Abstract
Purpose
The effect of exercise on stress has been demonstrated in several studies which have shown that exercise intensity and duration have various effects on the reproductive axis. This study evaluated the effect of different intensities and durations of exercise on the hormonal indices of stress, such as corticosterone (CORT), norepinephrine (NEP), and also reproductive performance indices, including gonadotropin-releasing hormone (GnRH), luteinizing hormone (LH), and testosterone (T).
Methods
In this experimental study, 30 adult Wistar rats were randomly divided into five groups as follows: no-exercise, RME-1 (regular moderate exercise for 1 month), RME-6 (regular moderate exercise for 6 months), RIE-1 (regular intensive exercise for 1 month), and RIE-6 (regular intensive exercise for 6 months). At the end of the experiment, the serum levels of the abovementioned hormones and hypothalamic expression of the Gnrh gene were measured using the enzyme-linked immunosorbent assay and the real-time polymerase chain reaction method, respectively.
Results
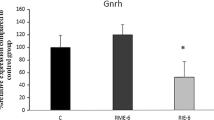
The levels of stress hormones, including CORT and NEP, increased only in the RIE-1 group compared with the no-exercise group. In addition, an increase was observed in T hormone levels in the RME-1 group compared with those in the no-exercise group, whereas LH and T hormone levels showed a greater decrease in the RIE-6 group than in the no-exercise group. Gnrh expression levels showed an increase and a decrease in the RME-1 and RIE-6 groups compared with the no-exercise group, respectively.
Conclusion
These results confirmed the effects of different intensities and durations of exercise on sex hormone levels.





Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available.
References
Cano Sokoloff N, Misra M, Ackerman KE (2016) Exercise, training, and the hypothalamic-pituitary-gonadal axis in men and women. Front Horm Res. 47:27–43
Clark JE (2016) The impact of duration on effectiveness of exercise, the implication for periodization of training and goal setting for individuals who are overfat, a meta-analysis. Biol Sport 33(4):309–333
Pauli SA, Berga SL (2010) Athletic amenorrhea: energy deficit or psychogenic challenge? Ann N Y Acad Sci 1205:33–38
Kumagai H et al (2018) Vigorous physical activity is associated with regular aerobic exercise-induced increased serum testosterone levels in overweight/obese men. Horm Metab Res 50(1):73–79
Steeves JA et al (2016) Cross-sectional association between physical activity and serum testosterone levels in US men: results from NHANES 1999–2004. Andrology 4(3):465–472
Bliss EL, Frischat A, Samuels L (1972) Brain and testicular function. Life Sciences. 11(5, Part 1):231–238
Härkönen M et al (1990) Pituitary and gonadal function during physical exercise in the male rat. J Steroid Biochem 35(1):127–132
Chen JX et al (2007) Influence of acute and chronic treadmill exercise on rat plasma lactate and brain NPY, L-ENK, DYN A1–13. Cell Mol Neurobiol 27(1):1–10
Hill EE et al (2008) Exercise and circulating cortisol levels: the intensity threshold effect. J Endocrinol Invest 31(7):587–591
Anderson T, Wideman L (2017) Exercise and the cortisol awakening response: a systematic review. Sports medicine - open 3(1):37–37
Radahmadi M et al (2015) Effect of forced exercise and exercise withdrawal on memory, serum and hippocampal corticosterone levels in rats. Exp Brain Res 233(10):2789–2799
Szawka RE et al (2013) Release of norepinephrine in the preoptic area activates anteroventral periventricular nucleus neurons and stimulates the surge of luteinizing hormone. Endocrinology 154(1):363–374
Molaei A et al (2020) Synergistic effects of quercetin and regular exercise on the recovery of spatial memory and reduction of parameters of oxidative stress in animal model of Alzheimer’s disease. EXCLI J 8(19):596–612
Shahidi S, et al (2018) Intracerebroventricular microinjection of the 5-HT1F receptor agonist LY 344864 inhibits methamphetamine conditioned place preference reinstatement in rats. Pharmacol Biochem Behav 173(1873–5177 (Electronic)):27–35
Shahidi S, et al (2019) Different doses of methamphetamine alter long-term potentiation, level of BDNF and neuronal apoptosis in the hippocampus of reinstated rats. J Physiol Sci. 69(2)(1880–6562 (Electronic)):409–419
Khajehnasiri N, Khazali H, Sheikhzadeh F (2018) Various responses of male pituitary–gonadal axis to different intensities of long-term exercise: Role of expression of KNDY-related genes. J Biosci 43(4):569–574
