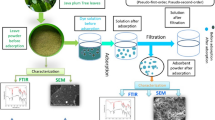
Abstract
This research focuses on developing a supported catalyst by impregnating Moroccan Sebou River sediment with copper (Cu) through a wet impregnation method. Different catalyst loading levels (5, 10, and 15 wt.%) were employed. These catalysts were tested in catalytic wet peroxide oxidation (CWPO) to degrade malachite green (MG) dye using hydrogen peroxide (H2O2) as oxidant. Moreover, the response surface methodology based on the Box–Behnken design (BBD) was used to investigate and optimize the operating parameters affecting the decolorization process, such as temperature, catalytic dose, metal loading, and hydrogen peroxide amount, which were systematically investigated. An MG degradation efficiency of 99.84% was achieved after a reaction time of 60 min under the following conditions: [MG] = 100 mg/L, dose = 0.2 g/L (catalyst 15 wt.%), [H2O2] = 1 mM, and T = 40 °C. The mineralization efficiency reached 74% in terms of chemical oxygen demand (COD) removal. As a result, the solution treated under these optimized conditions showed low toxicity based on the corn grain germination test. Reusability tests on the 15 wt.% catalyst showed a decrease in degradation efficiency by 8.45%, indicating high catalytic stability.
Graphical abstract











Similar content being viewed by others
Data availability
All relevant data are within the paper.
References
Ansari A, Nematollahi D (2018) A comprehensive study on the electrocatalytic degradation, electrochemical behavior and degradation mechanism of malachite green using electrodeposited nanostructured Β-PbO2 electrodes. Water Res 144:462–473. https://doi.org/10.1016/j.watres.2018.07.056
Bassi A, Kanungo K, Koo BH, Hasan I (2023) Cellulose nanocrystals doped silver nanoparticles immobilized agar gum for efficient photocatalytic degradation of malachite green. Int J Biol Macromol 244:125221. https://doi.org/10.1016/j.ijbiomac.2023.125221
Belghiti M, Tanji K, El Mersly L et al (2022) Fast and non-selective photodegradation of basic yellow 28, malachite green, tetracycline, and sulfamethazine using a nanosized ZnO synthesized from zinc ore. React Kinet Mech Catal 135:2265–2278. https://doi.org/10.1007/s11144-022-02232-8
Bhattacharyya S, Algieri C, Davoli M et al (2023) Polymer-based TiO2 membranes: an efficient route for recalcitrant dye degradation. Chem Eng Res Des 193:641–648. https://doi.org/10.1016/J.CHERD.2023.04.018
Candamano S, Coppola G, Mazza A et al (2023) Batch and fixed bed adsorption of methylene blue onto foamed metakaolin-based geopolymer: A. Chem Eng Res Des 197:761–773. https://doi.org/10.1016/j.cherd.2023.08.014
Chithira PR, Theresa John T (2020) Correlation among oxygen vacancy and doping concentration in controlling the properties of cobalt doped ZnO nanoparticles. J Magn Magn Mater 496:165928. https://doi.org/10.1016/j.jmmm.2019.165928
Coppola G, Bhattacharyya S, Pugliese V et al (2023) Metal–organic framework application in wastewater treatment: a review. Euro-Mediterr J Environ Integr. https://doi.org/10.1007/s41207-023-00411-0
Das KC, Dhar SS (2020) Rapid catalytic degradation of malachite green by MgFe2O4 nanoparticles in presence of H2O2. J Alloys Compd 828:154462. https://doi.org/10.1016/j.jallcom.2020.154462
Dra A, Tanji K, Arrahli A et al (2020) Valorization of Oued Sebou Natural Sediments (Fez-Morocco area) as adsorbent of methylene blue dye: kinetic and thermodynamic study. Sci World J 2020:8. https://doi.org/10.1155/2020/2187129
Dra A, Khallouk K, Tanji K et al (2023) Removal of crystal violet dye from aqueous solution using Oued Sebou sediment (Fez-Morocco): Box–Behnken optimization and germination studies. Water Air Soil Pollut 234:1–14. https://doi.org/10.1007/S11270-023-06110-4
Du LN, Wang S, Li G et al (2011) Biodegradation of malachite green by Pseudomonas sp. strain DY1 under aerobic condition: characteristics, degradation products, enzyme analysis and phytotoxicity. Ecotoxicology 20:438–446. https://doi.org/10.1007/s10646-011-0595-3
El Gaidoumi A, BenabdallahChaouni A, Lahrichi A, Kherbeche A (2015) Adsorption du phénol en milieu aqueux par une pyrophyllite marocaine brute et traitée (Adsorption of phenol in aqueous medium by a raw and treated. J Mater Environ Sci 6:2247–2259
