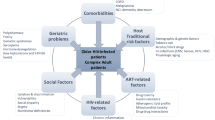
Abstract
Polypharmacy is a well-described problem in the geriatric population. It is a relatively new problem for people living with HIV (PLWH), as this group now has a life expectancy approaching that of the general population. Defining polypharmacy for PLWH is difficult, since the most common traditional definition of at least five medications would encompass a large percentage of PLWH who are on antiretrovirals (ARVs) and medications for other medical comorbidities. Even when excluding ARVs, the prevalence of polypharmacy in PLWH is higher than the general population, and not just in resource-rich countries. Using a more nuanced approach with “appropriate” or “safer” polypharmacy allows for a better framework for discussing how to mitigate the associated risks. Some of the consequences of polypharmacy include adverse effects of medications such as increased risk of geriatric syndromes, drug–drug interactions, decreased adherence, and over- and undertreatment of medical comorbidities. Interventions to combat polypharmacy include decreasing pill burden—specifically with fixed-dose combination tablets—and medication reconciliation/de-prescription using established criteria. The goal of these interventions is to decrease drug interactions and improve quality of life and outcomes. Some special populations of interest within the community of PLWH include those with chronic pain, substance abuse, or requiring end of life care. A final look into the future of antiretroviral therapy shows the promise of possible two-drug regimens, which can help reduce the above risks of polypharmacy.
Similar content being viewed by others
Change history
20 May 2019
The original version of this article unfortunately contained a mistake.
References
UNAIDS UNAIDS—AIDSinfo. http://aidsinfo.unaids.org/. Accessed 8 Oct 2018
CDC HIV among people aged 50 and older. https://www.cdc.gov/hiv/group/age/olderamericans/index.html. Accessed 29 Sept 2018
Antiretroviral Therapy Cohort Collaboration (2017) Survival of HIV-positive patients starting antiretroviral therapy between 1996 and 2013: a collaborative analysis of cohort studies. Lancet HIV 4:e349–e356. https://doi.org/10.1016/S2352-3018(17)30066-8
Duerden M, Avery T, Payne R (2013) Polypharmacy and medicines optimisation. Making it safe and sound. The King’s Fund, London. https://www.kingsfund.org.uk/sites/default/files/field/field_publication_file/polypharmacy-and-medicinesoptimisation-kingsfund-nov13.pdf. Accessed 12 Sept 2018
Masnoon N, Shakib S, Kalisch-Ellett L, Caughey GE (2017) What is polypharmacy? A systematic review of definitions. BMC Geriatr 17:230. https://doi.org/10.1186/s12877-017-0621-2
Whitman AM, DeGregory KA, Morris AL, Ramsdale EE (2016) A comprehensive look at polypharmacy and medication screening tools for the older cancer patient. Oncologist 21:723–730. https://doi.org/10.1634/theoncologist.2015-0492
Bushardt RL, Massey EB, Simpson TW et al (2008) Polypharmacy: misleading, but manageable. Clin Interv Aging 3:383–389
Burt J, Elmore N, Campbell SM et al (2018) Developing a measure of polypharmacy appropriateness in primary care: systematic review and expert consensus study. BMC Med 16:91. https://doi.org/10.1186/s12916-018-1078-7
Hanlon JT, Schmader KE (2013) The medication appropriateness index at 20: where it started, where it has been, and where it may be going. Drugs Aging 30:893–900. https://doi.org/10.1007/s40266-013-0118-4
By the American Geriatrics Society 2015 Beers Criteria Update Expert Panel (2015) American geriatrics society 2015 updated beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc 63:2227–2246. https://doi.org/10.1111/jgs.13702
O’Mahony D, O’Sullivan D, Byrne S et al (2015) STOPP/START criteria for potentially inappropriate prescribing in older people: version 2. Age Ageing 44:213–218. https://doi.org/10.1093/ageing/afu145
Payne RA, Abel GA, Avery AJ et al (2014) Is polypharmacy always hazardous? A retrospective cohort analysis using linked electronic health records from primary and secondary care. Br J Clin Pharmacol 77:1073–1082. https://doi.org/10.1111/bcp.12292
