Abstract
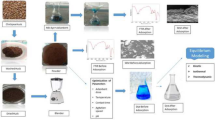
The textile industry has experienced significant advances accompanied by the production of large volumes of polluted water released into the environment, which requires treatment that varies according to the nature of the pollutants. The present work aimed at testing 13X type zeolites synthesized from Djebel Debagh DD1, DD2, and DD3 kaolins in order to remove dyes and other pollutants from the effluent of the COTITEX textile industry (Batna, Algeria). Several experimental parameters were studied (pH, temperature, initial concentration, mass of adsorbent) and physicochemical characterization was carried out on the synthesized adsorbents and the effluent to be treated. The 13X zeolites synthesized from DD1, DD2, and DD3 kaolins were characterized using X-ray diffraction (XRD), Fourier transform infrared (FTIR) spectroscopy, and scanning electron microscopy (SEM). Adsorption experiments were performed using the anionic dye present in the effluent and using real textile effluent. The results showed that the dye adsorption is efficient and feasible at ambient temperature on the 13X zeolite synthesized from DD1 kaolin compared with 13X zeolites synthesized from DD2 and DD3, with good color removal efficiency. Experiments with real textile wastewater showed a significant improvement in the physicochemical characteristics (COD, BOD5, TSS, PO43−, and NO2−) compared with the untreated effluent.
Graphical abstract















Similar content being viewed by others
References
Adamson AW, Gast AP (1997) Physical chemistry of surfaces, 6th edn. Wiley-Interscience, New York
AFNOR NF EN 1899–2 standard (1998) Determination of biochemical oxygen demand after n days (BOD), part 2: Method for undiluted samples (Classification index T90–103–2)
Aiello R, Colella C, Casey DG, Sand LB (1980) Experimental zeolite crystallization in rhyolitic ash-sodium salt systems. In: Rees LVC (ed) Proceedings of the 5th international conference on zeolites, Naples, Italy, 2–6 June 1980. Heyden, London
Akolekar D, Chaffee A, Howe RF (1997) The transformation of kaolin to low-silica X zeolite. Zeolites. https://doi.org/10.1016/S0144-2449(97)00132-2
Alshahidy BA, Abbas AS (2021) Comparative study on the catalytic performance of a 13X zeolite and its dealuminated derivative for biodiesel production. Bull Chem React Engcatal. https://doi.org/10.9767/bcrec.16.4.11436.763-772
Alver E, Metin AÜ (2012) Anionic dye removal from aqueous solutions using modified zeolite: adsorption kinetics and isotherm studies. Chem Eng J. https://doi.org/10.1016/j.cej.2012.06.038
APHA (1998) Standard methods for the exmination of water and waste water, 20th edn. American Public Health Association, Washington
Barrer RM, Beaumont R, Colella C (1974) Action of some basic solutions of metakaolinite and kaolinite. J Chem Soc Dalton Trans 9:934–941
Baur WH (1964) On the cation and water positions in faujasite. Am Min 49(1964):697–704
Belaid KD, Kacha S (2011) Étude cinétique et thermodynamique de l’adsorption d’un colorant basique sur la sciure de bois. J Water Sci 24(2):131–144. https://doi.org/10.7202/1006107ar
Bertolini TCR, Alcantara RR et al (2015) Adsorption of acid orange 8 dye from aqueous solution onto unmodified and modified zeolites. Orbital Electron J Chem 7(4):358–368. https://doi.org/10.17807/orbital.v7i4.76
Calligaris M, Nardin G, Randaccio L (1982) Cation-site location in a natural chabazite. Acta Crystallogr B 38:602–605
Candamano S, Policicchio A, Macario A et al (2019) CO2 adsorption investigation on an innovative nanocomposite material with hierarchical porosity. J NanosciNanotechnol. https://doi.org/10.1166/jnn.2019.16650
Casaburi O, Petrosino F, Marra F (2021) Modeling aspects in simulation of mef processing of solid behaving foods. Chem Eng Trans. https://doi.org/10.3303/CET2187038
Chen D, Hu X, Shi L et al (2012) Synthesis and characterization of zeolite X from lithium slag. Appl Clay Sci. https://doi.org/10.1016/j.clay.2012.02.017
Chowdam R (2016) Rapid and complete degradation of sulphur mustard adsorbed on M/zeolite-13X supported (M = 5 wt% Mn, Fe, Co) metal oxide catalysts with ozone. RSC Adv. https://doi.org/10.1039/C6RA17215F
