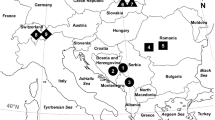
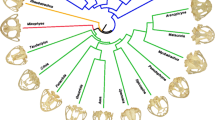
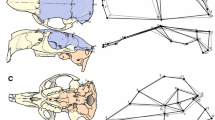
Abstract
Morphological integration refers to the phenotypic interdependence of two or more traits and is estimated by the degree of covariation or correlation among traits at different levels, such as at the intraspecific and evolutionary scales. Intraspecific integration of morphological traits results from the interaction among traits at the genetic, developmental, and functional levels and it has been proposed that it channels morphological evolution by modulating variability. In this work, we test whether the intraspecific integration might have channeled the morphological evolution of the skull roof in a major tetrapod radiation, that of extinct temnospondyl amphibians. To do this, we quantified the patterns of intraspecific integration of different species and explored their relationships with the evolutionary patterns of integration and disparity of three clades of temnospondyls using geometric morphometrics. We recovered that, at the intraspecific level, the integration patterns of the total shape of the skull roof are conserved across the clade and over geological time, but that the integration among individual bones varies in every species considered. We did not find a correlation between the patterns of integration among individual bones at the intraspecific and evolutionary levels, nor between the strength of intraspecific integration of each bone and their respective disparity. These results suggest that the intraspecific integration might have not affected significantly the morphological evolution of the skull roof in temnospondyls over geological time. Thus, it seems that the morphological evolution of this skeletal part might have been driven more by selective pressures than by shared developmental constraints inherited from the temnospondyl ancestor.
Resumen
La integración morfológica refiere a la interdependencia fenotípica de dos o más rasgos y es estimada por el grado de covariación o correlación de dichos rasgos a diferentes niveles, como el intraespecífico y el evolutivo. La integración intraespecífica de rasgos morfológicos resulta de la interacción de caracteres a nivel genético, del desarrollo y funcional y ha sido propuesta como canalizadora de la evolución morfológica al modular la variabilidad. En este trabajo, ponemos a prueba si la integración intraespecífica pudo haber canalizado la evolución morfológica del techo craneano en una de las grandes radiaciones de tetrápodos, la de los anfibios temnospóndilos extintos. Para ello, cuantificamos los patrones de integración intraespecífica de diferentes especies y exploramos su relación con los patrones evolutivos de integración y disparidad en tres clados de temnospóndilos usando morfometría geométrica. Obtuvimos que, a nivel intraespecífico, los patrones de integración de la forma total del techo craneano son conservados dentro del clado y a través del tiempo geológico, pero que la integración entre huesos individuales varía en todas las especies consideradas. No encontramos una correlación entre los patrones de integración entre los huesos individuales a nivel intraespecífico y evolutivo, ni entre la magnitud de la integración intraespecífica de cada hueso y su respectiva disparidad. Estos resultados sugieren que la integración intraespecífica pudo no haber afectado de manera significativa la evolución morfológica del techo craneano de los temnospóndilos a través del tiempo geológico. Por lo tanto, la evolución morfológica de esta parte del esqueleto habría sido dirigida en mayor medida por presiones selectivas que por restricciones del desarrollo heredadas del ancestro temnospóndilo.



Similar content being viewed by others
References
Ackermann, R. R., & Cheverud, J. M. (2000). Phenotypic covariance structure in tamarins (genus Saguinus): A comparison of variation patterns using matrix correlation and common principal component analysis. American Journal of Physical Anthropology: The Official Publication of the American Association of Physical Anthropologists, 111(4), 489–501.
Adams, D. C., & Otárola-Castillo, E. (2013). geomorph: An R package for the collection and analysis of geometric morphometric shape data. Methods in Ecology and Evolution, 4(4), 393–399.
Badyaev, A. V., Foresman, K. R., & Young, R. L. (2005). Evolution of morphological integration: Developmental accommodation of stress-induced variation. The American Naturalist, 166(3), 382–395.
Bell, E., Andres, B., & Goswami, A. (2011). Integration and dissociation of limb elements in flying vertebrates: A comparison of pterosaurs, birds and bats. Journal of Evolutionary Biology, 24(12), 2586–2599.
