Abstract
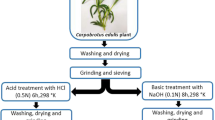
The purpose of this study was to enhance the biosorption capacity of the aquatic biomass Callitriche obtusangula for the anionic dye Direct Red 89 in aqueous solution through chemical pretreatment. Batch experiments were carried out to determine the optimal pretreatment parameters of the biomass, such as the nature and the concentration of the activating agent and the effect of pretreatment time. The best dye removal efficiency was found to be 83%, which was obtained using the biomass treated with H3PO4, an initial dye concentration of 40 mg/L, a temperature of 20 °C, a biomass dose of 2 g/L, and a pH of 1. A number of methods of probing the structure of the biomass, including Fourier-transform infrared spectroscopy, scanning electron microscopy, energy-dispersive X-ray analysis, specific surface area calculation, point of zero charge measurement, and the Boehm titration method, indicated that phosphoric acid acts as a dehydrating and oxidizing agent that enhances the surface area and functional groups of the biomass. Moreover, phosphoric acid promoted dye biosorption by grafting phosphate functions onto the biomass and enhancing the acid functions involved in dye fixation. Adsorption isotherm studies showed that the Freundlich model agreed well with the experimental data. In addition, the kinetics of biosorption were best described by a pseudo-second-order model.
Graphic abstract












Similar content being viewed by others
References
Abdullah RH (2015) Adsorption study for removal of direct red 89 and basic blue 41 dyes from aqueous solutions using sawdust as a high efficient adsorbent. Euphrates J Agric Sci 7:16–28
Akpomie KG, Dawodu FA, Adebowale KO (2015) Mechanism on the sorption of heavy metals from binary solution by a low cost montmorillonite and its desorption potential. Alex Eng J 54:757–767. https://doi.org/10.1016/j.aej.2015.03.025
Al-Degs Y, Allen KM, Ahmad MS (2000) Effect of carbon surface chemistry on the removal of reactive dyes from textile effluents. Water Res 34:927–935. https://doi.org/10.1016/S0043-1354(99)00200-6
Alinejad-Mir A, Amooey AA, Ghasemi S (2018) Adsorption of direct yellow 12 from aqueous solutions by an iron oxide-gelatin nanoadsorbent; kinetic, isotherm and mechanism analysis. J Clean Prod 170:570–580. https://doi.org/10.1016/j.jclepro.2017.09.101
Aljeboree AM, Alkaim AF, Alshirifi AN (2017) Kinetics and equilibrium study for the adsorption of textile dyes on coconut shell activated carbon. Arab J Chem 2017:3381–3393. https://doi.org/10.1016/j.arabjc.2014.01.020
Ausavasukhi A, Kampoosaen C, Kengnok O (2015) Adsorption characteristics of Congo red on carbonized leonardite. J Clean Prod 134:506–514. https://doi.org/10.1016/j.jclepro.2015.10.034
Azzaz AA, Jellali S, Akrout H, Assadi AA, Bousselmi L (2017) Optimization of a cationic dye removal by a chemically modified agriculture by-product using response surface methodology: biomasses characterization and adsorption properties. Environ Sci Pollut Res 24:9831–9846. https://doi.org/10.1007/s11356-016-7698-6
Azzaz AA, Jellali S, Bengharez Z, Bousselmi L, Akrout H (2018a) Investigations on a dye desorption from modified biomass by using a low-cost eluent: hysteresis and mechanisms exploration. Int J Environ Sci Technol 16(11):7393–7408. https://doi.org/10.1007/s13762-018-2171-3
Azzaz AA, Jellali S, Akrout H, Assadi AA, Bousselmi L (2018b) Dynamic investigations on cationic dye desorption from chemically modified lignocellulosic material using a low-cost eluent: dye recovery and anodic oxidation efficiencies of the desorbed solutions. J Clean Prod 201:28–38. https://doi.org/10.1016/j.jclepro.2018.08.023
Baldy V, Trémolières M, Andrieu M, Belliard J (2007) Changes in phosphorous content of two aquatic macrophytes according to water velocity, trophic status and time period in hardwater streams. Hydrobiologia 575:343–351. https://doi.org/10.1007/s10750-006-0380-0
