Abstract
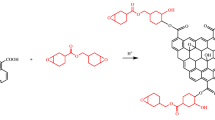
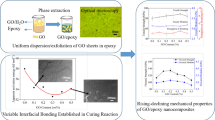
This work investigated the mechanical properties of epoxy resin composites embedded with graphene oxide (GO) using a novel two-phase extraction method. The graphene oxide from water phase was transferred into epoxy resin forming homogeneous suspension. Hyperbranched polyamine-ester (HBPE) anchored graphene oxide (GOHBPE) was prepared by modifying GO with HBPE using a neutralization reaction. Fourier transform-infrared spectroscopy (FTIR), Raman spectroscopy, X-ray diffraction (XRD), and transmission electron microscopy (TEM) showed that the HBPE was successfully grafted to the GO surface. The mechanical properties and dynamic mechanical analysis (DMA) of the composites demonstrated that GOHBPE played a critical role in mechanical reinforcement owing to the layered structure of GO, wrinkled topology, surface roughness and surface area ascending from various oxygen groups of GO itself, and the inarching of HBPE and the reaction among GO, HBPE, and epoxy resin. The transferred GOHBPE/epoxy resin composites showed 69.1% higher impact strength, 129.1% more tensile strength, 45.3% larger modulus, and 70.8% higher strain compared to that of cured neat epoxy resin. The glass transition temperature (Tg) of GOHBPE/epoxy resin composites was increased from 135 to 141 °C and their damping capacity was also improved from 0.71 to 0.91. This study provides guidelines for the fabrication of strengthened polymer composites using phase transfer approach.

ᅟ









Similar content being viewed by others
References
Balandin AA, Ghosh S, Bao W, Calizo I, Teweldebrhan D, Miao F, Lau CN (2008) Superior thermal conductivity of single-layer graphene. Nano Lett 8(3):902–907
Lee C, Wei X, Kysar JW, Hone J (2008) Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 321(5887):385–388
Nair RR, Blake P, Grigorenko AN, Novoselov KS, Booth TJ, Stauber T, Peres NMR, Geim AK (2008) Fine structure constant defines visual transparency of graphene. Science 320(5881):1308
Stauber T, Peres NMR, Geim AK (2008) Optical conductivity of graphene in the visible region of the spectrum. Phys Rev B 78(8):1–8
Geim AK (2009) Graphene: status and prospects. Science 324(5934):1530–1534
Shen W, Wang Y, Yan J, Wu H, Guo S (2015) Enhanced electrochemical performance of lithium iron(II) phosphate modified cooperatively via chemically reduced graphene oxide and polyaniline. Electrochimi Acta 173:310–315
Sun Y, Hu X, Luo W, Xia F, Huang Y (2013) Reconstruction of conformal nanoscale MnO on graphene as a high-capacity and long-life anode material for lithium ion batteries. Adv Funct Mater 23(19):2436–2444
Bonaccorso F, Colombo L, Yu G, Stoller M, Tozzini V, Ferrari AC, Ruoff RS, Pellegrini V (2015) Graphene, related two-dimensional crystals, and hybrid systems for energy conversion and storage. Science. 347(6217):1246501
Wei W, Yang S, Zhou H, Lieberwirth I, Feng X, Muellen K (2013) 3D graphene foams cross-linked with pre-encapsulated Fe3O4 nanospheres for enhanced lithium storage. Adv Mater 25(21):2909–2914
Li G, Xu C (2015) Hydrothermal synthesis of 3D NixCo1-xS2 particles/graphene composite hydrogels for high performance supercapacitors. Carbon 90:44–52
Lee JW, Hall AS, Kim J-D, Mallouk TE (2012) A facile and template-free hydrothermal synthesis of Mn3O4 nanorods on graphene sheets for supercapacitor electrodes with long cycle stability. Chem Mater 24(6):1158–1164
Liu L, Niu Z, Zhang L, Zhou W, Chen X, Xie S (2014) Nanostructured graphene composite papers for highly flexible and foldable supercapacitors. Adv Mater 26(28):4855–4862
Hu W, Peng C, Lv M, Li X, Zhang Y, Chen N, Fan C, Huang Q (2011) Protein corona-mediated mitigation of cytotoxicity of graphene oxide. ACS Nano 5(5):3693–3700
Liao K-H, Lin Y-S, Macosko CW, Haynes CL (2011) Cytotoxicity of graphene oxide and graphene in human erythrocytes and skin fibroblasts. ACS Appl Mater Inte 3(7):2607–2615
Zhang X, Yin J, Peng C, Hu W, Zhu Z, Li W, Fan C, Huang Q (2011) Distribution and biocompatibility studies of graphene oxide in mice after intravenous administration. Carbon 49(3):986–995
Kim H, Abdala AA, Macosko CW (2010) Graphene/polymer nanocomposites. Macromolecules 43(16):6515–6530
