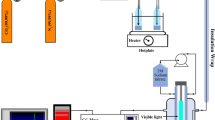
Abstract
The nanocomposite photocatalytic material, TiO2/g-C3N4, was successfully fabricated via spray drying and calcination to reduce CO2 to methanol. XRD, TEM, SEM and BET result shows the coordination of TiO2 nanoparticles with g-C3N4 nanosheets. Band edge calculation of TiO2/g-C3N4 heterostructure depicts type II heterojunction photocatalyst, for enhanced charge separation or reduced charge recombination. Our optimized result suggests the layered g-C3N4 material has close interaction with the TiO2 particles, which fosters the light-harvesting, charge transferring and electron hole pair separation to enhance the photoactivity. Significant enhancement in photo-reduction of CO2 to methanol was observed compared to bare TiO2 and g-C3N4. The yield of methanol over the composite was 31.7 µmol/gcat after 4 h of irradiation, which was 2.3 times more than those over pure C3N4. This work demonstrates the development of an efficient semiconductor composite with charge separation playing a crucial role in CO2 reduction reactions.
Graphic abstract










Similar content being viewed by others
References
Adekoya DO, Tahir M, Amin NAS (2017) g-C3N4/(Cu/TiO2) nanocomposite for enhanced photoreduction of CO2 to CH3OH and HCOOH under UV/visible light. J CO2 Utiliz 18:261–274. https://doi.org/10.1016/j.jcou.2017.02.004
Aguirre ME, Zhou R, Eugene AJ et al (2017) Cu2O/TiO2 heterostructures for CO2 reduction through a direct Z-scheme: protecting Cu2O from photocorrosion. Appl Catal B 217:485–493. https://doi.org/10.1016/j.apcatb.2017.05.058
Ao C, Feng B, Qian S et al (2020) Theoretical study of transition metals supported on g-C3N4 as electrochemical catalysts for CO2 reduction to CH3OH and CH4. J CO2 Utiliz 36:116–123. https://doi.org/10.1016/j.jcou.2019.11.007
Asahi R, Morikawa T, Irie H, Ohwaki T (2014) Nitrogen-doped titanium dioxide as visible-light-sensitive photocatalyst: designs, developments, and prospects. Chem Rev. https://doi.org/10.1021/cr5000738
Basavarajappa PS, Patil SB, Ganganagappa N et al (2020) Recent progress in metal-doped TiO2, non-metal doped/codoped TiO2 and TiO2 nanostructured hybrids for enhanced photocatalysis. Int J Hydrogen Energy 45:7764–7778. https://doi.org/10.1016/j.ijhydene.2019.07.241
Cao S, Li Y, Zhu B et al (2017) Facet effect of Pd cocatalyst on photocatalytic CO2 reduction over g-C3N4. J Catal 349:208–217. https://doi.org/10.1016/j.jcat.2017.02.005
Das S, Pérez-Ramírez J, Gong J et al (2020) Core-shell structured catalysts for thermocatalytic, photocatalytic, and electrocatalytic conversion of CO2. Chem Soc Rev 49:2937–3004
Fischer EM, Knutti R (2015) Anthropogenic contribution to global occurrence of heavy-precipitation and high-temperature extremes. Nat Clim Change 5:560–564. https://doi.org/10.1038/nclimate2617
Fu J, Zhu B, Jiang C et al (2017) Hierarchical porous O-doped g-C3N4 with enhanced photocatalytic CO2 reduction activity. Small. https://doi.org/10.1002/smll.201603938
Fung CM, Tang JY, Tan LL et al (2020) Recent progress in two-dimensional nanomaterials for photocatalytic carbon dioxide transformation into solar fuels. Mater Today Sustain 9:100037
Gao J, Shen Q, Guan R et al (2020) Oxygen vacancy self-doped black TiO2 nanotube arrays by aluminothermic reduction for photocatalytic CO2 reduction under visible light illumination. J CO2 Utiliz 35:205–215. https://doi.org/10.1016/j.jcou.2019.09.016
Goeppert A, Czaun M, Jones JP et al (2014) Recycling of carbon dioxide to methanol and derived products-closing the loop. Chem Soc Rev 43:7995–8048. https://doi.org/10.1039/c4cs00122b
Humayun M, Fu Q, Zheng Z et al (2018) Improved visible-light catalytic activities of novel Au/P-doped g-C3N4 photocatalyst for solar fuel production and mechanism. Appl Catal A 568:139–147. https://doi.org/10.1016/j.apcata.2018.10.007
