Abstract
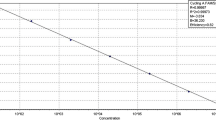
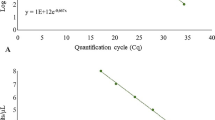
In order to develop a simultaneous and specific molecular detection of the three quarantine Phytophthora pathogens, Phytophthora hibernalis, Phytophthora cambivora and Phytophthora syringae that infect Malus Miller, three pairs of real-time PCR primers (PH-F/PH-R, PC-F/PC-R and PS-F/PS-R) and three probes (PH-Pr, PC-Pr and PS-Pr) labeled with HEX, FAM and ROX, respectively, were designed for P. hibernalis, P. cambivora and P. syringae by alignment analyses of enolase (Enol), ras-like protein Ypt1 and HSP90 gene sequences with other Phytophthora spp. Black Hole Quencher 1 (BHQ1) was used for P. hibernalis and P. cambivora and BHQ2 for P. syringae. Through the optimization of the reaction conditions, a triplex real-time PCR simultaneous detection for the three Phytophthora species infecting Malus Miller was developed. It could achieve simultaneous and specific detection with a sensitivity of 2 × 10−4, 2 × 10−4 and 2 × 10−2 ng/μL genomic DNA, respectively, for P. hibernalis, P. cambivora and P. syringae.



Similar content being viewed by others
References
Böhm J, Hahn A, Schubert R, Bahnweg G, Adler N, Nechwatal J, Oehlmann R, Obszwald W (1999) Real-time quantitative PCR: DNA determination in isolated spores of the Mycorrhizal Fungus Glomus mosseae and monitoring of Phytophthora infestans and Phytophthora citricola in their respective host plants. J Phytopathol 147(7/8):409–416
Chen Y, Roxby R (1996) Characterization of a Phytophthora infestans gene involved in the vesicle transport. Gene 181:89–94
Cubero J, Ayllon MA, Gell I, Melgarejo P, De Cal A, Martín-Sánchez PM, Pérez-Jiménez RM, Soria C, Segundo E, and Larena I. (2009) Detection of Strawberry pathogens by real-time PCR. In: Proceedings of VIth Internat. Strawberry Symposium
Du HZ, Wu PS, Yan J, Zhang QW (2013) Detection of Phytophthora hibernalis by real-time fluorescent PCR. Plant Quar 27(1):36–39
Feng LP, Liu HM, Wang YC, Li Y, Wu XH, Ji Y, Wu PS (2011) SN/T 2759–2011. The Entry-exit Inspection and Quarantine Industry Standards of the People’s Republic of China–Quarantine and Identification of Phytophthora cambivora (Petri) Buisman. China Standards Press, Beijing
Fujita K, Nakazawa N, Fukushima C, Harada Y (1994) Phytophthora fruit rot of apple, Japanese pear and European pear caused by Phytophthora syringae (Kleb). Ann Phytopathol Soc Jpn 6(6):717–724
Gao QF, Qin XY, Fang DH, Mo XH, Yang GH, Chen HR, Cai H (2012) A rapid method to detect Phytophthora nicotianae by PCR. J Yunnan Agric Univ 27(2):156–159
Grote D, Olmos A, Kofoet A, Tuset JJ, Bertolini E, Cambra M (2002) Specific and sensitive detection of Phytophthora nicotianae by simple and nested-PCR. Eur J Plant Pathol 108(3):197–207
Harris DC (1991) The Phytophthora diseases of apple. J Hortic Sci 66(5):513–544
Harris DC, Xu XM (2003) Conditions for infection of apple by Phytophthora syringae. J Phytopathol 151(1):190–194
Ippolito A, Schena L, Nigro F (2004) Real-time detection of Phytophthora nicotianae and P. citrophthora in citrus roots and soil. Eur J Plant Pathol 110:833–843
Judelson HS, Tooley PW (2000) Enhanced polymerase chain reaction methods for detecting and quantifying Phytophthora infestans in plants. Phytopathology 90:1112–1119
Kong P, Hong CX, Richardson PA (2003) Rapid detection of Phytophthora cinnamomi using PCR with primers derived from the Lpv putative storage protein genes. Plant Pathol 52:681–693
Lacourt I, Bonants PJM, Van Gent-Pelzer MP, Cooke DEL, Hagenaar-De Weerdt M, Surplus L, Duncan JM (1997) The use of nested primers in the polymerase chain reaction for the detection of Phytophthora fragariae and P. cactorum in strawberry. Acta Hortic 439:829–838
Liu Y, Zhu L, Li P, Liao F, Ren X, Li G (2015) Triplex-PCR molecular detection of two quarantine fungal diseases of Prunus-Phytophthora syringae and P. cambivora. J Plant Prot 42(4):571–577