Krentz HB, Cosman I, Lee K et al (2012) Pill burden in HIV infection: 20 years of experience. Antivir Ther (Lond) 17:833–840. https://doi.org/10.3851/IMP2076
Hasse B, Ledergerber B, Furrer H et al (2011) Morbidity and aging in HIV-infected persons: the Swiss HIV cohort study. Clin Infect Dis 53:1130–1139. https://doi.org/10.1093/cid/cir626
Guaraldi G, Malagoli A, Calcagno A et al (2018) The increasing burden and complexity of multi-morbidity and polypharmacy in geriatric HIV patients: a cross sectional study of people aged 65–74 years and more than 75 years. BMC Geriatr 18:99. https://doi.org/10.1186/s12877-018-0789-0
Edelman EJ, Gordon KS, Glover J et al (2013) The next therapeutic challenge in HIV: polypharmacy. Drugs Aging 30:613–628. https://doi.org/10.1007/s40266-013-0093-9
Gimeno-Gracia M, Crusells-Canales MJ, Armesto-Gómez FJ et al (2016) Polypharmacy in older adults with human immunodeficiency virus infection compared with the general population. Clin Interv Aging 11:1149–1157. https://doi.org/10.2147/CIA.S108072
Ssonko M, Stanaway F, Mayanja HK et al (2018) Polypharmacy among HIV positive older adults on anti-retroviral therapy attending an urban clinic in Uganda. BMC Geriatr 18:125. https://doi.org/10.1186/s12877-018-0817-0
Liverpool HIV drug interactions. https://www.hiv-druginteractions.org/checker. Accessed 29 Sept 2018
U.S. Department of Health and Human Services (DHHS) Guidelines for the use of antiretroviral agents in adults and adolescents living with HIV. https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv/0. Accessed 8 Oct 2018
Lepik KJ, Yip B, Ulloa AC et al (2018) Adverse drug reactions to integrase strand transfer inhibitors. AIDS 32:903–912. https://doi.org/10.1097/QAD.0000000000001781
Gilead Sciences (2018) Bictegravir/tenofovir alafenamide/emtricitabine [Package Insert]. https://www.gilead.com/~/media/files/pdfs/medicines/hiv/biktarvy/biktarvy_pi.pdf. Accessed 30 Sept 2018
Bedimo R, Maalouf NM, Zhang S et al (2012) Osteoporotic fracture risk associated with cumulative exposure to tenofovir and other antiretroviral agents. AIDS 26:825–831. https://doi.org/10.1097/QAD.0b013e32835192ae
Mills A, Arribas JR, Andrade-Villanueva J et al (2016) Switching from tenofovir disoproxil fumarate to tenofovir alafenamide in antiretroviral regimens for virologically suppressed adults with HIV-1 infection: a randomised, active-controlled, multicentre, open-label, phase 3, non-inferiority study. Lancet Infect Dis 16:43–52. https://doi.org/10.1016/S1473-3099(15)00348-5
Mallal S, Phillips E, Carosi G et al (2008) HLA-B*5701 screening for hypersensitivity to abacavir. N Engl J Med 358:568–579. https://doi.org/10.1056/NEJMoa0706135
Marcus JL, Neugebauer RS, Leyden WA et al (2016) Use of abacavir and risk of cardiovascular disease among HIV-infected individuals. J Acquir Immune Defic Syndr 71:413–419. https://doi.org/10.1097/QAI.0000000000000881
Mollan KR, Smurzynski M, Eron JJ et al (2014) Association between efavirenz as initial therapy for HIV-1 infection and increased risk for suicidal ideation or attempted or completed suicide: an analysis of trial data. Ann Intern Med 161:1–10. https://doi.org/10.7326/M14-0293
Hughes CA, Tseng A, Cooper R (2015) Managing drug interactions in HIV-infected adults with comorbid illness. Can Med Assoc J 187:36–43. https://doi.org/10.1503/cmaj.131626
Nan C, Shaefer M, Urbaityte R et al (2018) Abacavir use and risk for myocardial infarction and cardiovascular events: pooled analysis of data from clinical trials. Open Forum Infect Dis 5:ofy086. https://doi.org/10.1093/ofid/ofy086
Evans-Jones JG, Cottle LE, Back DJ et al (2010) Recognition of risk for clinically significant drug interactions among HIV-infected patients receiving antiretroviral therapy. Clin Infect Dis 50:1419–1421. https://doi.org/10.1086/652149
Greene M, Steinman MA, McNicholl IR, Valcour V (2014) Polypharmacy, drug–drug interactions, and potentially inappropriate medications in older adults with human immunodeficiency virus infection. J Am Geriatr Soc 62:447–453. https://doi.org/10.1111/jgs.12695
