Abstract
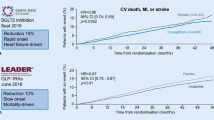
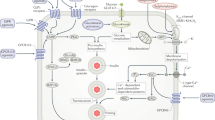
There is today an exponential increase in prevalence of type 2 diabetes mellitus (T2DM), especially in young people. This downward shift in age of onset of T2DM has been shown by abundant evidence to be due to an increase in obesity among the young, the latter mainly attributable to unhealthy dietary habits and a sedentary lifestyle. It is therefore obvious that the prevention of diabetes rather its treatment is of is paramount importance. In the past decade, because concerns about the safety of antidiabetic agents took precedence over the issue of efficacy, almost all studies have been diabetes CVOTs and not traditional CVOTs. Until 2015, the evidence showed that antidiabetic agents are effective in terms of reduction of microvascular, as opposed to macrovascular, complications. However, following publication of the results of some new studies, it became clear that the new class of antidiabetic drugs, e.g., SGLT 2 inhibitors and GLP-1 agonists, are also effective in reducing cardiovascular disease (CVD). In the coming decade, numerous health challenges are expected to arise, the most important being the greater expansion of the therapeutic armamentarium for T2DM and the adoption of strategies for prevention of CVDs. In parallel, the new generation of antidiabetic agents will target the recently investigated pathophysiologic disorders of diabetes, while, ideally, treatments should include smart drugs without side effects.
Similar content being viewed by others
References
Adams F (1990) The extant works of Aretaeus the Cappadocian. Published by Classics of Medicine Library
WHO (2017). http://www.who.int/mediacentre/factsheets/fs312/en/
Selvin E, Wang D, Lee AK et al (2017) Identifying trends in undiagnosed diabetes in U.S. adults by using a confirmatory definition: a cross-sectional study. Ann Intern Med. https://doi.org/10.7326/M17-1272
DeFronzo RA (2009) From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 58(4):773–795. https://doi.org/10.2337/db09-9028
Schwartz SS, Epstein S, Corkey BE et al (2016) The time is right for a new classification system for diabetes: rationale and implications of the β-cell–centric classification schema. Diabetes Care 39(2):179–186
Nissen SE, Wolski K et al (2007) Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med 356:2457–2247. https://doi.org/10.1056/NEJMoa072761
Gerstein HC, Miller ME, Byington RP et al (2008) Effects of intensive glucose lowering in type 2 diabetes. The action to control cardiovascular risk in diabetes study group. N Engl J Med 358(24):2545–2559. https://doi.org/10.1056/NEJMoa0802743
Patel A, MacMahon S, Chalmers J, Neal B et al (2008) Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 358:2560–2572. https://doi.org/10.1056/NEJMoa0802987
Duckworth W, Abraira C, Moritz T et al (2009) Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 360:129–139. https://doi.org/10.1056/NEJMoa080843
Home PD, Pocock SJ, Beck-Nielsen H, Curtis PS et al (2009 Jun 20) Rosiglitazone evaluated for cardiovascular outcomes in oral agent combination therapy for type 2 diabetes (RECORD): a multicentre, randomised, open-label trial. Lancet 373(9681):2125–2135. https://doi.org/10.1016/S0140-6736(09)60953-3
Holman RR, F.R.C.P, Paul SK, Bethel MA et al (2008) 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 359:1577–1589. https://doi.org/10.1056/NEJMoa0806470
Gerstein HC (2016) Cardiovascular Disease and Diabetes Nine-year effects of 3.7 years of intensive glycemic control on cardiovascular outcomes. Diabetes Care 39(5):701–708. https://doi.org/10.2337/dc15-2283
Zoungas S, Chalmers J, Neal B, Billot L et al (2014) Follow-up of blood-pressure lowering and glucose control in type 2 diabetes. N Engl J Med 371:1392–1406. https://doi.org/10.1056/NEJMoa1407963
