Abstract
Purpose
This study aimed to explore the role and underlying mechanism of circular RNA (circRNA) reticulon 1 (circRTN1) in thyroid cancer (TC).
Methods
The expression levels of circRTN1, microRNA-431-5p (miR-431-5p), and transforming growth factor-alpha (TGFA) mRNA were measured by quantitative real-time PCR (qRT-PCR). Cell proliferation was evaluated using colony formation and 5-ethynyl-2’-deoxyuridine (EdU) assays. Cell apoptosis was analyzed using flow cytometry. Cell migration and invasion were measured using the transwell assay. The protein levels of ki-67, Bax, matrix metalloproteinase 2 (MMP-2), and TGFA were detected using Western blot assay. The interaction between miR-431-5p and circRTN1 or TGFA was verified by dual-luciferase reporter assay and RNA immunoprecipitation (RIP) assay. The effect of circRTN1on TC in vivo was explored via xenograft tumor assay.
Results
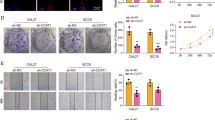
The expression of circRTN1 was increased in TC tissues and cells. Knockdown of circRTN1 suppressed TC cell proliferation, migration, and invasion, and increased cell apoptosis. MiR-431-5p was a target of circRTN1, and miR-431-5p downregulation reversed the role of circRTN1 knockdown in TC cells. TGFA was identified as a direct target of miR-431-5p, and miR-431-5p exerted the anti-tumor role in TC cells by downregulating TGFA. Moreover, circRTN1 sponged miR-431-5p to regulate TGFA expression. Furthermore, circRTN1 knockdown inhibited tumor growth in vivo.
Conclusion
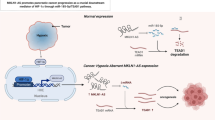
CircRTN1 acted as a cancer-promoting circRNA in TC by regulating the miR-431-5p/TGFA axis, providing a potential therapeutic strategy for TC treatment.
Highlights
-
CircRTN1 was highly expressed in thyroid cancer tissues and cells.
-
CircRTN1 knockdown inhibited thyroid cancer progression by sponging miR-431-5p.
-
MiR-431-5p targeted TGFA to exert the anti-cancer role in thyroid cancer cells.
-
CircRTN1 regulated TGFA expression by acting as a sponge of miR-431-5p.







Similar content being viewed by others
References
Cabanillas ME, McFadden DG, Durante C (2016) Thyroid cancer. Lancet 388:2783–2795
Brito JP, Morris JC, Montori VM (2013) Thyroid cancer: zealous imaging has increased detection and treatment of low risk tumours. BMJ 347:f4706
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249
Nguyen QT, Lee EJ, Huang MG, Park YI, Khullar A, Plodkowski RA (2015) Diagnosis and treatment of patients with thyroid cancer. Am Health Drug Benefits 8:30–40
De Falco V, Guarino V, Avilla E, Castellone MD, Salerno P, Salvatore G et al (2007) Biological role and potential therapeutic targeting of the chemokine receptor CXCR4 in undifferentiated thyroid cancer. Cancer Res 67:11821–11829
Blomberg M, Feldt-Rasmussen U, Andersen KK, Kjaer SK (2012) Thyroid cancer in Denmark 1943–2008, before and after iodine supplementation. Int J Cancer 131:2360–2366
Liu J, Liu T, Wang X, He A (2017) Circles reshaping the RNA world: from waste to treasure. Mol Cancer 16:58
Chen LL (2016) The biogenesis and emerging roles of circular RNAs. Nat Rev Mol Cell Biol 17:205–211
Arnaiz E, Sole C, Manterola L, Iparraguirre L, Otaegui D, Lawrie CH (2019) CircRNAs and cancer: biomarkers and master regulators. Semin Cancer Biol 58:90–99
Vo JN, Cieslik M, Zhang Y, Shukla S, Xiao L, Zhang Y et al (2019) The landscape of circular RNA in cancer. Cell 176:869-881.e813
Kristensen LS, Andersen MS, Stagsted LVW, Ebbesen KK, Hansen TB, Kjems J (2019) The biogenesis, biology and characterization of circular RNAs. Nat Rev Genet 20:675–691
Wang H, Yan X, Zhang H, Zhan X (2019) CircRNA circ_0067934 overexpression correlates with poor prognosis and promotes thyroid carcinoma progression. Med Sci Monit 25:1342–1349
Liu J, Zheng X, Liu H (2020) Hsa_circ_0102272 serves as a prognostic biomarker and regulates proliferation, migration and apoptosis in thyroid cancer. J Gene Med 22:e3209
Bach DH, Lee SK, Sood AK (2019) Circular RNAs in cancer. Mol Ther Nucleic Acids 16:118–129
Panda AC (2018) Circular RNAs act as miRNA sponges. Adv Exp Med Biol 1087:67–79
Kwan JY, Psarianos P, Bruce JP, Yip KW, Liu FF (2016) The complexity of microRNAs in human cancer. J Radiat Res 57(Suppl 1):i106–i111
Jiang Q, Cheng L, Ma D, Zhao Y (2019) FBXL19-AS1 exerts oncogenic function by sponging miR-431–5p to regulate RAF1 expression in lung cancer. Biosci Rep 39:BSR20181804
