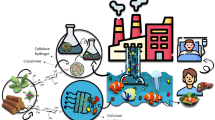
Abstract
The preparing biopolymer composite adsorbents for environmental friendliness applications remarkably attracted much attention. Herein, novel biopolymer nanocomposite of carboxymethyl cellulose (CMC), guar gum (GG), and graphene oxide (GO) was used to prepare the CMC/GG and CMC/GG/GO with different weight percents of GO (1, 3, and 5% denoted as CMC/GG/GO-1, CMC/GG/GO-3, and CMC/GG/GO-5, respectively) and their performance for removing cationic dye (MG: malachite green) from aqueous solution was studied. XRD, FTIR, SEM, EDX, and BET analyses were used to characterize the materials. Influential parameters such as adsorbent amount, initial dye concentration, contact time, temperature, and pH on the removal efficiency of contaminant from aqueous solutions were investigated. CMC/GG/GO-3 nanocomposite was selected as the optimal, and the max adsorption capacity 17.6 mg/g and removal efficiency 88.2% were obtained. Isotherm data (Langmuir, Freundlich, and Temkin) showed that the Langmuir isotherm corresponds to a correlation coefficient of 0.98. Thermodynamic data indicated that the adsorption was a spontaneous and endothermic process. Also, the study of pseudo-first-order and pseudo-second-order kinetic models signed a good agreement with the pseudo-second-order with R2 = 1.
Graphical abstract















Similar content being viewed by others
References
Jadhav SA, Khedkar MV, Somvanshi SB, Jadhav KM (2021) Magnetically retrievable nanoscale nickel ferrites: an active photocatalyst for toxic dye removal applications. Ceram Int 47(20):28623–28633. https://doi.org/10.1016/j.ceramint.2021.07
Ma S, Ji Y, Dong Y, Chen S, Wang Y, Lü S (2021) An environmental-friendly pesticide-fertilizer combination fabricated by in-situ synthesis of ZIF-8. Sci Total Environ 789:147845. https://doi.org/10.1016/j.scitotenv.2021.14
Kumar P, Bansal V, Kim K-H, Kwon EE (2018) Metal-organic frameworks (MOFs) as futuristic options for wastewater treatment. J Ind Eng Chem 62:130–145. https://doi.org/10.1016/j.jiec.2017.12.051
Zhang Y, Pan Y, Li P, Zeng X, Guo B, Pan J, Yin X (2021) Novel Schiff base-based cationic Gemini surfactants as corrosion inhibitors for Q235 carbon steel and printed circuit boards. Colloids Surf A 623:126717. https://doi.org/10.1016/j.colsurfa.2021.126
Noreen S, Perveen S, Shafiq N, Aslam S, Iqbal HMN, Ashraf SS, Bilal M (2021) Laccase-loaded functionalized graphene oxide assemblies with improved biocatalytic properties and decolorization performance. Environ Technol Innov 24:101884. https://doi.org/10.1016/j.eti.2021.101884
dos Reis LGT, Robaina NF, Pacheco WF, Cassella RJ (2011) Separation of malachite green and methyl green cationic dyes from aqueous medium by adsorption on Amberlite XAD-2 and XAD-4 resins using sodium dodecylsulfate as carrier. Chem Eng J 171:532–540. https://doi.org/10.1016/j.cej.2011.04.024
Didehban A, Zabihi M, Faghihi M, Akbarbandari F, Akhtarivand H (2021) Design and fabrication of core-shell magnetic and non-magnetic supported carbonaceous metal organic framework nanocomposites for adsorption of dye. J Phys Chem Solids 152:109930. https://doi.org/10.1016/j.jpcs.2020.109930
Gupta K, Khatri OP (2017) Reduced graphene oxide as an effective adsorbent for removal of malachite green dye: plausible adsorption pathways. J Colloid Interface Sci 501:11–21. https://doi.org/10.1016/j.jcis.2017.04.035