Ramanathan T, Abdala AA, Stankovich S, Dikin DA, Herrera-Alonso M, Piner RD, Adamson DH, Schniepp HC, Chen X, Ruoff RS, Nguyen ST, Aksay IA, Prud'Homme RK, Brinson LC (2008) Functionalized graphene sheets for polymer nanocomposites. Nat Nanotechnol 3(6):327–331
Dreyer DR, Park S, Bielawski CW, Ruoff RS (2010) The chemistry of graphene oxide. Chem Soc Rev 39(1):228–240
Novoselov KS, Fal'ko VI, Colombo L, Gellert PR, Schwab MG, Kim K (2012) A roadmap for graphene. Nature 490(7419):192–200
Sun S, Cao Y, Feng J, Wu P (2010) Click chemistry as a route for the immobilization of well-defined polystyrene onto graphene sheets. J Mater Chem 20(27):5605–5607
Yang H, Li F, Shan C, Han D, Zhang Q, Niu L, Ivaska A (2009) Covalent functionalization of chemically converted graphene sheets via silane and its reinforcement. J Mater Chem 19(26):4632–4638
Yang X, Ma L, Wang S, Li Y, Tu Y, Zhu X (2011) “Clicking” graphite oxide sheets with well-defined polystyrenes: a new strategy to control the layer thickness. Polymer 52(14):3046–3052
Lu CH, Yang HH, Zhu CL, Chen X, Chen GN (2009) A graphene platform for sensing biomolecules. Angew Chem Int Edt 48(26):4785–4787
Gu J, Liang C, Zhao X, Gan B, Qiu H, Guo Y, Yang X, Zhang Q, Wang DY (2017) Highly thermally conductive flame-retardant epoxy nanocomposites with reduced ignitability and excellent electrical conductivities. Compos Sci Technol 139:83–89
Li Y, Zhu J, Wei S, Ryu J, Sun L, Guo Z (2011) Poly(propylene)/graphene nanoplatelet nanocomposites: melt rheological behavior and thermal, electrical, and electronic properties. Macromol Chem and Phys 212(18):1951–1959
Zhu J, Wei S, Haldolaarachchige N, He J, Young DP, Guo Z (2012) Very large magnetoresistive graphene disk with negative permittivity. Nanoscale 4(1):152–156
Zhu J, Wei S, Gu H, Rapole SB, Wang Q, Luo Z, Haldolaarachchige N, Young DP, Guo Z (2012) One-pot synthesis of magnetic graphene nanocomposites decorated with core@double-shell nanoparticles for fast chromium removal. Environ Sci Technol 46(2):977–985
Zhu J, Sadu R, Wei S, Chen DH, Haldolaarachchige N, Luo Z, Gomes JA, Young DP, Guo Z (2012) Magnetic graphene nanoplatelet composites toward arsenic removal. ECS J Solid State SC 1(1):M1–M5
Zhu J, Chen M, Qu H, Zhang X, Wei H, Luo Z, Colorado HA, Wei S, Guo Z (2012) Interfacial polymerized polyaniline/graphite oxide nanocomposites toward electrochemical energy storage. Polymer 53(25):5953–5964
Liu H, Li Y, Dai K, Zheng G, Liu C, Shen C, Yan X, Guo J, Guo Z (2016) Electrically conductive thermoplastic elastomer nanocomposites at ultralow graphene loading levels for strain sensor applications. J Mater Chem C 4(1):157–166
Liu H, Huang W, Yang X, Dai K, Zheng G, Liu C, Shen C, Yan X, Guo J, Guo Z (2016) Organic vapor sensing behaviors of conductive thermoplastic polyurethane-graphene nanocomposites. J Mater Chem C 4(20):4459–4469
Gu H, Ma C, Gu J, Guo J, Yan X, Huang J, Zhang Q, Guo Z (2016) An overview of multifunctional epoxy nanocomposites. J Mater Chem C 4(48):5890–5906
Cao Y, Lai Z, Feng J, Wu P (2011) Graphene oxide sheets covalently functionalized with block copolymers via click chemistry as reinforcing fillers. J Mater Chem 21(25):9271–9278
Jiang B, Zhao LW, Guo J, Yan XR, Ding DW, Zhu CC, Huang YD, Guo ZH (2016) Improved thermal stability of methylsilicone resins by compositing with N-doped graphene oxide/Co3O4 nanoparticles. J Nanopart Res 18(6):1–11
Li P, Zheng Y, Yang R, Fan W, Wang N, Zhang A (2015) Flexible nanoscale thread of MnSn(OH)6 crystallite with liquid-like behavior and its application in nanocomposites. ChemPhysChem 16:2524–2529
Lan L, Zheng YP, Zhang AB, Zhang JX, Wang N (2012) Study of ionic solvent-free carbon nanotube nanofluids and its composites with epoxy matrix. J Nanopart Res 14(3):1–10
Verdejo R, Barroso-Bujans F, Rodriguez-Perez MA, de Saja JA, Lopez-Manchado MA (2008) Functionalized graphene sheet filled silicone foam nanocomposites. J Mater Chem 18:2221–2226
Tang J, Zhou H, Liang Y, Shi X, Yang X, Zhang J (2014) Properties of graphene oxide/epoxy resin composites. J Nanomater 696859
Shen XJ, Pei XQ, Fu SY, Friedrich K (2013) Significantly modified tribological performance of epoxy nanocomposites at very low graphene oxide content. Polymer 54(3):1234–1242