Sun S, Fu L, Wang G, Wang J, Xu L (2020) MicroRNA-431-5p inhibits the tumorigenesis of osteosarcoma through targeting PANX3. Cancer Manag Res 12:8159–8169
Wen HL, Xu ZM, Wen D, Lin SY, Liang Y, Xie JP (2020) Long noncoding RNAs SET-binding factor 2-antisense RNA1 promotes cell growth through targeting miR-431-5p/CDK14 axis in human papillary thyroid cancer. Kaohsiung J Med Sci 36:808–816
Wu H, Liu Y, Shu XO, Cai Q (2016) MiR-374a suppresses lung adenocarcinoma cell proliferation and invasion by targeting TGFA gene expression. Carcinogenesis 37:567–575
Griffith OL, Melck A, Jones SJ, Wiseman SM (2006) Meta-analysis and meta-review of thyroid cancer gene expression profiling studies identifies important diagnostic biomarkers. J Clin Oncol 24:5043–5051
Vuong HG, Long NP, Anh NH, Nghi TD, Hieu MV, Hung LP et al (2018) Papillary thyroid carcinoma with tall cell features is as aggressive as tall cell variant: a meta-analysis. Endocr Connect 7:R286-r293
Murugan AK, Munirajan AK, Alzahrani AS (2018) Long noncoding RNAs: emerging players in thyroid cancer pathogenesis. Endocr Relat Cancer 25:R59-r82
Patop IL, Kadener S (2018) circRNAs in cancer. Curr Opin Genet Dev 48:121–127
Li Y, Zheng Q, Bao C, Li S, Guo W, Zhao J et al (2015) Circular RNA is enriched and stable in exosomes: a promising biomarker for cancer diagnosis. Cell Res 25:981–984
Shang Q, Yang Z, Jia R, Ge S (2019) The novel roles of circRNAs in human cancer. Mol Cancer 18:6
Xia F, Chen Y, Jiang B, Bai N, Li X (2020) Hsa_circ_0011385 accelerates the progression of thyroid cancer by targeting miR-361-3p. Cancer Cell Int 20:49
Shu T, Yang L, Sun L, Lu J, Zhan X (2020) CircHIPK3 promotes thyroid cancer tumorigenesis and invasion through the Mirna-338–3p/RAB23 axis. Med Princ Pract
Wang M, Chen B, Ru Z, Cong L (2018) CircRNA circ-ITCH suppresses papillary thyroid cancer progression through miR-22-3p/CBL/β-catenin pathway. Biochem Biophys Res Commun 504:283–288
Militello G, Weirick T, John D, Döring C, Dimmeler S, Uchida S (2017) Screening and validation of lncRNAs and circRNAs as miRNA sponges. Brief Bioinform 18:780–788
Huang W, Zeng C, Hu S, Wang L, Liu J (2019) ATG3, a target of miR-431-5p, promotes proliferation and invasion of colon cancer via promoting autophagy. Cancer Manag Res 11:10275–10285
Hu YT, Li XX, Zeng LW (2019) Circ_0001742 promotes tongue squamous cell carcinoma progression via miR-431-5p/ATF3 axis. Eur Rev Med Pharmacol Sci 23:10300–10312
Felekkis K, Touvana E, Stefanou C, Deltas C (2010) microRNAs: a newly described class of encoded molecules that play a role in health and disease. Hippokratia 14:236
Liu Y, Zhang A, Bao PP, Lin L, Wang Y, Wu H et al (2021) MicroRNA-374b inhibits breast cancer progression through regulating CCND1 and TGFA genes. Carcinogenesis 42:528–536
Liu X, Chen L, Tian XD, Zhang T (2017) MiR-137 and its target TGFA modulate cell growth and tumorigenesis of non-small cell lung cancer. Eur Rev Med Pharmacol Sci 21:511–517
Chen S, Sun KX, Liu BL, Zong ZH, Zhao Y (2016) MicroRNA-505 functions as a tumor suppressor in endometrial cancer by targeting TGF-α. Mol Cancer 15:11
Degl’Innocenti D, Alberti C, Castellano G, Greco A, Miranda C, Pierotti MA et al (2010) Integrated ligand-receptor bioinformatic and in vitro functional analysis identifies active TGFA/EGFR signaling loop in papillary thyroid carcinomas. PLoS One 5:e12701
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and informed consent
Written informed consent was obtained from all participants. This study was approved by the Ethics Committee of the First Affiliated Hospital of Anhui Medical University Hefei, Anhui, China.
Conflict of interest
The authors declare no competing interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Supplementary Figure 1. The effects of cabozantinib and TSH on the expression of circRTN1
. (A and B) The expression of circRTN1 was detected by qRT-PCR in TPC-1 cells treated with different doses of cabozantinib and TSH (ANOVA). All data are shown as mean ± SD.
Rights and permissions
About this article
Cite this article
Gao, Y., Wang, Y., Xu, L. et al. CircRTN1 acts as a miR-431-5p sponge to promote thyroid cancer progression by upregulating TGFA. Hormones 21, 611–623 (2022). https://doi.org/10.1007/s42000-022-00378-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42000-022-00378-7