Bookstein, F. (1991). Morphometric tools for landmark data: Geometry and biology. Cambridge: Cambridge University Press.
Burt, C. (1948). Factor analysis and canonical correlations. British Journal of Statistical Psychology, 1(2), 95–106.
Cheverud, J. M. (1996). Developmental integration and the evolution of pleiotropy. American Zoologist, 36(1), 44–50.
Fröbisch, N. B., & Schoch, R. R. (2009). Testing the impact of miniaturization on phylogeny: Paleozoic dissorophoid amphibians. Systematic Biology, 58(3), 312–327.
Garland, T., Jr., Harvey, P. H., & Ives, A. R. (1992). Procedures for the analysis of comparative data using phylogenetically independent contrasts. Systematic Biology, 41(1), 18–32.
Goswami, A. (2006). Cranial modularity shifts during mammalian evolution. The American Naturalist, 168(2), 270–280.
Goswami, A., Binder, W. J., Meachen, J., & O’Keefe, F. R. (2015). The fossil record of phenotypic integration and modularity: A deep-time perspective on developmental and evolutionary dynamics. Proceedings of the National Academy of Sciences, 112, 4891–4896.
Goswami, A., & Polly, P. D. (2010a). The influence of modularity on cranial morphological disparity in Carnivora and Primates (Mammalia). PLoS One, 5(3), e9517.
Goswami, A., & Polly, P. D. (2010b). Methods for studying morphological integration and modularity. The Paleontological Society Papers, 16, 213–243.
Goswami, A., Polly, P. D., Mock, O. B., & Sánchez-Villagra, M. R. (2012). Shape, variance and integration during craniogenesis: Contrasting marsupial and placental mammals. Journal of Evolutionary Biology, 25(5), 862–872.
Goswami, A., Smaers, J. B., Soligo, C., & Polly, P. D. (2014). The macroevolutionary consequences of phenotypic integration: From development to deep time. Philosophical Transactions of the Royal Society B, 369(1649), 20130254.
Hall, B. K. (2003). Unlocking the black box between genotype and phenotype: Cell condensations as morphogenetic (modular) units. Biology and Philosophy, 18(2), 219–247.
Kelly, E. M., & Sears, K. E. (2011). Reduced integration in marsupial limbs and the implications for mammalian evolution. Biological Journal of the Linnean Society, 102, 22–36.
Klingenberg, C. P. (2008). Morphological integration and developmental modularity. Annual Review of Ecology Evolution and Systematics, 39, 115–132.
Klingenberg, C. P. (2010). Evolution and development of shape: Integrating quantitative approaches. Nature Reviews Genetics, 11(9), 623.
Klingenberg, C. P. (2013). Cranial integration and modularity: Insights into evolution and development from morphometric data. Hystrix, the Italian Journal of Mammalogy, 24(1), 43–58.
Klingenberg, C. P. (2014). Studying morphological integration and modularity at multiple levels: Concepts and analysis. Philosophical Transactions of the Royal Society B, 369(1649), 20130249.
Klingenberg, C. P. (2015). Analyzing fluctuating asymmetry with geometric morphometrics: Concepts, methods, and applications. Symmetry, 7(2), 843–934.
Klingenberg, C. P., & Marugán-Lobón, J. (2013). Evolutionary covariation in geometric morphometric data: Analyzing integration, modularity, and allometry in a phylogenetic context. Systematic Biology, 62(4), 591–610.
Maxwell, E. E., & Dececchi, T. A. (2013). Ontogenetic and stratigraphic influence on observed phenotypic integration in the limb skeleton of a fossil tetrapod. Paleobiology, 39(1), 123–134.
Monteiro, L. R., Bonato, V., & Dos Reis, S. F. (2005). Evolutionary integration and morphological diversification in complex morphological structures: Mandible shape divergence in spiny rats (Rodentia, Echimyidae). Evolution and Development, 7(5), 429–439.
Monteiro, L. R., & Nogueira, M. R. (2009). Adaptive radiations, ecological specialization, and the evolutionary integration of complex morphological structures. Evolution: International Journal of Organic Evolution, 64(3), 724–744.
Pavlicev, M., Cheverud, J. M., & Wagner, G. P. (2009). Measuring morphological integration using eigenvalue variance. Evolutionary Biology, 36(1), 157–170.