Hayward RA, Reaven PD, Wiitala WL, for the VADT Investigators et al (2015) Follow-up of glycemic control and cardiovascular outcomes in type 2 diabetes. N Engl J Med 372:2197–2206. https://doi.org/10.1056/NEJMoa1414266
Simpson SH, Lee J, Choi S, Vandermeer B, Abdelmoneim AS, Travis R (2014) Mortality risk among sulfonylureas: a systematic review and network meta-analysis. Lancet Diabetes Endocrinol. https://doi.org/10.1016/S2213-8587(14)70213-X
Bristol-Myers Squibb Company (2006) Bristol-Myers Squibb announces discontinuation of development of muraglitazar, an investigational oral treatment for type 2 diabetes
Guidance for Industry Diabetes Mellitus (2008) Evaluating cardiovascular risk in new antidiabetic therapies to treat type 2 diabetes U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Clinical/Medical
Dormandy JA, Charbonnel B, Eckland DJA et al (2005) Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomised controlled trial. Lancet 366(9493):1279–1289
FDA Drug Safety Communication: Updated FDA review concludes that use of type 2 diabetes medicine pioglitazone may be linked to an increased risk of bladder cancer, 2011. updated 12-12-2016
Erdmann E, Song E, Spanheimer R, van Troostenburg de Bruyn AR, Perez A (2014 Jan) Observational follow-up of the PROactive study: a 6-year update. Diabetes Obes Metab 16(1):63–74. https://doi.org/10.1111/dom.12180
Lewis JD, Habel LA, Quesenberry CP, Strom BL et al (2015) Pioglitazone use and risk of bladder cancer and other common cancers in persons with diabetes. JAMA 314(3):265–277. https://doi.org/10.1001/jama.2015.7996
Lewis JD, Habel LA, Quesenberry CP et al (2015) Pioglitazone use and risk of bladder cancer and other common cancers in persons with diabetes. The new 10-year findings, from three large database analyses. J Am Med Assoc 314(3):265–277. https://doi.org/10.1001/jama.2015.7996
Kernan WN, Viscoli CM, Furie KL et al (2016) Pioglitazone after ischemic stroke or transient ischemic attack. N Engl J Med 374:1321–1331. https://doi.org/10.1056/NEJMoa1506930
Gerstein HC, Bosch J, Dagenais GR et al (2012) Basal insulin and cardiovascular and other outcomes in dysglycemia. N Engl J Med 367:319–328. https://doi.org/10.1056/NEJMoa1203858
Inzucchi SE (2012) Management of hyperglycemia in type 2 diabetes: a patient-centered approach. Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 35(6):1364–1379. https://doi.org/10.2337/dc12-0413
Murugan R (2015) Movement towards personalised medicine in the ICU. Lancet 3(1):10–12
Scirica BM, Bhatt DL, Braunwald E et al (2013) Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med 369:1317–1326. https://doi.org/10.1056/NEJMoa1307684
William B (2013) White, Christopher P. Cannon, Simon R. Heller, et al. Alogliptin after acute coronary syndrome in patients with type 2 diabetes. N Engl J Med 369:1327–1335. https://doi.org/10.1056/NEJMoa1305889
Green JB, Angelyn Bethel M, Armstrong PW (2015) Effect of sitagliptin on cardiovascular outcomes in type 2 diabetes. N Engl J Med 373:232–242J. https://doi.org/10.1056/NEJMoa1501352
Pfeffer MA, Claggett B, Diaz R et al (2015) Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med 373:2247–2257. https://doi.org/10.1056/NEJMoa1509225
Zinman B, Wanner C, Lachin JM et al (2015) Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 373:2117–2128. https://doi.org/10.1056/NEJMoa1504720
Comment on Ferrannini et al. Diabetes Care 2016;39:1108–1114. Comment on Mudaliar et al. Comment on Mudaliar et al. Diabetes Care 2016;39:1115–1122 2016;39:1115–1122
FDA News Release (2016) FDA approves Jardiance to reduce cardiovascular death in adults with type 2 diabetes
Marso SP, Daniels GH, Brown-Frandsen K (2016) Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 375:311–322. https://doi.org/10.1056/NEJMoa1603827
