Abstract
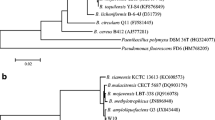
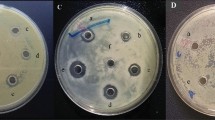
Bacillus species are often regarded as ideal biocontrol agents against plant pathogens, which can produce various antimicrobial peptides (AMPs). In this study, strain BU108 with good antimicrobial activity against Streptomyces scabiei was isolated from the rhizosphere soil of a potato field, which was then identified as Bacillus subtilis through 16S rDNA sequence analysis. An antimicrobial protein was purified by ammonium sulfate precipitation, ion exchange chromatography and ultrafiltration. The active sample showed a single band on SDS-PAGE gel, which was identified by mass spectrometry as partial fragments of gamma-glutamyl transpeptidase (GGT). To verify this new finding, three potential AMPs (KT20, IF20 and AE26) were predicted based on the sequence and structure of GGT, and the antimicrobial activity test confirmed two AMPs (KT20 and IF20). As GGT was involved in the metabolism of glutathione in former references, in this study, we reported the antimicrobial activity of its degradation fragments for the first time, which not only enriched the antimicrobial substance of Bacillus subtilis, but also provided a new choice for potato common scab biocontrol.




Similar content being viewed by others
References
Agrios GN (1997) Plant pathology, 4th edn. Academic, New York
Arrebola E, Jacobs R, Korsten L (2010) Iturin A is the principal inhibitor in the biocontrol activity of Bacillus amyloliquefaciens PPCB004 against postharvest fungal pathogens. J Appl Microbiol 108:386–395
Bindal S, Gupta R (2014) L-theanine synthesis using γ-glutamyl transpeptidase from Bacillus licheniformis ER-15. J Agric Food Chem 62(37):9151–9159
Brogden KA (2005) Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? Nat Rev Microbiol 3:238–250
Brotz H, Bierbaum G, Reynolds PE, Sahl HG (1997) The lantibiotic Mersacidin inhibits peptidoglycan biosynthesis at the level of transglycosylation. Eur J Biochem 246:193–199
Campos MA, Vargas MA, Regueiro V, Llompart CM, Albertí S, Bengoechea JA (2004) Capsule polysaccharide mediates bacterial resistance to antimicrobial peptides. Infect Immun 72:7107–7114
Chen X, Su L, Wu D, Wu J (2014) Application of recombinant Bacillus subtilis γ-glutamyltranspeptidase to the production of l-theanine. Process Biochem 49(9):1429–1439
Combet C, Blanchet C, Geourjon C, Deléage G (2000) Nps@: network protein sequence analysis. Trends Biochem Sci 25:147–150
Han JS, Cheng JH, Yoon TM, Song J, Rajkarnikar A, Kima WG (2010) Biological control agent of common scab disease by antagonistic strain Bacillus sp. sunhua. J Appl Microbiol 99:213–221
Hill J, Lazarovits G (2005) A mail survey of growers to estimate potato common scab prevalence and economic loss in Canada. Can J Plant Pathol 27:46–52
Hooker WJ (1990) Compendium of potato diseases. The American Phytopathological Society, St. Paul
Hu LB, Shi ZQ, Zhang T, Yang ZM (2007) Fengycin antibiotics isolated from B-FS01 culture inhibit the growth of Fusarium moniliforme Sheldon ATCC 38932. FEMS Microbiol Lett 272:91–98
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35(6):1547–1549
Lapwood DH, Wellings LW, Hawkins JH (1973) Irrigation as a practical means to control potato common scab (Streptomyces scabies): final experiment and conclusions. Plant Pathol 22:35–41
Loria R, Bukhalid RA, Fry BA, King RR (1997) Plant pathogenicity in the genus Streptomyces. Plant Dis 81:836–846
Meng QX, Jiang HH, Hanson LE, Hao JJ (2012) Characterizing a novel strain of Bacillus amyloliquefaciens BAC03 for potential biological control application. J Appl Microbiol 113:1165–1175
Moallic C, Dabonne S, Colas B, Sine JP (2006) Identification and characterization of a γ-glutamyltranspeptidase from a thermo-alcalophile strain of Bacillus pumilus. Protein J 25(6):391–397