El Gaidoumi A, Doña-Rodríguez JM, Pulido Melián E et al (2019a) Catalytic efficiency of Cu-supported pyrophyllite in heterogeneous catalytic oxidation of phenol. Arab J Sci Eng 447(44):6313–6325. https://doi.org/10.1007/S13369-019-03757-2
El Gaidoumi A, Loqman A, Benadallah AC et al (2019b) Co(II)-pyrophyllite as catalyst for phenol oxidative degradation: optimization study using response surface methodology. Waste Biomass Valoriz 10:1043–1051. https://doi.org/10.1007/s12649-017-0117-5
El Gaidoumi A, Arrahli A, Loqman A et al (2022a) Efficient sol–gel nanocomposite TiO2-clay in photodegradation of phenol: comparison to labe-made and commercial photocatalysts. SILICON 14:5401–5414. https://doi.org/10.1007/s12633-021-01275-1
El Gaidoumi A, Tanji K, Loqman A et al (2022b) Cu(II) impregnated clay-derived HS zeolite: synthesis, characterization and catalytic activity on catalytic wet peroxide oxidation (CWPO) of phenol. J Coord Chem 75:19–24. https://doi.org/10.1080/00958972.2022.2154156
El Hajam M, Kandri NI, Harrach A, Zerouale A (2019) Adsorptive removal of brilliant green dye from aqueous solutions using cedar and mahogany sawdusts. Sci Study Res Chem Chem Eng Biotechnol Food Ind 20:395–409
El Hassani AA, Tanji K, El Mrabet I et al (2023) A combined molecular dynamics simulation, DFT calculations, and experimental study of the adsorption of Rhodamine B dye on kaolinite and hydroxyapatite in aqueous solutions. Surf Interfaces 36:102647. https://doi.org/10.1016/j.surfin.2023.102647
El Mrabet I, Ihssane B, Valdés H, Zaitan H (2022) Optimization of Fenton process operating conditions for the treatment of the landfill leachate of Fez city (Morocco). Int J Environ Sci Technol 19:3323–3336. https://doi.org/10.1007/s13762-021-03393-0
Fahoul Y, Tanji K, Zouheir M et al (2022) Novel River Sediment@ZnO–Co nanocomposite for photocatalytic degradation and COD reduction of crystal violet under visible light. J Mol Struct 1253:132298. https://doi.org/10.1016/j.molstruc.2021.132298
Gao G, Zhang A, Zhang M et al (2008) Photocatalytic degradation mechanism of malachite green under visible light irradiation over novel biomimetic photocatalyst HMS-FePcs. Chin J Catal 29:426–430. https://doi.org/10.1016/S1872-2067(08)60043-1
Garcia-Mora AM, Torres-Palma RA, García H et al (2021) Removal of dissolved natural organic matter by the Al/Fe pillared clay-activated-catalytic wet peroxide oxidation: statistical multi-response optimization. J Water Process Eng 39:101755. https://doi.org/10.1016/j.jwpe.2020.101755
Gote YM, Sinhmar PS, Gogate PR (2023) Sonocatalytic degradation of chrysoidine R dye using ultrasonically synthesized NiFe2O4 catalyst. Catalysts 13:597. https://doi.org/10.3390/catal13030597
Hammud HH, Hammoud MH, Hussein AA et al (2023) Removal of malachite green using hydrochar from PALM leaves. Sustainability 15:8939. https://doi.org/10.3390/su15118939
Haounati R, Ighnih H, Ouachtak H et al (2023) Z-scheme g-C3N4/Fe3O4/Ag3PO4 @Sep magnetic nanocomposites as heterojunction photocatalysts for green malachite degradation and dynamic molecular studies. Colloids Surfaces A Physicochem Eng Asp 671:131509. https://doi.org/10.1016/j.colsurfa.2023.131509
Hassan AK, Atiya MA, Mahmoud ZA (2022) Photo-Fenton-like degradation of direct blue 15 using fixed bed reactor containing bimetallic nanoparticles: effects and Box–Behnken optimization. Environ Technol Innov 28:102907. https://doi.org/10.1016/j.eti.2022.102907
Ibrahim SM, Al-Hossainy AF, El-Aal MA (2023) Novel synthesis of nitro-acriflavine dye thin film by kinetics and mechanism of oxidation of acriflavine hydrochloride by permanganate ion: characterization, optical properties and TD-DFT. Surf Interfaces 36:102598. https://doi.org/10.1016/j.surfin.2022.102598
Ju YM, Yang SG, Ding YC et al (2009) Microwave-enhanced H2O2-based process for treating aqueous malachite green solutions: intermediates and degradation mechanism. J Hazard Mater 171:123–132. https://doi.org/10.1016/j.jhazmat.2009.05.120
Kerkez D, Bečelić-Tomin M, Gvoić V et al (2023) Pyrite cinder as an effective Fenton-like catalyst for the degradation of reactive azo dye: effects of process parameters and complete effluent characterization. Catalysts 13:424. https://doi.org/10.3390/catal13020424