Curcio S, Petrosino F, Morrone M, De Luca G (2018) Interactions between proteins and the membrane surface in multi scale modeling of organic fouling. J Chem Inf Modeli. https://doi.org/10.1021/acs.jcim.8b00298
de Carvalho TEM, Fungaro DA, Magdalena CP et al (2011) Adsorption of indigo carmine from aqueous solution using coal fly ash and zeolite from fly ash. J Radioanal Nucl Chem. https://doi.org/10.1007/s10967-011-1125-8
Doǧan M, Özdemir Y, Alkan M (2006) Adsorption kinetics and mechanism of cationic methyl violet and methylene blue dyes onto sepiolite. J Dyes Pigm. https://doi.org/10.1016/j.dyepig.2006.07.023
Fardjaoui NH, El Berrichi FZ, Ayari F (2017) Kaolin-issued zeolite A as efficient adsorbent for Bezanyl Yellow and Nylomine Green anionic dyes. Micropor Mesopor Mat. https://doi.org/10.1016/j.micromeso.2017.01.008
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57:385–471
Frisch JM et al (2009) Gaussian 09, Revision E.01. Gaussian, Inc., Wallingford CT
Genethliou C, Triantaphyllidou IE, Giannakis D et al (2021) Simultaneous removal of ammonium nitrogen, dissolved chemical oxygen demand and color from sanitary landfill leachate using natural zeolite. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2020.124679
Hartati H et al (2020) A review on synthesis of kaolin-based zeolite and the effect of impurities. J Chin Chem Soc. https://doi.org/10.1002/jccs.201900047
Hassan I, Antao SM, Parise JB (2004) Sodalite: high-temperature structures obtained from synchrotron radiation and rietveld refinements. Am Mineralog 89:359–364. https://doi.org/10.2138/am-2004-2-315
Ho YS (1995) Adsorption of Heavy Metals from Waste Streams by Peat. Ph.D. Thesis. The university of Birmingham, Birmingham, U.K.
Ho YS, McKay G (2000) The kinetics of sorption of divalent metal ions onto sphagnum moss peat. Water Res 34:735–742. https://doi.org/10.1016/S0043-1354(99)00232-8
IZA-SC (2017) http://www.iza-structure.org/databases/. Accessed 21 Jan 2021
Jaycock MJ, Parfitt GD (1981) Chemistry of interfaces. Ellis Horwood Ltd., Chichester, p 279
Lagergren S (1898) Zur theorie der sogenannten adsorption geloster stoffe. Kungliga Svenska Vetenskapsakademiens. Handl Band 24:1–39
Lakhera SK, Harsha S, Suman AS (2015) Synthesis and characterization of 13X zeolite/activated carbon composite. Int J ChemTech Res 7:1364–1368
Langmuir I (1916) The constitution and fundamental properties of solids and liquids. J Am Chem Soc 38:2221–2295
Li Y, Gao B et al (2009) Adsorption properties of aluminum magnesium mixed hydroxide for the model anionic due reaction brilliant red K-2BP. Hazard Mater 164:1098–1104. https://doi.org/10.1016/j.jhazmat.2008.09.00
Liang G, Li Y et al (2021) Synthesis of ZSM-5 zeolites from biomass power plant ash for removal of ionic dyes from aqueous solution: equilibrium isotherm, kinetic and thermodynamic analysis. RSC Adv 11:22365–22375. https://doi.org/10.1039/d1ra03847h
Ma Y, Yan C, Alshameri A et al (2014) Synthesis and characterization of 13X zeolite from low-grade natural kaolin. Adv Powder Technol. https://doi.org/10.1016/j.apt.2013.08.002
Maarof HI, Hameed BH, Ahmad AL (2003) Aqueous-phase adsorption of phenolic compounds on activated carbon. Environ Manage 25(2003):569–572
Naushad M, Alqadami A et al (2019) Adsorption of textile dye using para-amino benzoic acid modified activated carbon: kinetic and equilibrium studies. J Mol Liq. https://doi.org/10.1016/j.molliq.2019.112075
NF EN 872 (2005) European standards published by AFNOR (French standardization association), water quality—determination of suspended solids
Petrosino F, Curcio S, Chakraborty S, De Luca G (2019) Enzyme Immobilization on Polymer Membranes: A Quantum and Molecular Mechanics Study. Computation. https://doi.org/10.3390/computation70400566
Petrosino F, Hallez Y, De Luca G, Curcio S (2020) Osmotic pressure and transport coefficient in ultrafiltration: a Monte Carlo study using quantum surface charges. Chem Eng Sci 224:115762. https://doi.org/10.1016/j.ces.2020.115762
Pub©Chem (2015) Open Chemistry Database, CID 23668793.