Buvaneswari N, Kannan C (2011) Plant toxic and non-toxic nature of organic dyes through adsorption mechanism on cellulose surface. J Hazard Mater 189:294–300. https://doi.org/10.1016/j.jhazmat.2011.02.036
Pinar Gumus Z, Soylak M (2021) Metal organic frameworks as nanomaterials for analysis of toxic metals in food and environmental applications. TrAC, Trends Anal Chem 143:116417. https://doi.org/10.1016/j.trac.2021.116417
Salleh MAM, Mahmoud DK, Karim WAWA, Idris A (2011) Cationic and anionic dye adsorption by agricultural solid wastes: a comprehensive review. Desalination 280:1–13. https://doi.org/10.1016/j.desal.2011.07.019
Eltaweil AS, Elgarhy GS, El-Subruiti GM, Omer AM (2020) Carboxymethyl cellulose/carboxylated graphene oxide composite microbeads for efficient adsorption of cationic methylene blue dye. Int J Biol Macromol 154:307–318. https://doi.org/10.1016/j.ijbiomac.2020.03.122
Xu G-R, An Z-H, Xu K, Liu Q, Das R, Zhao H-L (2021) Metal organic framework (MOF)-based micro/nanoscaled materials for heavy metal ions removal: the cutting-edge study on designs, synthesis, and applications. Coord Chem Rev 427:213554. https://doi.org/10.1016/j.ccr.2020.213554
Lin Z, Pang S, Zhou Z, Wu X, Bhatt P, Chen S (2021) Current insights into the microbial degradation for butachlor: strains, metabolic pathways, and molecular mechanisms. Appl Microbiol Biotechnol 105(11):4369–4381. https://doi.org/10.1007/s00253-021-11346-3
Nouri L, Hemidouche S, Boudjemaa A, Kaouah F, Sadaoui Z, Bachari K (2020) Elaboration and characterization of photobiocomposite beads, based on titanium (IV) oxide and sodium alginate biopolymer, for basic blue 41 adsorption/photocatalytic degradation. Int J Biol Macromol 151:66–84. https://doi.org/10.1016/j.ijbiomac.2020.02
Vatanpour V, Faghani S, Keyikoglu R, Khataee A (2020) Enhancing the permeability and antifouling properties of cellulose acetate ultrafiltration membrane by incorporation of ZnO@graphitic carbon nitride nanocomposite. Carbohyd Polym. https://doi.org/10.1016/j.carbpol.2020.1174
Yu Z, Hu C, Dichiara AB, Jiang W, Gu J (2020) Cellulose nanofibril/carbon nanomaterial hybrid aerogels for adsorption removal of cationic and anionic organic dyes. Nanomaterials 10:1–20. https://doi.org/10.3390/nano10010169
Zhang L, Zhang H, Guo W, Tian Y (2014) Removal of malachite green and crystal violet cationic dyes from aqueous solution using activated sintering process red mud. Appl Clay Sci 93–94:85–93. https://doi.org/10.1016/j.clay.2014.03.004
Karimzadeh Z, Javanbakht S, Namazi H (2019) Carboxymethylcellulose/MOF-5/graphene oxide bio-nanocomposite as antibacterial drug nanocarrier agent. BioImpacts 9:5–13. https://doi.org/10.15171/bi.2019.02
Juengchareonpoon K, Wanichpongpan P, Boonamnuayvitaya V (2021) Graphene oxide and carboxymethylcellulose film modified by citric acid for antibiotic removal. J Chem Eng Environ. https://doi.org/10.1016/j.jece.2020.104637
Kaur K, Jindal R (2019) Meenu, Self-assembled GO incorporated CMC and chitosan-based nanocomposites in the removal of cationic dyes. Carbohydr Polym 225:115245. https://doi.org/10.1016/j.carbpol.2019.115245
Varaprasad K, Jayaramudu T, Sadiku ER (2017) Removal of dye by carboxymethyl cellulose, acrylamide and graphene oxide via a free radical polymerization process. Carbohydr Polym 164:186–194. https://doi.org/10.1016/j.carbpol.2017.01.094