Kočí K, Obalová L, Matějová L et al (2009) Effect of TiO2 particle size on the photocatalytic reduction of CO2. Appl Catal B 89:494–502. https://doi.org/10.1016/j.apcatb.2009.01.010
Kong L, Mu X, Fan X et al (2018) Site-selected N vacancy of g-C3N4 for photocatalysis and physical mechanism. Appl Mater Today 13:329–338. https://doi.org/10.1016/j.apmt.2018.10.003
Kumar S, Malik T, Sharma D, Ganguli AK (2019) NaNbO3/MoS2 and NaNbO3/BiVO4 core-shell nanostructures for photoelectrochemical hydrogen generation. ACS Appl Nano Mater 2:2651–2662. https://doi.org/10.1021/acsanm.9b00098
Li W, Liang R, Hu A et al (2014) Generation of oxygen vacancies in visible light activated one-dimensional iodine TiO2 photocatalysts. RSC Adv 4:36959–36966. https://doi.org/10.1039/c4ra04768k
Li J, Zhang M, Li Q, Yang J (2017) Enhanced visible light activity on direct contact Z-scheme g-C3N4–TiO2 photocatalyst. Appl Surf Sci 391:184–193. https://doi.org/10.1016/j.apsusc.2016.06.145
Li X, Yu J, Jaroniec M, Chen X (2019) Cocatalysts for selective photoreduction of CO2 into solar fuels. Chem Rev 119:3962–4179
Linsebigler AL, Guangquan Lu, Yates JT (1995) Photocatalysis on TiO2 surfaces: principles, mechanisms, and selected results. Chem Rev 95:735–758. https://doi.org/10.1021/cr00035a013
Makuła P, Pacia M, Macyk W (2018) How to correctly determine the band gap energy of modified semiconductor photocatalysts based on UV-vis spectra. J Phys Chem Lett 9:6814–6817
Maťejová L, Kǒcí K, Troppová I et al (2018) TiO2 and nitrogen doped TiO2 prepared by different methods; on the (micro)structure and photocatalytic activity in CO2 reduction and N2O decomposition. J Nanosci Nanotechnol 18:688–698. https://doi.org/10.1166/jnn.2018.13936
Modak A, Bhaumik A (2016) Surface-exposed Pd nanoparticles supported over nanoporous carbon hollow tubes as an efficient heterogeneous catalyst for the C[sbnd]C bond formation and hydrogenation reactions. J Mol Catal A 425:147–156. https://doi.org/10.1016/j.molcata.2016.09.037
Modak A, Yamanaka K, Goto Y, Inagaki S (2016) Photocatalytic H2 evolution by Pt-loaded 9,9′-spirobifluorene-based conjugated microporous polymers under visible-light irradiation. Bull Chem Soc Jpn 89:887–891. https://doi.org/10.1246/bcsj.20160105
Monshi A, Foroughi MR, Monshi MR (2012) Modified Scherrer equation to estimate more accurately nano-crystallite size using XRD. World J Nano Sci Eng 02:154–160. https://doi.org/10.4236/wjnse.2012.23020
Nethercot AH (1974) Prediction of Fermi energies and photoelectric thresholds based on electronegativity concepts. Phys Rev Lett 33:1088–1091. https://doi.org/10.1103/PhysRevLett.33.1088
Qi Y, Liang Q, Lv R et al (2018) Synthesis and photocatalytic activity of mesoporous g-C3N4/MoS2 hybrid catalysts. Royal Soc Open Sci 5:180187. https://doi.org/10.1098/rsos.180187
Qin H, Guo RT, Liu XY et al (2018) Z-Scheme MoS2/g-C3N4 heterojunction for efficient visible light photocatalytic CO2 reduction. Dalton Trans 47:15155–15163. https://doi.org/10.1039/c8dt02901f
Reli M, Šihor M, Kočí K, et al (2012) Influence of reaction medium on CO2 photocatalytic reduction yields over ZnS-MMT vliv reakčního prostření na výtěžky fotokatalytické redukce CO2 v přítomnosti ZnS-MMT. 34 GeoScience Engineering LVIII
Sagara N, Kamimura S, Tsubota T, Ohno T (2016) Photoelectrochemical CO2 reduction by a p-type boron-doped g-C3N4 electrode under visible light. Appl Catal B 192:193–198. https://doi.org/10.1016/j.apcatb.2016.03.055
Seinfeld JH, Pandis SN, Noone K (1998) Atmospheric chemistry and physics: from air pollution to climate change. Phys Today 51:88–90
Tang J, Zhou W, Guo R et al (2018) Enhancement of photocatalytic performance in CO2 reduction over Mg/g-C3N4 catalysts under visible light irradiation. Catal Commun 107:92–95. https://doi.org/10.1016/j.catcom.2018.01.006