Luo JF, Liu YT, Liao F, Hu XH, Liu P, Huang MG (2012) Interception of Phytophthora syringae on citrus fruits imported from California, USA. Mycosystema 31(1):24–30
Matheron ME, Young J, Matejka JC (1988) Phytophthora root and crown rot in apple trees in Arizona. Plant Dis 72(6):481–484
Minerdi D, Moretti M, Li Y, Gaggero L, Garibald A, Gullino ML (2008) Conventional PCR and real-time quantitative PCR detection of Phytophthora cryptogea on Gerbera jamesonii. Eur J Plant Pathol 12(2):227–237
Montenegro D, Aguínl O, Pintos C, Sainz MJ, Mansilla JP (2008) A selective PCR-based method for the identification of Phytophthora hibernalis Carne. Span J Agric Res 6(1):78–84
Qian GZ, Tang GG (2005) A review on the plant taxonomic study on the genus Malus Miller. J Nanjing For Univ 29(3):94–98
Ristaino JB, Madritch M, Trout CL (1998) PCR amplification of ribosomal DNA for species identification in the plant pathogen genus Phytophthora. Appl Environ Microbiol 64(3):948–954
Schena L, Nigro F, Ippolito A (2004) Real-time PCR detection and quantification of Soilborne Fungal pathogens: the case of Rosellinia necatrix, Phytophthora nicotianae, P. citrophthora, and Verticillium dahliae. Phytopathol Mediterr 43:273–280
Schena L, Hughes KJD, David E (2006) Detection and quantification of Phytophthoraramorum, P. kernoviae, P. citricola and P. quercina in symptomatic leaves by multiplex real-time PCR. Mol Plant Pathol 7(5):365–379
Silval C, Duncan JM, Cooke DEL (2005) Development of specific PCR primers for identification and detection of Phytophthora capsici Leon. Eur J Plant Pathol 112(1):43–52
Wang H, Zhu RL, Tan YL, Wei K, Wang XJ, Sun ZH, Sheng PC (2011a) Establishment and application of multiple PCR for diagnosing Proteus mirabilis, Salmonella and Listeria monocytogenes. Sci Agric Sin 44(11):2334–2340
Wang YC, Li Y, Wu PS, Shao XL, Zhang JX, Yu DD, Gan QH, Song T (2011b) SN/T 2756–2011. The Entry-exit Inspection and Quarantine Industry Standards of the People’s Republic of China-Inspection and Identification of Phytophthora syringe (Berk) Kleb. China Standards Press, Beijing
Wilcox WF (1993) Incidence and severity of crown and root rots on four apple rootstocks following exposure to Phytophthora species and waterlogging. J Am Soc Hortic Sci (JASHS) 118(1):63–67
Wu PS, Du HZ, Yan J (2010) SN/T 2617–2010. The Entry-exit Inspection and Quarantine Industry Standards of the People’s Republic of China–Detection and Identification of Phytophthroa hibernalis Carne. China Standards Press, Beijing
Zhang ZG, Li YQ, Fan H, Wang YC, Zheng XB (2006) Molecular detection of Phytophthora capsici in infected plant tissues, soil and water. Plant Pathol 55(6):770–775
Zhang HF, Ren Z, Liu X, Zhang ZG, Wang YC, Wu XH, Zheng XB (2008) Rapid molecular detection of Phytophthora hilbernalis by PCR. Acta Phytopathol Sin 38(3):231–237
Zhuc Linhui, Guo Jingze, Liao Fang, Luo Jiafeng, Huang Guoming, Ren Xueyi, Li Guanrong (2015) Simutaneous triplex PCR detection of two quarantine fungal pathogens of Citrus, Phytoghthora hiberalis and Phytoghthora syringae. J Southwest Univ 37(5):1–8
Acknowledgements
This work was funded by Rapid Identification of Quarantine Fungal Phytophthora Pathogens of Imported Fruits and Seedlings, a Program from The General Administration of Quality Supervision, Inspection and Quarantine of PRC (No: 2012IK286) and Plant Quarantine Pathogen Detection and Application by DNA Barcoding, National “12th Five-Year” Science and Technology Support Program (No: 2012BAK11B02).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical statement
This research did not involve any animal and/or human participants.
Rights and permissions
About this article
Cite this article
Liao, F., Zhang, Y., Zhu, LH. et al. Triplex real-time PCR detection of three quarantine Phytophthora pathogens infecting Malus Miller. J Plant Dis Prot 125, 325–330 (2018). https://doi.org/10.1007/s41348-017-0144-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41348-017-0144-2