Abstract
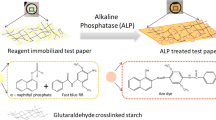
A paper analytical device (PAD) was developed for the colourimetric estimation of alkaline phosphatase (ALP). ALP catalyzes the hydrolysis of disodium phenyl phosphate (DSPP) to phenol, which then undergoes oxidative coupling with 4-aminoantipyrine (4-AAP) producing a reddish brown-coloured quinone imine. The colourimetric reaction tested in solution has been translated onto a PAD for the point of care (PoC) testing of ALP. A uniform colour was obtained in 25 min with the introduction of ALP onto the reagent drop cast paper device. The images obtained by scanning the PAD were processed using ImageJ software. The colour intensity obtained by image processing was proportional to the concentration of ALP present in the sample. ALP in serum and saliva samples were tested using the PAD in the range of 0 to 528 U/L and 0 to 187 U/L, respectively. The interference studies revealed that the PAD was selective to ALP in the presence of other biomolecules. The PAD exhibited a limit of detection (LOD) of 3.3 U/L and a sensitivity of 48.36 a.u/Log(U/L) for serum ALP and LOD of 1.69 U/L and a sensitivity of 28.5 a.u/Log (U/L) for salivary ALP. The PAD showed good agreement with the clinical methods for real samples.





Similar content being viewed by others
Data availability
Data will be made available on request.
References
Shaban SM, Moon BS, Pyun DG, Kim DH. A colorimetric alkaline phosphatase biosensor based on p-aminophenol-mediated growth of silver nanoparticles. Colloids Surf B Biointerfaces. 2021;205:111835.
Sharma U, Pal D, Prasad R. Alkaline phosphatase: an overview. Indian J Clin Biochem. 2014;29(3):269–78.
Liu SG, Han L, Li N, Xiao N, Ju YJ, Li NB, Luo HQ. A fluorescence and colorimetric dual-mode assay of alkaline phosphatase activity: via destroying oxidase-like CoOOH nanoflakes. J Mater Chem B. 2018;6(18):2843–50.
Luo L, Liu J, Liu Y, Chen H, Zhang Y, Liu M, Yao S. In situ formation of fluorescence species for the detection of alkaline phosphatase and organophosphorus pesticide via the ascorbate oxidase mimetic activity of AgPd bimetallic nanoflowers. Food Chem. 2024;430:137062.
Yang Q, Wang X, Peng H, Arabi M, Li J, Xiong H, Choo J, Chen L. Ratiometric fluorescence and colorimetry dual-mode assay based on manganese dioxide nanosheets for visual detection of alkaline phosphatase activity. Sens Actuators B Chem. 2020;302:127176.
Fan S, Jiang X, Yang M, Wang X. Sensitive colorimetric assay for the determination of alkaline phosphatase activity utilizing nanozyme based on copper nanoparticle-modified Prussian blue. Anal Bioanal Chem. 2021;413(15):3955–63.
Hafez E, Moon BS, Shaban SM, Pyun DG, Kim DH. Multicolor diagnosis of salivary alkaline phosphatase triggered by silver-coated gold nanobipyramids. Microchim Acta. 2021;188(12):1–10.
Tang Z, Chen H, He H, Ma C. Assays for alkaline phosphatase activity: progress and prospects. TrAC Trends Anal Chem. 2019;113:32–43.
Zhou X, Wang M, Wang M, Su X. Nanozyme-based detection of alkaline phosphatase. ACS Appl Nano Mater. 2021;4(8):7888–96.
Wang AL, Teng JX, Yang CG, Xu ZR. Rapid and facile electrospray preparation of CsPbBr3@PMMA fluorescent microspheres for fluorescent detection of ALP in biological samples. Colloids Surf A Physicochem Eng Asp. 2022;634:127909.
Xi CY, Zhang M, Jiang L, Chen HY, Lv J, He Y, Hafez ME, Qian RC, Li DW. MOFs-functionalized regenerable SERS sensor based on electrochemistry for pretreatment-free detection of serum alkaline phosphatase activity. Sens Actuators B Chem. 2022;369:132264.