Marso SP, Bain SC, Consoli A et al (2016) Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 375:1834–1844. https://doi.org/10.1056/NEJMoa1607141
Holman RR, F. Med. Sci, Bethel MA, Mentz RJ et al (2017) Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N Engl J Med 377:1228–1239. https://doi.org/10.1056/NEJMoa1612917
Press Releases (2016) Intarcia announces successful cardiovascular safety results in phase FREEDOM-CVO trial for ITCA 650, an investigational therapy for type 2 diabetes
Neal B, Perkovic V, Mahaffey KW et al (2017) Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 377:644–657. https://doi.org/10.1056/NEJMoa1611925
Kosiborod M, Cavender MA, Alex ZF et al (2017) Lower risk of heart failure and death in patients initiated on SGLT-2 inhibitors versus other glucose-lowering drugs: the CVD-REAL Study. Circulation. https://doi.org/10.1161/CIRCULATIONAHA.117.029190
(2017) FDA approves SGLT2 inhibitor ertugliflozin for type 2 diabetes. Medscape
Marso SP, McGuire DK, Zinman B et al (2017) Efficacy and safety of degludec versus glargine in type 2 diabetes. N Engl J Med 377:723–732. https://doi.org/10.1056/NEJMoa1615692
Riddle MC, Bolli GB, Ziemen M, Muehlen-Bartmer I, Bizet F, Home PD (2014) New insulin glargine 300 units/mL versus glargine 100 units/mL in people with type 2 diabetes using basal and mealtime insulin: glucose control and hypoglycemia in a 6-month randomized controlled trial (EDITION 1). Diabetes Care 37(10):2755–2762
Aroda VR, Rosenstock J, Wysham C et al Efficacy and safety of LixiLan, a titratable fixed-ratio combination of insulin glargine plus lixisenatide in type 2 diabetes inadequately controlled on basal insulin and metformin: the LixiLan-L randomized trial. Diabetes Care:dc161495. https://doi.org/10.2337/dc16-1495
Lingvay I, Pérez Manghi F, García-Hernández P et al (2016) Effect of insulin glargine up-titration vs insulin degludec/liraglutide on glycated hemoglobin levels in patients with uncontrolled type 2 diabetes: the DUAL V randomized clinical trial. JAMA 315(9):898–907. https://doi.org/10.1001/jama..1252
DeFronzo RA (2015) Combination of empagliflozin and linagliptin as second-line therapy in subjects with type 2 diabetes inadequately controlled on metformin. Diabetes Care 38(3):384–393
FDA website (2017) FDA approves once-daily QTERN® (dapagliflozin and saxagliptin) tablets for adults with type-2 diabetes
1st ever “biosimilar” insulin approved in US – potential to come cheaper than other insulins, with launch in December 2016 - https://diatribe.org/fda-approves-new-insulin-glargine-basaglar-first-biosimilar-insulin-us#sthash.Soz14Urj.dpuf
ADA (2017) Standards of Medical Care in Diabetes
(2015) Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: the Diabetes Prevention Program Outcomes Study, Vol 3, Issue 11, pp 866–875. https://doi.org/10.1016/S2213-8587(15)00291-0
AACE/ACE comprehensive type 2 diabetes management algorithm 2018
CAROLINA: cardiovascular outcome study of linagliptin versus glimepiride in patients with type 2 diabetes, ClinicalTrials.gov Identifier: NCT01243424
Cardiovascular and renal microvascular outcome study with linagliptin in patients with type 2 diabetes mellitus (CARMELINA), ClinicalTrials.gov Identifier: NCT01897532
Multicenter trial to evaluate the effect of dapagliflozin on the incidence of cardiovascular events (DECLARE-TIMI58), ClinicalTrials.gov Identifier: NCT01730534
Cardiovascular outcomes following ertugliflozin treatment in type 2 diabetes mellitus participants with vascular disease, the VERTIS CV study (MK-8835-004), ClinicalTrials.gov Identifier: NCT01986881
Evaluation of the effects of canagliflozin on renal and cardiovascular outcomes in participants with diabetic nephropathy (CREDENCE) ,ClinicalTrials.gov Identifier: NCT02065791
EMPagliflozin outcomE tRial in patients with chrOnic heaRt failure with reduced ejection fraction (EMPEROR-Reduced), ClinicalTrials.gov Identifier: NCT03057977