Tang JY, Kong XY, Ng BJ et al (2019) Midgap-state-mediated two-step photoexcitation in nitrogen defect-modified g-C3N4 atomic layers for superior photocatalytic CO2 reduction. Catal Sci Technol 9:2335–2343. https://doi.org/10.1039/c9cy00449a
Wang WH, Himeda Y, Muckerman JT et al (2015) CO2 hydrogenation to formate and methanol as an alternative to photo- and electrochemical CO2 reduction. Chem Rev 115:12936–12973
Wang YL, Tian Y, Lang ZL et al (2018) A highly efficient Z-scheme B-doped g-C3N4/SnS2 photocatalyst for CO2 reduction reaction: a computational study. J Mater Chem A 6:21056–21063. https://doi.org/10.1039/c8ta07352j
Wang K, Fu J, Zheng Y (2019) Insights into photocatalytic CO2 reduction on C3N4: strategy of simultaneous B, K co-doping and enhancement by N vacancies. Appl Catal B 254:270–282. https://doi.org/10.1016/j.apcatb.2019.05.002
Wang Z, Lee H, Chen J et al (2020) Synergistic effects of Pd–Ag bimetals and g-C3N4 photocatalysts for selective and efficient conversion of gaseous CO2. J Power Sources 466:228306. https://doi.org/10.1016/j.jpowsour.2020.228306
Wei ZH, Wang YF, Li YY et al (2018) Enhanced photocatalytic CO2 reduction activity of Z-scheme CdS/BiVO4 nanocomposite with thinner BiVO4 nanosheets. J CO2 Utiliz 28:15–25. https://doi.org/10.1016/j.jcou.2018.09.008
Xia P, Zhu B, Yu J et al (2017) Ultra-thin nanosheet assemblies of graphitic carbon nitride for enhanced photocatalytic CO2 reduction. J Mater Chem A 5:3230–3238. https://doi.org/10.1039/c6ta08310b
Xu Q, Zhang L, Yu J et al (2018) Direct Z-scheme photocatalysts: principles, synthesis, and applications. Mater Today 21:1042–1063
Yang Y, Wu J, Xiao T et al (2019) Urchin-like hierarchical CoZnAl-LDH/RGO/g-C3N4 hybrid as a Z-scheme photocatalyst for efficient and selective CO2 reduction. Appl Catal B 255:117771. https://doi.org/10.1016/j.apcatb.2019.117771
Ye L, Wu D, Chu KH et al (2016) Phosphorylation of g-C3N4 for enhanced photocatalytic CO2 reduction. Chem Eng J 304:376–383. https://doi.org/10.1016/j.cej.2016.06.059
Ye W, Hu J, Hu X et al (2019) Rational construction of Z-scheme CuInS2/Au/g-C3N4 heterostructure: experimental results and theoretical calculation. Chem Cat Chem 11:6372–6383. https://doi.org/10.1002/cctc.201901227
Yuan YP, Ruan LW, Barber J et al (2014) Hetero-nanostructured suspended photocatalysts for solar-to-fuel conversion. Energy Environ Sci 7:3934–3951
Zhang G, Zhu X, Chen D et al (2020a) Hierarchical: Z-scheme g-C3N4/Au/ZnIn2S4 photocatalyst for highly enhanced visible-light photocatalytic nitric oxide removal and carbon dioxide conversion. Environ Sci 7:676–687. https://doi.org/10.1039/c9en01325c
Zhang H, Tang Y, Liu Z et al (2020b) Study on optical properties of alkali metal doped g-C3N4 and their photocatalytic activity for reduction of CO2. Chem Phys Lett 751:137467. https://doi.org/10.1016/j.cplett.2020.137467
Zou W, Deng B, Hu X et al (2018) Crystal-plane-dependent metal oxide-support interaction in CeO2/g-C3N4 for photocatalytic hydrogen evolution. Appl Catal B 238:111–118. https://doi.org/10.1016/j.apcatb.2018.07.022
Acknowledgements
The authors would like to thank Indian Institute of Technology Delhi for financial support. The authors would like to thank Department of Science and Technology, Government of India under Technology Mission Division (Sanction no. TM/EWO/MI/CCUS/19C).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Singh, S., Modak, A. & Pant, K.K. CO2 Reduction to Methanol Using a Conjugated Organic–Inorganic Hybrid TiO2–C3N4 Nano-assembly. Trans Indian Natl. Acad. Eng. 6, 395–404 (2021). https://doi.org/10.1007/s41403-021-00201-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41403-021-00201-6