Pashaei-Fakhri S, Peighambardoust SJ, Foroutan R, Arsalani N, Ramavandi B (2021) Crystal violet dye sorption over acrylamide/graphene oxide bonded sodium alginate nanocomposite hydrogel. Chemosphere 270:129419. https://doi.org/10.1016/j.chemosphere.2020
Sharma G, Sharma S, Kumar A, Al-Muhtaseb AH, Naushad M, Ghfar AA, Mola GT, Stadler FJ (2018) Guar gum and its composites as potential materials for diverse applications: a review. Carbohydr Polym 199:534–545. https://doi.org/10.1016/j.carbpol.2018.07.053
Thombare N, Jha U, Mishra S, Siddiqui MZ (2016) Guar gum as a promising starting material for diverse applications: a review. Int J Biol Macromol 88:361–372. https://doi.org/10.1016/j.ijbiomac.2016.04.001
Jain V, Tammishetti V, Joshi K, Kumar D, Pradip, Rai B (2017) Guar gum as a selective flocculant for the beneficiation of alumina rich iron ore slimes: density functional theory and experimental studies. Miner Eng 109:144–152. https://doi.org/10.1016/j.mineng.2017.03.007
Ahmad R, Haseeb S (2015) Absorptive removal of Pb2+, Cu2+ and Ni2+ from the aqueous solution by using groundnut husk modified with guar gum (GG): kinetic and thermodynamic studies, Groundw. Sustain Dev 1:41–49. https://doi.org/10.1016/j.gsd.2015.11.001
Mudgil D, Barak S, Khatkar BS (2014) Guar gum: processing, properties and food applications - a review. J Food Sci Technol 51:409–418. https://doi.org/10.1007/s13197-011-0522-x
Pathania D, Katwal R, Sharma G, Naushad M, Khan MR, Al-Muhtaseb AH (2016) Novel guar gum/Al2O3 nanocomposite as an effective photocatalyst for the degradation of malachite green dye. Int J Biol Macromol 87:366–374. https://doi.org/10.1016/j.ijbiomac.2016.02.073
Hattori K, Abe E, Yoshida T, Cuculo JA (2004) New solvents for cellulose. II. Ethylenediamine/thiocyanate salt system. Polym J 36:123–130. https://doi.org/10.1295/polymj.36.123
Rachtanapun P (2009) Blended films of carboxymethyl cellulose from papaya peel (CMCp) and corn starch, Kasetsart J. -. Nat Sci 43:259–266
Lakshmi DS, Trivedi N, Reddy CRK (2017) Synthesis and characterization of seaweed cellulose derived carboxymethyl cellulose. Carbohydr Polym 157:1604–1610. https://doi.org/10.1016/j.carbpol.2016.11.042
Yang Z, Peng H, Wang W, Liu T (2010) Crystallization behavior of poly(ε-caprolactone)/layered double hydroxide nanocomposites. J Appl Polym Sci 116:2658–2667. https://doi.org/10.1002/app
Tijsen CJ, Kolk HJ, Stamhuis EJ, Beenackers AACM (2001) An experimental study on the carboxymethylation of granular potato starch in non-aqueous media. Carbohydr Polym 45:219–226. https://doi.org/10.1016/S0144-8617(00)00243-5
Kong Q, Wang X, Lou T (2020) Preparation of millimeter-sized chitosan/carboxymethyl cellulose hollow capsule and its dye adsorption properties. Carbohydr Polym 244:116481. https://doi.org/10.1016/j.carbpol.2020.116481
Bono A, Ying PH, Yan FY, Muei CL, Sarbatly R, Krishnaiah D (2009) Synthesis and characterization of carboxymethyl cellulose from palm kernel cake. Adv Nat Appl Sci 3:5–11
Keilany Z (1978) Book Reviews. Rev Soc Econ 36:228–229. https://doi.org/10.1080/00346767800000037
Sharma R, Kalia S, Kaith BS, Srivastava MK (2016) Synthesis of guar gum-acrylic acid graft copolymers based biodegradable adsorbents for cationic dye removal. Int J Plast Technol 20:294–314. https://doi.org/10.1007/s12588-016-9156-1