Qiang Z, Yan Z, Jing O, Yi Z, Yang H, Chen D (2018) Chemically modified kaolinite nanolayers for the removal of organic pollutants. Appl Clay Sci. https://doi.org/10.1016/j.clay.2018.03.009
Radoor S, Karayil J, Jayakumar A, Parameswaranpillai J, Siengchin S (2021) Efficient removal of methyl orange from aqueous solution using mesoporous ZSM-5 zeolite: Synthesis, kinetics and isotherm studies. Colloids Surf A Physicochem Eng Asp. https://doi.org/10.1016/j.colsurfa.2020.125852
Ramdani TZ, Guendouzi A, Kadeche A, Herbache H, Mostefai A, Taleb S, Deratani A (2020) Mechanism study of metal ion adsorption on porous hydroxyapatite: experiments and modeling. Can J Chem. https://doi.org/10.1139/cjc-2019-03155
Rita K (2012) Adsorption of yellow dye: Acid yellow RR from its aqueous solution using two different samples of activated carbon by static batch method. J Nat Sci. https://doi.org/10.4236/ns.2012.42016 (10.17807/orbital.v7i4.766)
Sanxin N, Xiaokang X, Zheng W et al (2019) Enhanced removal performance for Congo red by coal-series kaolin with acid treatment. Env Tech. https://doi.org/10.1080/09593330.2019.1670269
Schwanke AJ, Balzer R, Pergher S (2019) Microporous and mesoporous materials from natural and inexpensive sources. In: Martínez L, Kharissova O, Kharisov B (eds) Handbook of ecomaterials. Springer, Cham. https://doi.org/10.1007/978-3-319-68255-6_43
Shigemoto N, Hayashi H, Miyaura K (1993) Selective formation of Na-X zeolite from coal fly ash by fusion with sodium hydroxide prior to hydrothermal reaction. J Mater. https://doi.org/10.1007/BF00414272
Singer A, Berkgaut V (1995) Cation exchange properties of hydrothermally treated coal fly ash. Environ Sci Technol. https://doi.org/10.1021/es00007a009
Slatni I, El Berrichi FZ, Duplay J et al (2020) Mesoporous silica synthesized from natural local kaolin as an effective adsorbent for removing of Acid Red 337 and its application in the treatment of real industrial textile. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-020-08615-5
Stewart JJP (2007) Optimization of parameters for semi empirical methods V: Modification of NDDO approximations and application to 70 elements. J Mol Model. https://doi.org/10.1007/s00894-007-0233-4
Suquet H (1989) Effects of dry grinding and leaching on the crystal structure of chrysotile. Clays Clay Miner. https://doi.org/10.1346/CCMN.1989.0370507
Tabti A, Djafri F, Launay F, Serier M, Benchikh (2021) Adsorption of bromothymol blue (BTB) dye using four zeolites as adsorbent. Kem Ind. https://doi.org/10.15255/KUI.2020.052
Treacy MMJ, Higgins JB (2007) Collection of simulated XRD powder patterns for Zeolites. Fifth (5th) revised edition. Elsevier. https://doi.org/10.1016/B978-0-444-53067-7.X5470-77
Villaquirán-Caicedo MA, De Gutiérrez RM, Gordillo M, Gallego NC (2016) Synthesis of zeolites from a low-quality Colombian kaolin. Clays Clay Miner. https://doi.org/10.1346/CCMN.2016.0640201
Weber WJ, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng ASCE 89:31–59
Xu HY, Wu LC, Shi T et al (2014) Adsorption of acid fuchsin onto LTA-type zeolite derived from fly ash. Sci China Technol Sci. https://doi.org/10.1007/s11431-014-5542-0
Yan L, Qin L, Yu H, Li S, Shan R, Du B (2015) Adsorption of acid dyes from aqueous solution by CTMAB modified bentonite: kinetic and isotherm modeling. J Mol Liq 211:1074–1081. https://doi.org/10.1016/j.molliq.2015.08.032
Yunan MH, Yan C et al (2014) Synthesis and characterization of 13X zeolite from low-grade natural kaolin. Adv Powder Technol 25:495–499. https://doi.org/10.1016/J.APT.2013.08.002
Zen S, El Berrichi FZ, Abidi N et al (2018) Activated kaolin’s potential adsorbents for the removal of Derma Blue R67 acid dye: kinetic and thermodynamic studies. Desal Water Treat. https://doi.org/10.5004/dwt.2018.21996
Zhan BZ, White MA, Lumsden M et al (2002) Control of particle size and surface properties of crystals of NaX zeolite. Chem Mater. https://doi.org/10.1021/cm011635f
Acknowledgements
This paper is supported by the PRIMA program under grant agreement No. 2024—TRUST project. The PRIMA program is supported by the European Union. It was also supported by the General Directorate of Scientific Research and Technological Development (DGRSDT) in Algeria under the authority of the Minister in charge of scientific research and France (National Research Agency, ANR). The authors would like to gratefully acknowledge Mrs. Rabia Gueraiche (Engineer in the EATIT company) for providing the textile effluent, and the industrial analysis and materials engineering laboratory (LAIGM) in Guelma for performing the COD analyses.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Sudip Chakraborty.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Slatni, I., Dhiffalah, A., Elberrichi, F.Z. et al. Investigation of adsorption properties of modified DD kaolins to microporous material type 13X zeolite in treatment of textile industry effluent: experiments and theoretical approach. Euro-Mediterr J Environ Integr 7, 415–432 (2022). https://doi.org/10.1007/s41207-022-00324-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41207-022-00324-4