Holtzman C, Armon C, Tedaldi E et al (2013) Polypharmacy and risk of antiretroviral drug interactions among the aging HIV-infected population. J Gen Intern Med 28:1302–1310. https://doi.org/10.1007/s11606-013-2449-6
Foisy MM, Yakiwchuk EM, Hughes CA (2008) Induction effects of ritonavir: implications for drug interactions. Ann Pharmacother 42:1048–1059. https://doi.org/10.1345/aph.1K615
Gellad WF, Grenard JL, Marcum ZA (2011) A systematic review of barriers to medication adherence in the elderly: looking beyond cost and regimen complexity. Am J Geriatr Pharmacother 9:11–23. https://doi.org/10.1016/j.amjopharm.2011.02.004
Cantudo-Cuenca MR, Jiménez-Galán R, Almeida-Gonzalez CV, Morillo-Verdugo R (2014) Concurrent use of comedications reduces adherence to antiretroviral therapy among HIV-infected patients. J Manag Care Spec Pharm 20:844–850. https://doi.org/10.18553/jmcp.2014.20.8.844
Prosperi MCF, Fabbiani M, Fanti I et al (2012) Predictors of first-line antiretroviral therapy discontinuation due to drug-related adverse events in HIV-infected patients: a retrospective cohort study. BMC Infect Dis 12:296. https://doi.org/10.1186/1471-2334-12-296
Huang ES, Karter AJ, Danielson KK et al (2010) The association between the number of prescription medications and incident falls in a multi-ethnic population of adult type-2 diabetes patients: the diabetes and aging study. J Gen Intern Med 25:141–146. https://doi.org/10.1007/s11606-009-1179-2
Erlandson KM, Allshouse AA, Jankowski CM et al (2012) Risk factors for falls in HIV-infected persons. J Acquir Immune Defic Syndr 61:484–489. https://doi.org/10.1097/QAI.0b013e3182716e38
Jyrkkä J, Enlund H, Lavikainen P et al (2011) Association of polypharmacy with nutritional status, functional ability and cognitive capacity over a three-year period in an elderly population. Pharmacoepidemiol Drug Saf 20:514–522. https://doi.org/10.1002/pds.2116
Gandhi TK, Weingart SN, Borus J et al (2003) Adverse drug events in ambulatory care. N Engl J Med 348:1556–1564. https://doi.org/10.1056/NEJMsa020703
Schambelan M, Wilson PWF, Yarasheski KE et al (2008) Development of appropriate coronary heart disease risk prediction models in HIV-infected patients. Circulation 118:e48–e53. https://doi.org/10.1161/CIRCULATIONAHA.107.189627
Deeks SG, Phillips AN (2009) HIV infection, antiretroviral treatment, ageing, and non-AIDS related morbidity. BMJ 338:a3172. https://doi.org/10.1136/bmj.a3172
Neuhaus J, Jacobs DR, Baker JV et al (2010) Markers of inflammation, coagulation, and renal function are elevated in adults with HIV infection. J Infect Dis 201:1788–1795. https://doi.org/10.1086/652749
Freiberg MS, Leaf DA, Goulet JL et al (2009) The association between the receipt of lipid lowering therapy and HIV status among veterans who met NCEP/ATP III criteria for the receipt of lipid lowering medication. J Gen Intern Med 24:334–340. https://doi.org/10.1007/s11606-008-0891-7
Guaraldi G, Menozzi M, Zona S et al (2017) Impact of polypharmacy on antiretroviral prescription in people living with HIV. J Antimicrob Chemother 72:511–514. https://doi.org/10.1093/jac/dkw437
Portsmouth SD, Osorio J, McCormick K et al (2005) Better maintained adherence on switching from twice-daily to once-daily therapy for HIV: a 24-week randomized trial of treatment simplification using stavudine prolonged-release capsules. HIV Med 6:185–190. https://doi.org/10.1111/j.1468-1293.2005.00287.x
Airoldi M, Zaccarelli M, Bisi L et al (2010) One-pill once-a-day HAART: a simplification strategy that improves adherence and quality of life of HIV-infected subjects. Patient Prefer Adherence 4:115–125
Fogarty L, Roter D, Larson S et al (2002) Patient adherence to HIV medication regimens: a review of published and abstract reports. Patient Educ Couns 46:93–108. https://doi.org/10.1016/S0738-3991(01)00219-1
Saag MS, Benson CA, Gandhi RT et al (2018) Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2018 recommendations of the international antiviral society-USA panel. JAMA 320:379–396. https://doi.org/10.1001/jama.2018.8431