Pérez-Ben, C. M., Schoch, R. R., & Báez, A. M. (2018). Miniaturization and morphological evolution in Paleozoic relatives of living amphibians: A quantitative approach. Paleobiology, 44(1), 58–75.
R Core Team (2016). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/. Accessed Mar 2016.
Rohlf, F. J. (2016). TPSDIG2 2.26. Stony Brook: Department of Ecology and Evolution, State University of New York at Stony Brook.
Rohlf, F. J., & Slice, D. (1990). Extensions of the Procrustes method for the optimal superimposition of landmarks. Systematic Biology, 39(1), 40–59.
Schoch, R. R. (1992). Comparative ontogeny of early Permian branchiosaurid amphibians from southwestern Germany. Developmental stages. Palaeontographica Abteilung A, 222, 43–83.
Schoch, R. R. (2008). A new stereospondyl from the German Middle Triassic, and the origin of the Metoposauridae. Zoological Journal of the Linnean Society, 152(1), 79–113.
Schoch, R. R. (2013). The evolution of major temnospondyl clades: An inclusive phylogenetic analysis. Journal of Systematic Palaeontology, 11(6), 673–705.
Schoch, R. R., & Milner, A. R. (2000). Handbook of Paleoherpetology—Temnospondyli II. Munich: Dr. Friedrich Pfeil.
Schoch, R. R., & Milner, A. R. (2014). Handbook of Paleoherpetology—Temnospondyli I. Munich: Dr. Friedrich Pfeil.
Schoch, R. R., & Witzmann, F. (2009a). The temnospondyl Glanochthon from the Lower Permian Meisenheim Formation of Germany. Special Papers in Palaeontology, 81, 121–136.
Schoch, R. R., & Witzmann, F. (2009b). Osteology and relationships of the temnospondyl genus Sclerocephalus. Zoological Journal of the Linnean Society, 157(1), 135–168.
Wagner, G. P., & Altenberg, L. (1996). Perspective: Complex adaptations and the evolution of evolvability. Evolution, 50(3), 967–976.
Willmore, K. E., Young, N. M., & Richtsmeier, J. T. (2007). Phenotypic variability: Its components, measurement and underlying developmental processes. Evolutionary Biology, 34(3–4), 99–120.
Young, N. M., & Hallgrímsson, B. (2005). Serial homology and the evolution of mammalian limb covariation structure. Evolution, 59(12), 2691–2704.
Young, R. L., & Badyaev, A. V. (2006). Evolutionary persistence of phenotypic integration: influence of developmental and functional relationships on complex trait evolution. Evolution, 60(6), 1291–1299.
Zelditch, M. L., Swiderski, D. L., & Sheets, H. D. (2012). Geometric morphometrics for biologists: A primer. New York: Academic Press.
Zelditch, M. L., Wood, A. R., Bonett, R. M., & Swiderski, D. L. (2008). Modularity of the rodent mandible: Integrating bones, muscles, and teeth. Evolution and Development, 10(6), 756–768.
Zelditch, M. L., Wood, A. R., & Swiderski, D. L. (2009). Building developmental integration into functional systems: Function-induced integration of mandibular shape. Evolutionary Biology, 36(1), 71–87.
Acknowledgements
The authors thank U. Göhlich (NHMW), H. Hagdorn (MHI), H. Lutz (NHMM), M. A. Norell (AMNH), K. Padian (UCMP), S. E. Pierce (MCZ), O. W. M. Rauhut (BSM), D. Vasilyan (GPIT), and F. Witzmann (MB) for specimen access. C. P.-B. also thanks R. Schoch and A. M. Báez for fruitful discussions and J. Fortuny, G. Cassini, and M. Ramírez for helpful comments on the doctoral thesis on which this work is based. The article was greatly improved by suggestions of J. Fortuny and two anonymous reviewers. This work was funded by doctoral fellowships from CONICET, DAAD together with the Ministry of Education of Argentina, and the Konrad Lorenz Institute awarded to C.P.-B.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pérez-Ben, C.M., Gómez, R.O. Morphological integration and evolution of the skull roof in temnospondyl amphibians. J Iber Geol 45, 341–351 (2019). https://doi.org/10.1007/s41513-018-0088-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41513-018-0088-9