Effect of albiglutide, when added to standard blood glucose lowering therapies, on major cardiovascular events in subjects with type 2 diabetes mellitus, ClinicalTrials.gov Identifier: NCT02465515
Researching cardiovascular events with a weekly incretin in diabetes (REWIND) , ClinicalTrials.gov Identifier: NCT01394952
Prato D, Foley JE, Kothny W, Kozlovski P, Stumvoll M, Paldánius PM, Matthews DR (2014) Diabet Med 31(10):1178–1184. https://doi.org/10.1111/dme.12508
Nathan DM, Buse JB, Kahn SE, Krause-Steinrauf H et al (2013) Rationale and design of the glycemia reduction approaches in diabetes: a comparative effectiveness study (GRADE). Diabetes Care 36(8):2254–2261. https://doi.org/10.2337/dc13-0356
RISE Consortium (2014) Restoring Insulin Secretion (RISE): design of studies of β-cell preservation in prediabetes and early type 2 diabetes across the life span. Diabetes Care 37(3):780–788. https://doi.org/10.2337/dc13-1879
Davies M, Pieber TR, Hartoft-Nielsen M-L et al (2017) Effect of oral semaglutide compared with placebo and subcutaneous semaglutide on glycemic control in patients with type 2 diabetes: a randomized clinical trial. JAMA 318(15):1460–1470. https://doi.org/10.1001/jama.14752
Zhen G, Aimetti A, Zhang Y et al (2013) Dang Injectable nano-network for glucose-mediated insulin delivery. ACS Nano. https://doi.org/10.1021/nn400630x
Wang C, Ye Y, Sun W et al (2017) Red blood cells for glucose-responsive insulin delivery. Adv Mater. https://doi.org/10.1002/adma.201606617
Ang K, Tamborlane WV, Weinzimer SA (2015) Combining glucose monitoring and insulin delivery into a single device: current progress and ongoing challenges of the artificial pancreas. Expert Opin Drug Deliv 12(10):1579–1582. https://doi.org/10.1517/17425247.2015.1074174
(2015) The future of diabetes management. The Medical Futurist℠ Newsletter
Singh R, Lather V, Pandita D et al (2017) Synthesis, docking and antidiabetic activity of some newer benzamide derivatives as potential glucokinase activators. Lett Drug Design Discov 14(5):540–553 (14)
Jiang C et al (2017) Dibenzazepine-loaded nanoparticles induce local browning of white adipose tissue to counteract obesity. Mol Ther. https://doi.org/10.1016/j.ymthe.2017.05.020
Zhang S, Liu H, Chuang CL et al (2014) The pathogenic mechanism of diabetes varies with the degree of overexpression and oligomerization of human amylin in the pancreatic islet cells. FASEB J. https://doi.org/10.1096/fj.14-251744
Tsai S, Clemente-Casares X, Revelo XS et al (2015) Are obesity-related insulin resistance and type 2 diabetes autoimmune diseases? Diabetes Care 64(6):1886–1897. https://doi.org/10.2337/db14-1488
Muñoz-Garach A, Diaz-Perdigones C, Tinahones FJ et al (2016) Gut microbiota and type 2 diabetes mellitus. Endocrinol Nutr 63(10):560–568. https://doi.org/10.1016/j.endonu.2016.07.008
Chen L, Tuo B, Dong H (2016) Regulation of intestinal glucose absorption by ion channels and transporters. Nutrients 8(1):43. https://doi.org/10.3390/nu8010043
Cappon G, Acciaroli G, Vettoretti M, Facchinetti A, Sparacino G (2017) Wearable continuous glucose monitoring sensors: a revolution in diabetes treatment. Electronics 6(3):65. https://doi.org/10.3390/electronics6030065
Lean MEJ, Leslie WS, Barnes AC (2017) Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet. https://doi.org/10.1016/S0140-6736(17)33102-1
Pi-Sunyer X (2014) The Look AHEAD Trial: a review and discussion of its outcomes. Curr Nutr Rep 3(4):387–391. https://doi.org/10.1007/s13668-014-0099-x
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Haddad, J.A., Haddad, A.N. The past decade in type 2 diabetes and future challenges. Hormones 17, 451–459 (2018). https://doi.org/10.1007/s42000-018-0080-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42000-018-0080-y