Zhang S, Li R, Liu X, Yang L, Lu Q, Liu M, Li H, Zhang Y, Yao S. A novel multiple signal amplifying immunosensor based on the strategy of in situ-produced electroactive substance by ALP and carbon-based Ag-Au bimetallic as the catalyst and signal enhancer. Biosens Bioelectron. 2017;92:457–64.
Guo J, Liu Y, Zhang L, Pan J, Wang Y, Wang Y, Cai H, Ju H, Lu G. An ascorbic acid-responsive chemo-chromic SERS sensing chip for synergistic dual-modal on-site analysis of alkaline phosphatase. Sens Actuators B Chem. 2022;371:132527.
Liu X, Fan N, Wu L, Wu C, Zhou Y, Li P, Tang B, Li R. Lighting up alkaline phosphatase in drug-induced liver injury using a new chemiluminescence resonance energy transfer nanoprobe. Chem Commun. 2018;54:2023.
Zhu X, Fan L, Wang S, Lei C, Huang Y, Nie Z, Yao S. Phospholipid-tailored titanium carbide nanosheets as a novel fluorescent nanoprobe for activity assay and imaging of phospholipase D. Anal Chem. 2018;90(11):6742–8.
Fang A, Chen H, Li H, Liu M, Zhang Y, Yao S. Glutathione regulation-based dual-functional upconversion sensing-platform for acetylcholinesterase activity and cadmium ions. Biosens Bioelectron. 2017;87:545–51.
Wei YY, Zhang YZ, Song D, Li J, Xu ZR. Alkaline phosphatase-regulated in situ formation of chromogenic probes for multicolor visual sensing of biomarkers. Talanta. 2021;228:122222.
Li Q, Wang Y, Zhu Q, Liu H, Liu J, Meng HM, Li Z. A dual-mode system based on molybdophosphoric heteropoly acid and fluorescent microspheres for the reliable and ultrasensitive detection of alkaline phosphatase. Analyst. 2023;148(6):1259–64.
Hu Q, Zhou B, Dang P, Li L, Kong J, Zhang X. Facile colorimetric assay of alkaline phosphatase activity using Fe(II)-phenanthroline reporter. Anal Chim Acta. 2017;950:170–7.
Wu T, Hou W, Ma Z, Liu M, Liu X, Zhang Y, Yao S. Colorimetric determination of ascorbic acid and the activity of alkaline phosphatase based on the inhibition of the peroxidase-like activity of citric acid-capped Prussian Blue nanocubes. Microchimica Acta. 2019. https://doi.org/10.1007/s00604-018-3224-5.
Wang J, Ni P, Chen C, Jiang Y, Zhang C, Wang B, Cao B, Lu Y. Colorimetric determination of the activity of alkaline phosphatase by exploiting the oxidase-like activity of palladium cube@CeO2 core-shell nanoparticles. Microchimica Acta. 2020. https://doi.org/10.1007/s00604-019-4070-9.
Ding Z, Li Z, Zhao X, Miao Y, Yuan Z, Jiang Y, Lu Y. Self-deposited ultrasmall Ru nanoparticles on carbon nitride with high peroxidase-mimicking activity for the colorimetric detection of alkaline phosphatase. J Colloid Interf Sci. 2023;631:86–95.
Xie X, Wang Y, Zhou X, Chen J, Wang M, Su X. Fe-N-C single-atom nanozymes with peroxidase-like activity for the detection of alkaline phosphatase. Analyst. 2021;146(3):896–903.
Dhara K, Debiprosad RM. Review on nanomaterials-enabled electrochemical sensors for ascorbic acid detection. Anal Biochem. 2019;586:113415.
Hou Y, Lv CC, Guo YL, Ma XH, Liu W, Jin Y, Li BX, Yang M, Yao SY. Recent advances and applications in paper-based devices for point-of-care testing. J Anal Test. 2022;6:247–73.
Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9(7):671–5.
Resmi PE, Stanley J, Kumar S, Soman KP, Ramachandran T, Satheesh Babu TG 2018 Fabrication of paper microfluidics POCT device for the colorimetric assay of alkaline phosphatase. In: INDICON 2018 - 15th IEEE India Council International Conference, 2018. p.1–4
Chen X, Chen J, Zhang HY, Wang FB, Wang FF, Ji XH, He ZK. Colorimetric detection of alkaline phosphatase on microfluidic paper-based analysis devices. Chin J Anal Chem. 2016;44(4):591–6.
Tai W, Yin F, Bi Y, Lin JM, Zhang Q, Wei Y, Hu Q, Yu L. Paper-based sensors for visual detection of alkaline phosphatase and alpha-fetoprotein via the distance readout. Sens Actuators B Chem. 2023;384:133666.
Zhu Y, Tong X, Wei Q, Cai G, Cao Y, Tong C, Shi S, Wang F. 3D origami paper-based ratiometric fluorescent microfluidic device for visual point-of-care detection of alkaline phosphatase and butyrylcholinesterase. Biosens Bioelectron. 2022;196:113691.
Lakshmi Devi A, Resmi PE, Pradeep A, Suneesh PV, Nair BG, Satheesh Babu TG. A paper-based point-of-care testing device for the colourimetric estimation of bilirubin in blood sample. Spectrochim Acta A Mol Biomol Spectrosc. 2023;287(P1):122045.
Resmi PE, Sachin Kumar S, Alageswari D, Suneesh PV, Ramachandran T, Nair BG, Satheesh Babu TG. Development of a paper-based analytical device for the colourimetric detection of alanine transaminase and the application of deep learning for image analysis. Anal Chim Acta. 2021;1188:339158.
Edachana RP, Kumaresan A, Balasubramanian V, Thiagarajan R, Nair BG, Thekkedath Gopalakrishnan SB. Paper-based device for the colorimetric assay of bilirubin based on in-situ formation of gold nanoparticles. Microchimica Acta. 2020. https://doi.org/10.1007/s00604-019-4051-z.
Emerson E, Kelly K. The condensation of aminoantipyrine. vi. a study of the effect of excess base on the reaction of amino-antipyrine with phenolic compounds in the presence of oxidizing agents. J Org Chem. 1948;13(4):532–4.
Kim HY, Lee HJ, Chang SK. Reaction-based colorimetric signaling of Cu2+ ions by oxidative coupling of phenols with 4-aminoantipyrine. Talanta. 2015;132:625–9.
Baş D. Sensitive and reliable paper-based glucose sensing mechanisms with smartphone readout using the: L * a * b * color space. Anal Methods. 2017;9(47):6698–704.
Mahato K, Chandra P. Paper-based miniaturized immunosensor for naked eye ALP detection based on digital image colorimetry integrated with smartphone. Biosens Bioelectron. 2019;128:9–16.
Zhang L, Nie J, Wang H, Yang J, Wang B, Zhang Y, Li J. Instrument-free quantitative detection of alkaline phosphatase using paper-based devices. Anal Methods. 2017;9(22):3375–9.
Acknowledgements
The authors thank the Department of Biotechnology (DBT), Government of India for financial support. Project Sanction order No. 102/IFD/SAN/2238/2016-17 and Project Sanction order No.102/IFD/SAN/1555/2018-2019.
Funding
Department of Biotechnology, Ministry of Science and Technology, India, 102/IFD/SAN/1555/2018-2019, 102/IFD/SAN/2238/2016-17
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests relevant to the content of this article to declare.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Alageswari, D., Lakshmi Devi, A., Resmi, P.E. et al. Paper Analytical Device for the Colourimetric Detection of Alkaline Phosphatase in Serum and Saliva. J. Anal. Test. (2024). https://doi.org/10.1007/s41664-024-00304-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41664-024-00304-3