Stankovich S, Dikin DA, Dommett GHB, Kohlhaas KM, Zimney EJ, Stach EA, Piner RD, Nguyen ST, Ruoff RS (2006) Graphene-based composite materials. Nature 442(7100):282–286
Yang L, Wang ZQ, Ji YC, Wang JN, Xue G (2014) Highly ordered 3D graphene-based polymer composite materials fabricated by “particle-constructing” method and their outstanding conductivity. Macromolecules 47(5):1749–1756
Yang H, Shan C, Li F, Zhang Q, Han D, Niu L (2009) Convenient preparation of tunably loaded chemically converted graphene oxide/epoxy resin nanocomposites from graphene oxide sheets through two-phase extraction. J Mater Chem 19(46):8856–8860
Zheng YP, Zhang JX, Yang YD, Lan L (2013) The synthesis of hyperbranched poly (amine-ester) and study on the properties of its UV-curing film. J Adhes Sci Technol 27(24):2666–2775
Bao C, Song L, Xing W, Yuan B, Wilkie CA, Huang J, Guo Y, Hu Y (2012) Preparation of graphene by pressurized oxidation and multiplex reduction and its polymer nanocomposites by masterbatch-based melt blending. J Mater Chem 22:6088–6096
Gu H, Zhang H, Ma C, Lyu S, Yao F, Liang C, Yang X, Guo J, Guo Z, Gu J (2017) Polyaniline assisted uniform dispersion for magnetic ultrafine barium ferrite nanorods reinforced epoxy metacomposites with tailorable negative permittivity. J Phys Chem C 121(24):13265–13273
Zhang X, Alloul O, He Q, Zhu J, Verde MJ, Li Y, Wei S, Guo Z (2013) Strengthened magnetic epoxy nanocomposites with protruding nanoparticles on the graphene nanosheets. Polymer 54(14):3594–3604
Lee Y (2017) Mechanical properties of epoxy composites reinforced with ammonia-treated graphene oxides. Carbon Lett 21:1–7
Song P, Cao Z, Cai Y, Zhao L, Fang Z, Fu S (2011) Fabrication of exfoliated graphene-based polypropylene nanocomposites with enhanced mechanical and thermal properties. Polymer 52(18):4001–4010
Gu H, Ma C, Liang C, Meng X, Gu J, Guo Z (2017) Low loading of grafted thermoplastic polystyrene strengthened and toughened transparent epoxy composites. J Mater Chem C 5(17):4275–4285
Jiang D, Xing L, Liu L, Sun S, Zhang Q, Wu Z, Yan X, Guo J, Huang Y, Guo Z (2015) Enhanced mechanical properties and anti-hydrothermal ageing behaviors of unsaturated polyester composites by carbon fibers interfaced with POSS. Compos Sci Technol 117:168–175
Jiang D, Xing L, Liu L, Yan X, Guo J, Zhang X, Zhang Q, Wu Z, Zhao F, Huang Y, Wei S, Guo Z (2014) Interfacially reinforced unsaturated polyester composites by chemically grafting different functional POSS onto carbon fibers. J Mater Chem A 2(43):18293–18303
Guo ZH, Pereira T, Choi O, Wang Y, Hahn HT (2006) Surface functionalized alumina nanoparticle filled polymeric nanocomposites with enhanced mechanical properties. J Mater Chem 16(27):2800–2808
Chen X, Wei S, Yadav A, Patil R, Zhu J, Ximenes R, Sun L, Guo Z (2011) Poly(propylene)/carbon nanofiber nanocomposites: ex situ solvent-assisted preparation and analysis of electrical and electronic properties. Macromol Mater Eng 296(5):434–443
Bafana AP, Yan X, Wei X, Patel M, Guo Z, Wei S, Wujcik EK (2017) Polypropylene nanocomposites reinforced with low weight percent graphene nanoplatelets. Compos Part B-Eng 109:101–107
Lu N, Oza S (2015) A comparative study of the mechanical properties of hemp fiber with virgin and recycled high denisty polyethylene matrix. Composites Part B- Eng 45(1):1651–1656
Lu N, Oza S, Ferguson I (2012) Effect of alkali and silane treatment on the therma stability of hemp fiber as reinforcement in composite structures. Adv Mater Res 415:666–670
Acknowledgements
We gratefully acknowledge the supports from the Priority Academic Program Development of Jiangsu Higher Education Institution; the Key Laboratory Funded by Jiangsu advanced welding technology, National Natural Science Foundation of China (No. 51402132), Jiangsu Provincial Natural Science Foundation of China (Grant No. BK2012279 and No. BK20140505), and US National Science Foundation under grants of CMMI-1560834 and IIP-1700628.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, JX., Liang, YX., Wang, X. et al. Strengthened epoxy resin with hyperbranched polyamine-ester anchored graphene oxide via novel phase transfer approach. Adv Compos Hybrid Mater 1, 300–309 (2018). https://doi.org/10.1007/s42114-017-0007-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42114-017-0007-0