Abstract
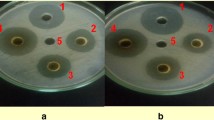
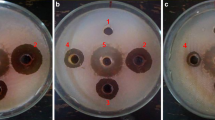
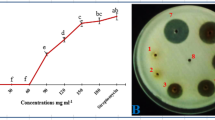
Because of its known anti-bacterial properties, we explored the potential of Xanthium strumarium, an invasive, enormous mass-producing weed, for the control of Ralstonia solanacearum which causes bacterial wilt (BW) of tomato. Both in-vitro and in-planta experiments were conducted, using different concentrations of the dried powders of the plant parts applied to infested soil at different times. Addition of a 20% (w/v) aqueous extract of leaf powder or succulent shoot powder to wells cut in nutrient agar inhibited growth of R. solanacearum. In in-planta experiments, 4.5% (w/w) leaf powder applied to artificially infested soil 10 days before transplant (DBT), produced the best effect and enhanced root length, shoot length, and plant fresh bio-mass by 64%, 37%, and 42%, respectively, as compared to inoculated control. Leaf powder also lowered the area under disease progress curve (AUDPC) by 38%, and the pathogen counts (g−1 dry soil) by 1.202 log10 units. Succulent shoot powder (4.5% w/w) applied 20 DBT proved to be better than other application times and increased root length, shoot length, and plant fresh bio-mass by 55%, 42%, and 57%, respectively, as compared to inoculated control. Succulent shoot powder also decreased AUDPC by 35%, and the pathogen counts (g−1 dry soil) by 1.294 log10 units. Our data strongly suggest that 4.5% (w/w) of leaf or succulent shoot powder, applied 20 DBT, can be an effective component of the integrated disease management (IDM) against BW.

Similar content being viewed by others
References
Abdel-Monaaim MF, Abo-Elyousr KAM, Morsy KM (2011) Effectiveness of plant extracts on suppression of damping-off and wilts diseases of lupine (Lupinustermis forsik). Crop Prot 30:185–191. https://doi.org/10.1016/j.cropro.2010.09.016
Aliyu TH, Balogun OS, Adeoti OM (2011) Pathogenic responses of cowpea (vigna unguiculata) inoculated with cucumber mosaic virus to soil amendment with neem leaf powder. Agrosearch 11:99–110. https://doi.org/10.4314/agrosh.v11i1.10
Al-Obaidi O (2014) Studes on anti bacterial and anticancer activity of Nerium oleander extracts. Eur Chem Bull 3:259–262. https://doi.org/10.17628/ecb.2014.3.259-262
Anith KN, Momol MT, Kloepper JW, Marois JJ, Olson SM, Jones JB (2004) Efficacy of plant growth-promoting rhizobacteria, acibenzolar-s-methyl, and soil amendment for integrated management of bacterial wilt on tomato. Plant Dis 88:669–673. https://doi.org/10.1094/PDIS.2004.88.6.669
Arthy JR, Akiew EB, Kirkegaard JA, Trevorrow PR (2005) Using Brassica spp. as biofumigants to reduce the population of Ralstonia solanacearum. Bacterial wilt disease and the Ralstonia solanacearum species complex. American Phytopathological Society Press, St Paul, pp 159–165
Askarne L, Talibi I, Boubaker H, Boudyach EH, Msanda F, Saadi B, Aoumar AB (2012) Use of Moroccan medicinal plant extracts as botanical fungicide against citrus blue mould. Lett Appl Microbiol 56(1):37–43. https://doi.org/10.1111/lam.12012
Aysan Y, Karatas A, Cinar O (2003) Biological control of bacterial stem rot caused by Erwinia chrysanthemi on tomato. Crop Prot 22:807–811. https://doi.org/10.1016/S0261-2194(03)00030-9
