjc.2020.05.037
Oguwike F, Onubueze D, Ughachukwu P (2013) Evaluation of activities of marigold extract on wound healing of albino wister rat. IOSR J Dent Med Sci 8:67–70
Pietrarelli L, Balestra G, Varvaro L (2006) Effects of simulated rain on Pseudomonas syringae pv. Tomato populations on tomato plants. J Plant Pathol 2:245–251. https://www.jstor.org/stable/41998328
Pirzada A, Shaikh W, Usmanghani K, Mohiuddin E (2010) Antifungal activity of Dodonaea viscosa Jacq extract on pathogenic fungi isolated from super ficial skin infection. Pak J Pharm Sci 23:337–340
Raj A, Lawerence R (2018) Green synthesis and characterization of ZnO nanoparticles from leaves extracts of Rosa indica and its antibacterial activity. Ras J Chem 11:1339–1348. https://doi.org/10.31788/RJC.2018.1132009
Ramya R, Sivasakthi R, Senthilkumar C, Anudeepa J, Santhi N, Narayanan RV (2011) Preliminary phytochemical and antifertility studies on Dodonea viscosa Linn. Asian J Res Pharm Sci 1:77 – 9. https://www.indianjournals.com/ijor.aspx?target=ijorajrps&volume=1&issue=3&article=007
Ranjit PM, Santhipriya T, Nagasri S, Chowdary Y, Pasumarthy N, Gopal V (2012) Preliminary phytochemical screening and antibacterial activities of ethanolic extract of Calotropis procera flowers against human pathogenic strains. Asian J Pharm Clin Res 5:127–131
Rivard C, O’connell S, Peet M, Welker R, Louws F (2012) Grafting tomato to manage bacterial wilt caused by Ralstonia solanacearum in the southeastern United States. Plant Dis 96:973–978. https://doi.org/10.1094/PDIS-12-10-0877
Schönfeld J, Gelsomino A, Van Overbeek L, Gorissen A, Smalla K, Van Elsas J (2003) Effects of compost addition and simulated solarisation on the fate of Ralstonia solanacearum biovar 2 and indigenous bacteria in soil. FEMS Microbio Ecol 43:63–74. https://doi.org/10.1111/j.1574-6941.2003.tb01046.x
Scotti R, Bonanomi G, Scelza R, Zoina A, Rao M (2015) Organic amendments as sustainable tool to recovery fertility in intensive agricultural systems. Soil Sci Plant Nutr 15:333–352. https://doi.org/10.4067/S0718-95162015005000031
Shafek RE, Shafik NH, Michael HN, El-Hagrassi AM, Osman AF (2015) Phytochemical studies and biological activity of Dodonaea viscosa flowers extract. J Chem Pharm Res 7:109–116
Shahbaz MU, Mukhtar T, Begum N (2015) Biochemical and serological characterization of Ralstonia solanacearum Associated with Chilli Seeds from Pakistan. Int J Agric Biol 17:31–40
Tahir MI, Inam-Ul-Haq M, Ashfaq M, Abbasi NA (2014) Surveillance of Ralstonia solanacearum infecting potato crop in Punjab. Pak J Phytopathol 26:45–52. http://pakps.com/pjp/index.php/PJP/article/view/89/42
Treonis AM, Austin EE, Buyer JS, Maul JE, Spicer L, Zasada IA (2010) Effects of organic amendment and tillage on soil microorganisms and microfauna. Appl Soil Ecol 46:103–110. https://doi.org/10.1016/j.apsoil.2010.06.017
Van Elsas JD, Kastelein P, Van Bekkum P, Van Der Wolf JM, De Vries PM, Van Overbeek LS (2000) Survival of Ralstonia solanacearum biovar 2, the causative agent of potato brown rot, in field and microcosm soils in temperate climates. Phytopathology 90:1358–1366. https://doi.org/10.1094/PHYTO.2000.90.12.1358
Varma J, Dubey N (1999) Prospectives of botanical and microbial products as pesticides of tomorrow. Curr Sci: 172–9. https://www.jstor.org/stable/24101232
Venkatesh S, Reddy Y, Ramesh M, Swamy M, Mahadevan N, Suresh B (2008) Pharmacognostical studies on Dodonaea viscosa leaves. Afr J Pharm Pharmacol 2:83–88. https://doi.org/10.5897/AJPP.9000220
Wai KPP, Lee J, Mo HS, Kim BS (2013) Sources of resistance to bacterial wilt and restorer-of-fertility genotype for cytoplasmic male sterility in Capsicum pepper. Hortic Environ Biotechnol 54:266–271. https://doi.org/10.1007/s13580-013-0006-1
Wang JF, Lin CH (2005) Integrated management of tomato bacterial wilt. AVRDC-The World Vegetable Center; 2005. https://worldveg.tind.io/record/12956/
Whipps J (2007) Biological pesticides for control of seed-and soil-borne plant pathogen. Mod Soil Microbiol 10021000596 https://cir.nii.ac.jp/crid/1570854175613702528
Zanón M, Font M, Jordá C (2011) Use of tomato crop residues into soil for control of bacterial wilt caused by Ralstonia solanacearum. J Crop Prot 30:1138–1143. https://doi.org/10.1016/j.cropro.2011.03.025
Author information
Authors and Affiliations
Contributions
All authors have participated in (a) conception and design, or analysis and interpretation of the data; (b) drafting the article or revising it critically for important intellectual content. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Institutional review board statement
Not applicable.
Informed consent statement
Not applicable.
Competing interests
The authors declare that they have no competing interests, any financial or personal relationships that may be perceived as influencing their work.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ahmad, K., Khan, R.A.A., Ahmad, M. et al. Evaluating powder preparation of Dodonaea viscosa as a new potential control measure against bacterial wilt of tomato. J Plant Pathol 105, 921–932 (2023). https://doi.org/10.1007/s42161-023-01403-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-023-01403-3