Blanco JR, Caro AM, Pérez-Cachafeiro S et al (2010) HIV infection and aging. AIDS Rev 12:218–230
Zalma A, von Moltke LL, Granda BW et al (2000) In vitro metabolism of trazodone by CYP3A: inhibition by ketoconazole and human immunodeficiency viral protease inhibitors. Biol Psychiatry 47:655–661
Greenblatt DJ, von Moltke LL, Harmatz JS et al (2003) Short-term exposure to low-dose ritonavir impairs clearance and enhances adverse effects of trazodone. J Clin Pharmacol 43:414–422
Geraci MJ, McCoy SL, Crum PM, Patel RA (2010) Antipsychotic-induced priapism in an HIV patient: a cytochrome P450-mediated drug interaction. Int J Emerg Med 3:81–84. https://doi.org/10.1007/s12245-010-0175-y
Pollack TM, McCoy C, Stead W (2009) Clinically significant adverse events from a drug interaction between quetiapine and atazanavir–ritonavir in two patients. Pharmacotherapy 29:1386–1391. https://doi.org/10.1592/phco.29.11.1386
Hantson P, Di Fazio V, Wallemacq P (2010) Toxicokinetic interaction between quetiapine and antiretroviral therapy following quetiapine overdose. Drug Metab Lett 4:7–8
McNicholl IR, Gandhi M, Hare CB et al (2017) A pharmacist-led program to evaluate and reduce polypharmacy and potentially inappropriate prescribing in older HIV-positive patients. Pharmacotherapy 37:1498–1506. https://doi.org/10.1002/phar.2043
Janssen prezcobix (darunavir and cobicistat) [Package Insert]. http://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/PREZCOBIXpi.pdf. Accessed 30 Sept 2018
SPRINT Research Group, Wright JT, Williamson JD et al (2015) A randomized trial of intensive versus standard blood-pressure control. N Engl J Med 373:2103–2116. https://doi.org/10.1056/nejmoa1511939
American Diabetes Association (2018) 11. Older adults: standards of medical care in diabetes—2018. Diabetes Care 41:S119–S125. https://doi.org/10.2337/dc18-S011
Bruce RD, Merlin J, Lum PJ et al (2017) 2017 HIVMA of IDSA clinical practice guideline for the management of chronic pain in patients living with HIV. Clin Infect Dis 65:e1–e37. https://doi.org/10.1093/cid/cix636
Lawson E, Sabin C, Perry N et al (2015) Is HIV painful? An epidemiologic study of the prevalence and risk factors for pain in HIV-infected patients. Clin J Pain 31:813–819. https://doi.org/10.1097/AJP.0000000000000162
Canan CE, Chander G, Monroe AK et al (2018) High-risk prescription opioid use among people living with HIV. J Acquir Immune Defic Syndr 78:283–290. https://doi.org/10.1097/QAI.0000000000001690
Uebelacker LA, Weisberg RB, Herman DS et al (2015) Chronic pain in HIV-infected patients: relationship to depression, substance use, and mental health and pain treatment. Pain Med 16:1870–1881. https://doi.org/10.1111/pme.12799
Surratt HL, Kurtz SP, Levi-Minzi MA et al (2015) Pain treatment and antiretroviral medication adherence among vulnerable HIV-positive patients. AIDS Patient Care STDS 29:186–192. https://doi.org/10.1089/apc.2014.0104
CDC HIV/AIDS. https://www.cdc.gov/hiv/default.html. Accessed 29 Sept 2018
Cuzin L, Katlama C, Cotte L et al (2017) Ageing with HIV: do comorbidities and polymedication drive treatment optimization? HIV Med 18:395–401. https://doi.org/10.1111/hiv.12441
Gruta C, Goldschmidt R (2017) HIV/AIDS: implications for older adult patients. https://aging.arizona.edu/sites/aging/files/fact-sheet-pdfs/hiv_1.pdf. Accessed 29 Sept 2018
Burgess MJ, Zeuli JD, Kasten MJ (2015) Management of HIV/AIDS in older patients-drug/drug interactions and adherence to antiretroviral therapy. HIV AIDS (Auckl) 7:251–264. https://doi.org/10.2147/HIV.S39655
Silverberg MJ, Ray GT, Saunders K et al (2012) Prescription long-term opioid use in HIV-infected patients. Clin J Pain 28:39–46. https://doi.org/10.1097/AJP.0b013e3182201a0f
Moatti JP, Carrieri MP, Spire B et al (2000) Adherence to HAART in French HIV-infected injecting drug users: the contribution of buprenorphine drug maintenance treatment. The Manif 2000 study group. AIDS 14:151–155