Balestra GM, Heydari A, Ceccarelli D, Ovidi E, Quattrucci A (2009) Antibacterial effect of Allium sativum and Ficus carica extracts on tomato bacterial pathogens. Crop Prot 28(10):807–811. https://doi.org/10.1016/j.cropro.2009.06.004
Begum N, Haque MI, Mukhtar T, Naqvi SM, Wang JF (2012) Status of bacterial wilt caused by Ralstonia solanacearum in Pakistan. Pak J Phytopathol 24(1):11–20 http://pakps.com/pjp/files/11-20-irfan-paper.pdf
Bonanomi G, Antignani V, Pane C, Scala F (2007) Suppression of soilborne fungal disease with organic amendments. J Plant Pathol 89:311–324 https://www.jstor.org/stable/41998409
Brady N, Weil R (1999) Elements of the nature and properties of soil spp. Prentice-Hall, Upper Saddle River, p 559 https://trove.nla.gov.au/version/45005208
Burney K, Roshan Z, Iftikhar A (1999) Bacterial wilt caused by Ralstonia (Pseudomonas) solanacearum in Solanaceous crops of Pakistan. In: Proceeding 2nd national conference of plant pathology, pp 27–29
Campbell CL, Madden LV (1990) Introduction to plant disease epidemiology. Wiley, New York
Cardoso SC, Soares ACF, Brito ADS, Laranjeira FF, Ledo CAS, Santos AP (2006) Control of tomato bacterial wilt through the incorporation of aerial part of pigeon pea and crotalaria to soil. Summa Phytopathol 32:27–33. https://doi.org/10.1590/S0100-54052006000100004
Champoiseau PG, Jeffrey BJ, Timur MM (2009) Ralstonia solanacearum race 3 biovar 2; Detection, exclusion and analysis of select agent. Educational modules, Bacterial Wilt of Tomato, 1–17. https://www.ars.usda.gov/ARSUserFiles/00000000/opmp/RalstoniaR3b2May2010.pdf
Denny T (2007) Plant pathogenic Ralstonia species. In: Plant-associated bacteria. Springer, Dordrecht, pp 573–644
Devkota A, Das RK (2015) Antimicrobial activities of Xanthium strumarium L. J Nat Hist Mus 29:70–77. https://doi.org/10.3126/jnhm.v29i0.19039
Din N, Ahmad M, Siddique M, Ali A, Naz I, Ullah N, Ahmad F (2016) Phytobiocidal management of bacterial wilt of tomato caused by Ralstonia solanacearum (Smith) Yabuuchi. Span J Agric Res 14(3):1006. https://doi.org/10.5424/sjar/2016143-9012
Elphinstone JG (2005) The current bacterial wilt situation: a global overview. In: Allen C, Prior P, Hayward AC (eds) Bacterial wilt disease and the Ralstonia solanacearum species complex. American Phytopathological Society Press, St Paul, pp 9–28 https://ci.nii.ac.jp/naid/10029718988/#cit
Farooq U, Waseem B, Muzaffar R, Tripathi J, Tharani M, Sharma M (2014) A comparative study of phytochemical investigation of Xanthium strumarium medicinal plant. Int J Res Pharm Chem 4(1):96–100 http://www.ijrpc.com/files/15-426.pdf
Floyd J (2007) New pest response guidelines: Ralstonia solanacearum Race 3 biovar 2//USDA APHIS- PPQ, Emergency and Domestic Programs, 45 pp
Gomez KA, Gomez AA (1984) Statistical procedures for agricultural research. Wiley, New York
Goszczynska T, Serfontein JJ, Serfontein S (2000) Media and diagnostic tests, introduction to practical phytobacteriology. Bacterial Diseases Unit, ARC-Plant Protection Research Institute, Pretoria, pp 60–73
Gruter D, Schmid B, Brandl H (2006) Influence of plant diversity and elevated atmospheric carbon dioxide levels on below ground bacterial diversity. BMC Microbiol 6:1–8. https://doi.org/10.1186/1471-2180-6-68
Han T, Li H, Zhang Q, Zheng H, Qin L (2006) New thiazinediones and other components from Xanthium strumarium. Chem Nat Compd 42:567–570. https://doi.org/10.1007/s10600-006-0215-2