Kıpçak İ, KurtaranErsal E (2021) Catalytic wet peroxide oxidation of a real textile azo dye Cibacron Red P-4B over Al/Fe pillared bentonite catalysts: kinetic and thermodynamic studies. React Kinet Mech Catal 132:1003–1023. https://doi.org/10.1007/s11144-021-01962-5
LeovacMaćerak A, KulićMandić A, Pešić V et al (2023) “Green” nZVI-Biochar as Fenton catalyst: perspective of closing-the-loop in wastewater treatment. Molecules 28:1425. https://doi.org/10.3390/molecules28031425
Loqman A, El Bali B, El Gaidoumi A et al (2021) The first application of Moroccan perlite as industrial dyes removal. SILICON 14:2813–2838. https://doi.org/10.1007/S12633-021-01056-W
Luo Y, Liang J, Zeng G et al (2017) Seed germination test for toxicity evaluation of compost: its roles, problems and prospects. Waste Manag 71:109–114. https://doi.org/10.1016/j.wasman.2017.09.023
Mardi L, Fahoul Y, Tanji K et al (2023) The effect of COVID-19 lockdown on water quality and sediment release in Sebou River (Morocco). Sustain Water Resour Manag 9:1–13. https://doi.org/10.1007/S40899-023-00915-1
Mertah O, El Hajjaji K, El Gaidoumi A et al (2020) Efficient UV-light photocatalytic activity of TiO2@Phosphogypsum composite synthesized by sol–gel method. Opt Quantum Electron 52:1–20. https://doi.org/10.1007/S11082-020-02394-6
Moumen A, Belhocine Y, Sbei N et al (2022) Removal of malachite green dye from aqueous solution by catalytic wet oxidation technique using Ni/kaolin as catalyst. Molecules 27:7528. https://doi.org/10.3390/molecules27217528
Rashtbari Y, Abazari M, Arfaeinia L et al (2023) The optimization of reactive black 5 dye removal in the sono-catalytic process combined with local yellow montmorillonite and hydrogen peroxide using response surface methodology from aqueous solutions. Biomass Convers Biorefin 13:6067–6081. https://doi.org/10.1007/s13399-021-01773-7
Samoila P, Cojocaru C, Mahu E et al (2021) Boosting catalytic wet-peroxide-oxidation performances of cobalt ferrite by doping with lanthanides for organic pollutants degradation. J Environ Chem Eng 9:104961. https://doi.org/10.1016/J.JECE.2020.104961
Sharma A, Shrivastava B, Kuhad RC (2015) Reduced toxicity of malachite green decolorized by laccase produced from Ganoderma sp. rckk-02 under solid-state fermentation. 3 Biotech 5:621–631. https://doi.org/10.1007/s13205-014-0258-1
Shokoohi R, Godini K, Latifi Z (2023) Catalytic oxidation of reactive blue 222 dye using peroxymonosulfate activated by Mn3O4: parameter optimization using response surface methodology. Inorg Chem Commun 149:110400. https://doi.org/10.1016/j.inoche.2023.110400
Siddiqui H, Parra MR, Qureshi MS et al (2018) Studies of structural, optical, and electrical properties associated with defects in sodium-doped copper oxide (CuO/Na) nanostructures. J Mater Sci 53:8826–8843. https://doi.org/10.1007/s10853-018-2179-6
Tai LT, Dat NM, Nam NTH et al (2023) Green synthesis of copper oxide nanoparticles for photodegradation of malachite green and antibacterial properties under visible light. Opt Mater (Amst) 136:113489. https://doi.org/10.1016/j.optmat.2023.113489
Tanji K, El Mrabet I, Fahoul Y et al (2023) Epigrammatic progress on the photocatalytic properties of ZnO and TiO2 based hydroxyapatite@photocatlyst toward organic molecules photodegradation: a review. J Water Process Eng 53:103682. https://doi.org/10.1016/J.JWPE.2023.103682
Zhang H, Liu S, Wang K et al (2023) Optimization of photocatalytic degradation of malachite green using nano-scaled V/CeO2-functionalized steel-slag-based catalyst by response surface design approach. J Mater Res Technol 25:6061–6073. https://doi.org/10.1016/j.jmrt.2023.07.043
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
Not applicable.
Additional information
Responsible Editor: Mohamed Ksibi.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mardi, L., Tanji, K., El Gaidoumi, A. et al. Catalytic wet oxidation degradation of malachite green with Cu-coated sediment as catalyst: parameter optimization using response surface methodology. Euro-Mediterr J Environ Integr (2024). https://doi.org/10.1007/s41207-024-00492-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41207-024-00492-5