Ruiz M, Armstrong M, Reske T et al (2014) Antiretroviral therapy at the end of life: the experience of an academic HIV clinic. Am J Hosp Palliat Care 31:475–479. https://doi.org/10.1177/1049909113494459
Goodkin K, Kompella S, Kendell SF (2018) End-of-life care and bereavement issues in human immunodeficiency virus—AIDS. Nurs Clin N Am 53:123–135. https://doi.org/10.1016/j.cnur.2017.10.010
Slomka J, Prince-Paul M, Webel A, Daly BJ (2016) Palliative care, hospice, and advance care planning: views of people living with hiv and other chronic conditions. J Assoc Nurses AIDS Care 27:476–484. https://doi.org/10.1016/j.jana.2016.02.003
Rhodes RL, Nazir F, Lopez S et al (2016) Use and predictors of end-of-life care among HIV patients in a safety net health system. J Pain Symptom Manag 51:120–125. https://doi.org/10.1016/j.jpainsymman.2015.08.010
Schwarcz L, Chen M-J, Vittinghoff E et al (2013) Declining incidence of AIDS-defining opportunistic illnesses: results from 16 years of population-based AIDS surveillance. AIDS 27:597–605. https://doi.org/10.1097/QAD.0b013e32835b0fa2
Lima VD, Lourenço L, Yip B et al (2015) AIDS incidence and AIDS-related mortality in British Columbia, Canada, between 1981 and 2013: a retrospective study. Lancet HIV 2:e92–e97. https://doi.org/10.1016/S2352-3018(15)00017-X
Stewart A, Chan Carusone S, To K et al (2012) Causes of death in HIV patients and the evolution of an AIDS hospice: 1988–2008. AIDS Res Treat 2012:390406. https://doi.org/10.1155/2012/390406
Wardlaw LA (1994) Sustaining informal caregivers for persons with AIDS. Fam Soc J Contemp Soc Serv 75:373–384. https://doi.org/10.1177/104438949407500606
Van Deventer C, Wright A (2017) The psychosocial impact of caregiving on the family caregivers of chronically ill AIDS and/or HIV patients in home-based care: a qualitative study in Zimbabwe. South Afr J HIV Med. https://doi.org/10.4102/sajhivmed.v18i1.718
Friedland GH, Williams A (1999) Attaining higher goals in HIV treatment: the central importance of adherence. AIDS 13(Suppl 1):S61–S72
Llibre JM, Hung C-C, Brinson C et al (2018) Efficacy, safety, and tolerability of dolutegravir-rilpivirine for the maintenance of virological suppression in adults with HIV-1: phase 3, randomised, non-inferiority SWORD-1 and SWORD-2 studies. Lancet 391:839–849. https://doi.org/10.1016/S0140-6736(17)33095-7
Aboud M, Orkin C, Podzamczer D, Boger J (2018) Durable suppression 2 years after switch to DTG +RPV 2-drug regimen: SWORD-1 and SWORD-2 studies, 22nd International AIDS Conference, Amsterdam, Netherlands, July 23–27, 2018
ViiV Healthcare (2016) An efficacy, safety, and tolerability study comparing dolutegravir plus lamivudine with dolutegravir plus tenofovir/emtricitabine in treatment naïve HIV infected subjects (Gemini 1). In: NCT02831673. https://clinicaltrials.gov/ct2/show/NCT02831673. Accessed 27 Sept 2018
ViiV Healthcare (2016) An efficacy, safety, and tolerability study comparing dolutegravir (DTG) plus lamivudine (3TC) with dolutegravir plus tenofovir/emtricitabine in treatment naïve HIV infected subjects (Gemini 2). In: NCT02831764. https://clinicaltrials.gov/ct2/show/NCT02831764. Accessed 28 Sept 2018
Acknowledgements
Dr. Johnston receives support from NIH TL1 TR000459 and NIAID T32AI007613. Dr. Siegler receives support from Gilead Sciences, Inc. for an investigator-initiated study IN-US-311-4182.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Siegler receives support from Gilead Sciences for an investigator-initiated study. Dr. Faragon is on the speakers’ bureaus of AbbVie, Gilead Sciences, Merck, and Janssen Pharmaceutical. Drs. Freedman, Johnston, and Del Carmen report no conflicts of interest.
Ethical approval
The preparation of this review did not involve human or animal participants.
Informed consent
No informed consent was required.
Rights and permissions
About this article
Cite this article
Freedman, S.F., Johnston, C., Faragon, J.J. et al. Older HIV-infected adults: complex patients (III)—polypharmacy. Eur Geriatr Med 10, 199–211 (2019). https://doi.org/10.1007/s41999-018-0139-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41999-018-0139-y