Neeno-Eckwall EC, Kinkel LL, Schottel JL (2001) Competition and antibiosis in the biological control of potato scab. Can J Microbiol 47:332–340
Nguyen LT, Haney EF, Vogel HJ (2011) The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol 29:464–472
Peypoux F, Bonmatin JM, Wallach J (1999) Recent trends in the biochemistry of Surfactin. Appl Microbiol Biotechnol 51:553–563
Potter HS, Hooker WJ, Cargo W, Stachwick GT (1958) Pentachloronitrobenzene and urea-formaldehyde for potato scab control in Michigan. Plant Dis Rep 43:633–637
Schaad NW, Jones JB, Chun W (2001) Laboratory guide for the identification of plant pathogenic bacteria. The American Phyto-pathological Society, St. Paul
Schmiedeknecht G, Bochow H, Junge H (1998) Use of Bacillus subtilis as biocontrol agent. II. Biological control of potato diseases. Z Pflanzenk Pflanzen 105:376–386
Shuai Y, Zhang T, Mu W, Jiang B (2011) Purification and characterization of γ-glutamyltranspeptidase from Bacillus subtilis SK11.004. J Agric Food Chem 59(11):6233–6238
Spieβ T, Korn SM, Kötter P, Entian KD (2015) Autoinduction specificities of the lantibiotics Subtilin and Nisin. Appl Environ Microbiol 81:7914–7923
Stein T, Borchert S, Conrad B, Feesche J, Hofemeister B, Hofemeister J, Entian KD (2002) Two different lantibiotic-like peptides originate from the Ericin gene cluster of Bacillus subtilis A1/3. J Bacteriol 184:1703–1711
Taniguchi N, Ikeda Y (1998) Gamma-glutamyl transpeptidase: catalytic mechanism and gene expression. Adv Enzymol Relat Areas Mol Biol 72:239–278
Varkey J, Singh S, Nagaraj R (2006) Antibacterial activity of linear peptides spanning the carboxy-terminal beta-sheet domain of arthropod defensins. Peptides 27:2614–2623
Waghu FH, Barai RS, Gurung P, Idicula-Thomas S (2016) CAMPR3: a database on sequences, structures and signatures of antimicrobial peptides. Nucleic Acids Res 44:D1094–D1097
Wang NN, Gao XN, Yan X, Li ZP, Kang ZS, Huang LL, Han QM (2015) Purification, characterization, and heterologous expression of an antifungal protein from the endophytic Bacillus subtilis strain em7 and its activity against Sclerotinia sclerotiorum. Genet Mol Res 14:15488–15504
Wang G, Li X, Wang Z (2016a) APD3: the antimicrobial peptide database as a tool for research and education. Nucleic Acids Res 44:D1087–D1093
Wang NN, Yan X, Gao XN, Niu HJ, Kang ZS, Huang LL (2016b) Purification and characterization of a potential antifungal protein from Bacillus subtilis, e1r-j against Valsa mali. World J Microbiol Biotechnol 32:63
Wu Q, Xu H, Zhang L, Yao J, Ouyang P (2006) Production, purification and properties of γ-glutamyltranspeptidase from a newly isolated Bacillus subtilis NX-2. J Mol Catal B Enzym 43:113–117
Zhao X, Zhao X, Wei Y, Shang Q, Liu Z (2012) Isolation and identification of a novel antifungal protein from a rhizobacterium Bacillus subtilis strain F3. J Phytopathol 161:43–48
Acknowledgements
The authors thank the reviewers, whose comments and suggestions helped us to improve this manuscript.
Funding
This study was financially supported by Heilongjiang Bayi Agricultural University Support Program for San Heng San Zong (ZRCQC201904) and Heilongjiang Bayi Agricultural University Experimental Demonstration Base Project in Jiu San Farm.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest declared.
Human and animal rights statements
This article did not involve human participants and/or animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, Q., Shen, YR. & Yin, KD. The degradation fragments of gamma-glutamyl transpeptidase from Bacillus subtilis BU108 have antimicrobial activity against Streptomyces scabiei. J Plant Dis Prot 128, 279–285 (2021). https://doi.org/10.1007/s41348-020-00371-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41348-020-00371-5