Hackney AC (2006) Stress and the neuroendocrine system: the role of exercise as a stressor and modifier of stress. Expert Rev Endocrinol Metab 1(6):783–792
St-Pierre D, Richard D (2020) The Effect of Exercise on the Hypothalamic-Pituitary-Adrenal Axis. In: Hackney AC, Constantini NW (eds) Endocrinology of Physical Activity and Sport. Springer International Publishing, Cham, pp 41–54
Sandlund ES, Norlander T (2000) The effects of Tai Chi Chuan relaxation and exercise on stress responses and well-being: an overview of research. Int J Stress Manag 7(2):139–149
Nabkasorn C et al (2006) Effects of physical exercise on depression, neuroendocrine stress hormones and physiological fitness in adolescent females with depressive symptoms. Eur J Public Health 16(2):179–184
Jiang P et al (2014) The impacts of swimming exercise on hippocampal expression of neurotrophic factors in rats exposed to chronic unpredictable mild stress. Evidence-Based Complementary and Alternative Medicine 2014:729827
Liu W et al (2013) Swimming exercise ameliorates depression-like behavior in chronically stressed rats: relevant to proinflammatory cytokines and IDO activation. Behav Brain Res 242:110–116
Bobbert T et al (2005) Adaptation of the hypothalamic-pituitary hormones during intensive endurance training. Clin Endocrinol (Oxf) 63(5):530–536
Tsatsoulis A, Fountoulakis S (2006) The protective role of exercise on stress system dysregulation and comorbidities. Ann N Y Acad Sci 1083:196–213
Christensen NJ, Galbo H (1983) Sympathetic nervous activity during exercise. Annu Rev Physiol 45:139–153
Greiwe JS et al (1999) Norepinephrine response to exercise at the same relative intensity before and after endurance exercise training. J Appl Physiol (1985) 86(2):531–5
Ostman I, Nybäck H (1976) Adaptive changes in central and peripheral noradrenergic neurons in rats following chronic exercise. Neuroscience 1(1):41–47
Vaamonde D et al (2009) Response of semen parameters to three training modalities. Fertil Steril 92(6):1941–1946
Wang O (2012) Glucocorticoids Regulate Kisspeptin Neurons during Stress and Contribute to Infertility and Obesity in LeptinDeficient Mice. Doctoral dissertation. Harvard University
Arisha AH, Moustafa A (2019) Potential inhibitory effect of swimming exercise on the Kisspeptin-GnRH signaling pathway in male rats. Theriogenology 133:87–96
Bakos HW et al (2011) The effect of paternal diet-induced obesity on sperm function and fertilization in a mouse model. Int J Androl 34(5 Pt 1):402–410
Ghanayem BI et al (2010) Diet-induced obesity in male mice is associated with reduced fertility and potentiation of acrylamide-induced reproductive toxicity. Biol Reprod 82(1):96–104
Alves J, Toro V, Barrientos G, Bartolome I, Munoz D, Maynar, (2020) Hormonal Changes in High-Level Aerobic Male Athletes during a Sports Season. Int. J Environ Res Public Health 17(16):5833
Kujala UM, Alen M, Huhtaniemi IT (1990) Gonadotrophin-releasing hormone and human chorionic gonadotrophin tests reveal that both hypothalamic and testicular endocrine functions are suppressed during acute prolonged physical exercise. Clin Endocrinol (Oxf) 33(2):219–225
Vaamonde D, Garcia-Manso JM, Hackney AC (2017) Impact of physical activity and exercise on male reproductive potential: a new assessment questionnaire. Revista andaluza de medicina del deporte 10(2):79–93
Funding
The authors thank the Shahrekord University of Medical Sciences for the preparation of laboratory equipment.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All animal experimental procedures were performed in accordance with the guidelines for the proper conduct of animal experiments issued by the Ethics Committee of the Shahrekord University of Medical Sciences and performed according to The Guide for Care and Use of Laboratory Animals published by the National Institutes of Health, USA (NIH Publication No. 85–23, revised 1985).
Conflicts of interests
The authors declare no competing interests.
Competing financial interests
The authors declare no competing financial interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khajehnasiri, N., Dehkordi, M.B., Amini-Khoei, H. et al. Effect of exercise intensity and duration on the levels of stress hormones and hypothalamic-pituitary–gonadal axis in adult male rats: an experimental study. Hormones 20, 483–490 (2021). https://doi.org/10.1007/s42000-021-00303-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42000-021-00303-4