Hassan MAE, Bereika MFF, Abo-Elnaga HIG, Sallam MA (2009) Direct antimicrobial activity and induction of systemic resistance in potato plants against bacterial wilt disease by plant extracts. Plant Pathol J 25(4):352–360. https://doi.org/10.5423/PPJ.2009.25.4.352
Hayward AC (1991) Biology and epidemiology of bacterial wilt caused by Pseudomonas solanacearum. Annu Rev Phytopathol 29:65–87. https://doi.org/10.1146/annurev.py.29.090191.000433
Hayward AC (1994) The hosts of Pseudomonas solanacearum. In: Hayward AC, Hartman GL (eds) Bacterial wilt: the disease and its causative agent, Pseudomonas solanacearum. CAB International, Wallingford, pp 9–25 https://www.cabdirect.org/cabdirect/abstract/19952310916
Huang J, Li H, Yuan H (2006) Effect of organic amendments on Verticillium wilt of cotton. Crop Prot 25:1167–1173. https://doi.org/10.1016/j.cropro.2006.02.014
Huet G (2014) Breeding for resistance to Ralstonia solanacearum. Front Plant Sci 5:715–719. https://doi.org/10.3389/fpls.2014.00715
Ji P, Momol MT, Olson SM, Pradhanang PM, Jones JB (2005) Evolution of thymol as biofunmigant for control of bacterial wilt of tomato under field conditions. Plant Dis 89(5):497–500. https://doi.org/10.1094/PD-89-0497
Ji P, Momol MT, Olson SM, Rich JR, Jones JB (2007) Development of an integrated approach for managing bacterial wilt and root-knot on tomato under field conditions. Plant Dis 91(10):1321–1326. https://doi.org/10.1094/PDIS-91-10-1321
Kagale S, Marimuthu T, Thayumanavan B, Nandakumar R, Samiyappan R (2004) Antimicrobial activity and induction of systemic resistance in rice by leaf extract of Datura metel against Rhizoctonia solani and Xanthomonas oryzae pv. oryzae. Physiol Mol Plant Pathol 65(2):91–100. https://doi.org/10.1016/j.pmpp.2004.11.008
Kang JM, Lee MH, Shim JK, Seo ST, Sherestha R, Cho MS, Hahn JH, Park DS (2007) PCR-based specific detection of Ralstonia solanacearum by amplification of cytochrome c1 signal peptide sequences. J Microbiol Biotechnol 17(11):1765–1771 https://www.ncbi.nlm.nih.gov/pubmed/18092459
Khan RAA, Ahmad B, Ahmad M, Ali A, Naz I, Fahim M (2019) Management of Ralstonia solanacearum (Smith) Yabuuchi wilt in tomato (Solanum lycopersium L.) with dried powder of the medicinal plant Withania somnefera (L) Dunal. Pak J Bot 51(1):297–306. https://doi.org/10.30848/PJB2019-1(8)
Kim YS, Kim JS, Park S, Choi S, Lee CO, Kim S, Kim Y, Kim SH, Ryu SY (2003) Two cytotoxic Sesquiterpene lactones from the leaves of Xanthium strumarium and their in vitro inhibitory activity on Farnesyl transferase. Planta Med 69:375–377. https://doi.org/10.1055/s-2003-38879
Kokalis-Burelle N, Chellemi DO, Perie X (2005) Effect of soils from six management systems on root-knot nematodes and plant growth in greenhouse assays. J Nematol l37:467–472 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2620999/
Laterrot HE (1998) Resistance to bacteria in tomato. In: Scarasci Mugnozza GT, Porceddu E, Pagnotta MA (eds) Genetics and breeding for crop quality and resistance. Proceedings of the XV EUCARPIA Congress, Viterbo, Italy, September 20–25. Kluwer Academic Publishers, Dordrecht, pp 103–110
Lin CH, Tsai KC, Prior P, Wang JF (2014) Phylogenetic relationships and population structure of R alstonia solanacearum isolated from diverse origins in Taiwan. Plant Pathol 63(6):1395–1403. https://doi.org/10.1111/ppa.12209