Sharma G, Kumar A, Sharma S, Naushad M, Ghfar AA, Al-Muhtaseb AH, Ahamad T, Sharma N, Stadler FJ (2020) Carboxymethyl cellulose structured nano-adsorbent for removal of methyl violet from aqueous solution: isotherm and kinetic analyses. Cellulose 27:3677–3691. https://doi.org/10.1007/s10570-020-02989-y
Sabarish R, Unnikrishnan G (2018) Polyvinyl alcohol/carboxymethyl cellulose/ZSM-5 zeolite biocomposite membranes for dye adsorption applications. Carbohydr Polym 199:129–140. https://doi.org/10.1016/j.carbpol.2018.06.123
Lv Y, Xing B, Zheng M, Yi G, Huang G, Zhang C, Yuan R, Chen Z, Cao Y (2018) Hydrothermal synthesis of ultra-light coal-based graphene oxide aerogel for efficient removal of dyes from aqueous solutions. Nanomaterials 8:1–17. https://doi.org/10.3390/nano8090670
Hoseinzadeh H, Hayati B, Shahmoradi F, Seifpanahi-shabani K, Mohammad N (2021) Development of room temperature synthesized and functionalized metal-organic framework/graphene oxide composite and pollutant adsorption ability. Mater Res Bull 142:111408. https://doi.org/10.1016/j.materresbull.2021.111408
Huang D, Xin Q, Ni Y, Shuai Y, Wang S, Li Y, Ye H, Lin L, Ding X, Zhang Y (2018) Synergistic effects of zeolite imidazole framework@graphene oxide composites in humidified mixed matrix membranes on CO2 separation. RSC Adv 8:6099–6109. https://doi.org/10.1039/c7ra09794h
Rafi M, Samiey B, Cheng CH (2018) Study of adsorption mechanism of congo red on graphene oxide/PAMAM nanocomposite. Materials (Basel) 11:1–24. https://doi.org/10.3390/ma11040496
Kumar B, Priyadarshi R, Sauraj, Deeba F, Kulshreshtha A, Gaikwad KK, Kim J, Kumar A, Negi YS (2020) Nanoporous sodium carboxymethyl cellulose-g-poly (Sodium acrylate)/fecl3 hydrogel beads: synthesis and characterization. Gels 6:1–11. https://doi.org/10.3390/gels6040049
Kong W, Liu J (2019) Nitrogen-decorated, porous carbons derived from waste cow manure as efficient catalysts for the selective capture and conversion of CO 2. RSC Adv 9:4925–4931. https://doi.org/10.1039/c8ra10497b
Vall M, Hultberg J, Strømme M, Cheung O (2019) Inorganic carbonate composites as potential high temperature CO2 sorbents with enhanced cycle stability. RSC Adv 9:20273–20280. https://doi.org/10.1039/c9ra02843a
Thakur S, Sharma B, Verma A, Chaudhary J, Tamulevicius S, Thakur VK (2018) Recent approaches in guar gum hydrogel synthesis for water purification. Int J Polym Anal Charact 23:621–632. https://doi.org/10.1080/1023666X.2018.1488661
Pathania D, Sharma S, Singh P (2017) Removal of methylene blue by adsorption onto activated carbon developed from Ficus carica bast. Arab J Chem 10:S1445–S1451. https://doi.org/10.1016/j.arabjc.2013.04.021
Xiong G, Wang BB, You LX, Ren BY, He YK, Ding F, Dragutan I, Dragutan V, Sun YG (2019) Hypervalent silicon-based, anionic porous organic polymers with solid microsphere or hollow nanotube morphologies and exceptional capacity for selective adsorption of cationic dyes. J. Mater. Chem. A. 7:393–404. https://doi.org/10.1039/c8ta07109h
Gan Q, Shi W, Xing Y, Hou Y (2018) A polyoxoniobate/g-C3N4 nanoporous material with high adsorption capacity of methylene blue from aqueous solution. Front Chem 6:1–10. https://doi.org/10.3389/fchem.2018.00007
Shen J, Shahid S, Amura I, Sarihan A, Tian M, Emanuelsson EA (2018) Enhanced adsorption of cationic and anionic dyes from aqueous solutions by polyacid doped polyaniline. Synth Met 245:151–159. https://doi.org/10.1016/j.synthmet.2018.08.015