Lo Cantore P, Iacobellis NS, De Marco A, Capasso F, Senatore F (2004) Antibacterial activity of Coriandrum sativum L. and Foeniculum vulgare Miller var Vulgare (miller) essential oils. L Agric Food Chem 52(26):7862–7866 https://www.ncbi.nlm.nih.gov/pubmed/15612768
Madden LV, Hughes G, Van den Bosch F (2007) The study of plant disease epidemics. APS Press, St. Paul
Mazola M (2002) Mechanisms of natural soil suppressiveness to soilborne diseases. Antonie Van Leeuwenhoek 81:557–564. https://doi.org/10.1023/A:1020557523557
MINFAL (2009) Agriculture statistic of Pakistan. Ministry Of Food, Agriculture and Livestock (Economic Wing), Islamabad, pp 71–72
Mitra J, Paul PK (2017) A potent biocide formulation inducing SAR in plants. J Plant Dis Prot 124(2):163–175. https://doi.org/10.1007/s41348-016-0067-3
Najeeb S, Ahmad M, Khan RAA, Naz I, Ali A, Alam SS (2019) Management of bacterial wilt in tomato using dried powder of Withania coagulan (L) Dunal. Aust Plant Pathol. https://doi.org/10.1007/s13313-019-0618-8
Naz I, Saifullah, Palomeres-Rius JE, Khan SM, Ali S, Ahmad M, Ali A, Khan A (2015a) Control of southern Root-knot nematode, Meloidogyne incognita (Kofoid and white) chitwood on tomato using green manure of Fumaria parviflora Lam (Fumariaceae). Crop Prot 67:121–129. https://doi.org/10.1016/j.cropro.2014.10.005
Naz I, Saifullah, Palomeres-Rius JE, Block V, Khan SM, Ali S, Baig A (2015b) Sustainable management of the southern Root-knot nematode, Meloidogyne incognita (Kofoid and white) chitwood, by means of amendments of Fumaria parviflora. Int J Agric Biol 17:289–296 http://www.fspublishers.org/published_papers/72259_..pdf
Nguyen MT, Ranamukhaarachchi SL (2010) Soil-borne antagonists for biological control of bacterial wilt disease caused by Ralstonia solanacearum in tomato and pepper. J Plant Pathol 92(2):395–405 https://www.jstor.org/stable/41998815
Oguwike FN, Onubueze DPM, Ughachukwu P (2013) Evaluation of activities of marigold extract on wound healing of albino wister rat. IOSR J Dent Med Sci 8:67–70. https://doi.org/10.9790/0853-0856770
Perez C, Pauli M, Bazerque P (1990) An antibiotic assay by agar well diffusion method. Acta Biol Med Exp 15:113–115 https://ci.nii.ac.jp/naid/10024190094/
Philogène BJR, Regnault-Roger C, Vincent C (2005) Botanicals: yesterday’s and today’s promises. In: Biopesticides of plant origin. Intercept, Lavoisier, pp 1–15 https://pubs.acs.org/doi/abs/10.1021/np058244q
Pradhanang PM, Momol MT, Rich JR, Olson SM, Jones JB (2003) Effect of plant essential oils on Ralstonia solanacearum population density and bacterial wilt incidence in tomato. Plant Dis 87(4):423–427. https://doi.org/10.1094/PDIS.2003.87.4.423
Qasem JR, Abu-Blan HA (1996) Fungicidal activity of some common weed extracts against different plant pathogenic fungi. J Phytopathol 144(3):157–161. https://doi.org/10.1111/j.1439-0434.1996.tb01507.x
Saddler GS (2005) Management of bacterial wilt disease. In: Allen C, Prior P, Hayward AC (eds) Bacterial wilt disease and the Ralstonia solanacearum species complex. APS Press, St. Paul, pp 121–132
Saha D, Kumar R, Ghosh S, Kumari M, Saha A (2012) Control of foliar diseases of tea with Xanthium strumarium leaf extract. Ind Crop Prod 37:376–382. https://doi.org/10.1016/j.indcrop.2011.12.030