Mahini R, Esmaeili H, Foroutan R (2018) Adsorption of methyl violet from aqueous solution using brown algae Padina sanctae-crucis. Turkish J Biochem 43(6):623–631
Meteku BE, Huang J, Zeng J, Subhan F, Feng F, Zhang Y, Yan Z (2020) Magnetic metal–organic framework composites for environmental monitoring and remediation. Coord Chem Rev 413:213261. https://doi.org/10.1016/j.ccr.2020.213261
Cao X, Wang X, Chen M, Xiao F, Huang Y, Lyu X (2020) Synthesis of nanoscale zeolitic imidazolate framework-8 (ZIF-8) using reverse micro-emulsion for Congo red adsorption. Sep Purif Technol. https://doi.org/10.1016/j.seppur.2020.11806
Arifin NFT, Yusof N, Nordin NAHM, Bilad MR, Jaafar J, Ismail AF, Salleh WNW (2021) Comparison of different activated agents on biomass-derived graphene towards the hybrid nanocomposites with zeolitic imidazolate framework-8 for room temperature hydrogen storage. J Environ Chem Eng 9(2):105118. https://doi.org/10.1016/j.jece.2021.105118
Chowdhury A, Kumari S, Khan AA, Chandra MR, Hussain S (2021) Activated carbon loaded with Ni-Co-S nanoparticle for superior adsorption capacity of antibiotics and dye from wastewater: kinetics and isotherms. Colloids Surf, A 611:125868. https://doi.org/10.1016/j.colsurfa.2020.125
Nqombolo A, Munonde TS, Makhetha TA, Moutloali RM, Nomngongo PN (2021) Cobalt/zinc based metal organic frameworks as an effective adsorbent for improved removal of As(V) and Cr(VI) in a wide pH range. J Market Res 12:1845–1855. https://doi.org/10.1016/j.jmrt.2021.03.113
Palanisamy PN, Sivakumar P (2009) Kinetic and isotherm studies of the adsorption of Acid Blue 92 using a low-cost non-conventional activated carbon. Desalination 249:388–397. https://doi.org/10.1016/j.desal.2009.09.006
Banerjee D (1993) Experimental techniques in thermal analysis thermogravimetry (TG) & differential scanning calorimetry (DSC). Anal Proc 12:469–508
Wang J, Wang S (2016) Removal of pharmaceuticals and personal care products (PPCPs) from wastewater: a review. J Environ Manag 182:620–640. https://doi.org/10.1016/j.jenvman.2016.07.049
Cai X, Xie Z, Li D, Kassymova M, Zang SQ, Jiang HL (2020) Nano-sized metal-organic frameworks: synthesis and applications. Coord Chem Rev 417:213366. https://doi.org/10.1016/j.ccr.2020.213366
Rey-Mafull CA, Tacoronte JE, Garcia R, Tobella J, Llópiz JC, Iglesias A, Hotza D (2014) Comparative study of the adsorption of acetaminophen on activated carbons in simulated gastric fluid. Springerplus 3:1–12. https://doi.org/10.1186/2193-1801-3-48
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• An eco-friendly adsorbent was synthesized using CMC, GG, and GO.
• Dye (MG) adsorption onto CMC/GG/GO-3 fitted well with pseudo-second-order kinetics and the Langmuir isotherm.
• CMC/GG/GO-3 is the excellent potential candidate for removal of dye (MG) from aqueous solutions.
• Thermodynamic data indicated that the adsorption was a spontaneous and endothermic process.
Rights and permissions
About this article
Cite this article
Naeini, A.H., Kalaee, M.R., Moradi, O. et al. Synthesis, characterization and application of Carboxylmethyl cellulose, Guar gam, and Graphene oxide as novel composite adsorbents for removal of malachite green from aqueous solution. Adv Compos Hybrid Mater 5, 335–349 (2022). https://doi.org/10.1007/s42114-021-00388-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42114-021-00388-w