Schonfeld J, Gelsomino A, Van-Overbeek LS, Gorissen A, Smalla K, Van-Elas JD (2003) Effects of compost addition and simulated solarisation on the fate of Ralstonia solanacearum biovar 2and indigeneous bacteria in soil. FEMS Microbiol Ecol 43:63–74. https://doi.org/10.1111/j.1574-6941.2003.tb01046.x
Shahbaz MU, Mukhtar T, Ul-Haque MI, Begum N (2015) Biochemical and serological characterization of Ralstonia solanacearum associated with chilli seeds from Pakistan. Int J Agric Biol 17:31–40
Sharma JR, Cheema GS, Saini SS, Gill BS (2010) Soft rot disease of Aloebarbadensis and its management. J Res Punjab Agric Univ 47:18–19 https://www.cabdirect.org/cabdirect/abstract/20113213221
Sikkema J, dse Bont JAM, Poolman B (1994) Interactions of cyclic hydrocarbons with biological membranes. Chemical compounds from essential oils also act on cytoplasmic membrane proteins. J Biol Chem 269(11):8022–8028 http://www.jbc.org/content/269/11/8022.short
Tanaka JCA, da Silva CC, de Oliveira AJB, Nakamura CV, Dias BP (2006) Antimicrobial activity of indole alkaloids from Aspidosperma ramiflorum. Braz J Med Biol Res 39(3):387–391. https://doi.org/10.1590/S0100-879X2006000300009
Treonis AM, Austin EE, Buyer JS, Maul JE, Spicer L, Zasada IA (2010) Effects of organic amendment and tillage on soil microorganisms and microfauna. Appl Soil Ecol 46:103–110. https://doi.org/10.1016/j.apsoil.2010.06.017
Tripathi P, Dubey NK (2004) Exploitation of natural products as an alternative strategy to control post-harvest fungal rotting of fruits and vegetables. Postharvest Biol Technol 32:235–245. https://doi.org/10.1016/j.postharvbio.2003.11.005
Wai KPP, Lee J, Mo H, Kim B (2013) Sources of resistance to bacterial wilt and restorer-of-fertility genotype for cytoplasmic male sterility in Capsicum Pepper. Hortic Environ Biotechnol 54(3):266–271. https://doi.org/10.1007/s13580-013-0006-1
Walters D, Walsh D, Newton A, Lyon G (2005) Induced resistance for plant disease control: maximizing the efficacy of resistance elicitors. Phytopathology 95(12):1368–1373. https://doi.org/10.1094/PHYTO-95-1368
Wang JF, Lin CH (2005) Integrated management of tomato bacterial wilt. AVRDC-The World Vegetable Center, Taiwan
Wang Y, McAllister TA, Yanke LJ, Cheeke PR (2000) Effect of steroidal saponin from Yucca schidigera extract on ruminal mmicrobes. J Appl Microbiol 88(5):887–896. https://doi.org/10.1046/j.1365-2672.2000.01054.x
Wenneker M, Verdel M, Groaeneveld R, Kempennar C, van Beuningen A, Janse J (1999) Ralstonia (Pseudomonas) solanacearum race 3 (bio var 2) in surface water and natural weed hosts: first report on stinging nettle (Urtica dioica). Eur J Plant Pathol 105(3):307–315. https://doi.org/10.1023/A:1008795417575
Whipps JM, Gerhardson B (2007) Biological pesticides for control of seed- and soil-borne plant pathogens. A training course guide. In: Van Elsas JD, Jansson JD, Trevors JT (eds) Modern soil microbiology, 2nd edn. CRC Press, Boca Raton, pp 479–501 https://ci.nii.ac.jp/naid/10021000596
Yadava RN, Jharbade J (2007) Novel biologically active triterpenoid saponin from the leaves of Xanthium strumarium Linn. Asian J Chem 19:1224–1230
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khan, R.A.A., Ahmad, M., Naz, I. et al. Sustainable management of bacterial wilt of tomato using dried powder of Xanthium strumarium L.. J Plant Pathol 102, 421–431 (2020). https://doi.org/10.1007/s42161-019-00451-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42161-019-00451-y