Banisheykholeslami F, Hosseini M, Darzi GN (2021) Design of PAMAM grafted chitosan dendrimers biosorbent for removal of anionic dyes: adsorption isotherms, kinetics and thermodynamics studies. Int J Biol Macromol 177:306–316. https://doi.org/10.1016/j.ijbiomac.2021.02.118
Bello OS, Ahmad MA, Puad NAA (2014) Kinetic, equilibrium and thermodynamic studies of synthetic dye removal using pomegranate peel activated carbon prepared by microwave-induced KOH activation. Water Resour Ind 6:18–35. https://doi.org/10.1016/j.wri.2014
Ben Arfi R, Ghorbal A, Karoui S, Mougin K (2017) Adsorptive removal of cationic and anionic dyes from aqueous solution by utilizing almond shell as bioadsorbent. Euro-Mediterr J Environ Integr 2:20
Ben Arfi R, Ghorbal A, Karoui S, Mougin K (2019) Cetyltrimethylammonium bromide-treated Phragmites australis powder as novel polymeric adsorbent for hazardous Eriochrome Black T removal from aqueous solutions. Polym Bull 76:5077–5102. https://doi.org/10.1007/s00289-018-2648-8
Benabbas K, Hocini I, Khellaf N (2018) Biosorption of the anionic dye Direct Red 89 by the aquatic plant Callitriche obtusangula. In: Abdelbaki B, Safi B, Saidi M (eds) Proceedings of the Third International Symposium on Materials and Sustainable Development. Springer International, Cham, pp 540–548. doi:https://doi.org/10.1007/978-3-319-89707-3_58
Benabbas K, Zabat N, Hocini I (2020a) Azo dye removal by acid pretreated biomass and its regeneration by visible light photocatalysis with incorporated CuO. Environ Technol. https://doi.org/10.1080/09593330.2020.1786169
Benabbas K, Zabat N, Hocini I (2020b) Eco-friendly synthesis of self-regenerative low-cost biosorbent by the incorporation of CuO: a photocatalyst sensitive to visible light irradiation for azo dye removal. Environ Sci Pollut Res 27(25):31074–31091. https://doi.org/10.1007/s11356-020-09364-1
Boehm HP (1994) Some aspects of the surface chemistry of carbon blacks and other carbons. Carbon 32:759–769. https://doi.org/10.1016/0008-6223(94)90031-0
Carliell CM, Barclay SJ, Buckley CA (1996) Treatment of exhausted reactive dye bath effluent using anaerobic digestion: laboratory and full-scale trials. Water SA 22:225–231
Cazetta AL, Vargas AMM, Nogami EM, Kunita MH, Guilherme MR, Martins AC, Silva TL, Moraes JCG, Almeida VC (2011) NaOH-activated carbon of high surface area produced from coconut shell: kinetics and equilibrium studies from the methylene blue adsorption. Chem Eng J 174:117–125. https://doi.org/10.1016/j.cej.2011.08.058
Chen PJ, Shunnian W (2004) Acid/base-treated activated carbons: characterization of functional groups and metal adsorptive properties. Langmuir 20:2233–2242. https://doi.org/10.1021/la0348463
Chowdhury ZZ, Abd Hamid SB, Das R, Hasan MDR, Zain SM, Khalid K, Uddin MN (2013) Preparation of carbonaceous adsorbents from lignocellulosic biomass and their use in removal of contaminants from aqueous solution. BioResources 8:6523–6555. https://doi.org/10.15376/biores.8.4.6523-6555
Deischter J, Palkovits R, Tailoring WN (2020) Activated carbons for an efficient downstream processing: selective liquid-phase adsorption of lysine. ChemSusChem 13(14):3614–3621. https://doi.org/10.1002/cssc.202000885
Den W, Sharma VK, Lee M, Nadadur G, Varma RS (2018) Lignocellulosic biomass transformations via greener oxidative pretreatment processes: access to energy and value-added chemicals. Front Chem. https://doi.org/10.3389/fchem.2018.00141
Ding Y, Zhou P, Tang H (2016) Visible-light photocatalytic degradation of bisphenol A on NaBiO3 nanosheets in a wide pH range: a synergistic effect between photocatalytic oxidation and chemical oxidation. Chem Eng J 291:149–160. https://doi.org/10.1016/j.cej.2016.01.105
Dobele G, Dizhbite T, Rossinskaja G, Telysheva G, Meier D, Radtke S, Faix O (2003) Pre-treatment of biomass with phosphoric acid prior to fast pyrolysis. J Anal Appl Pyrol 68(69):197–211. https://doi.org/10.1016/s0165-2370(03)00063-9
El Sayed G, Yehia MM, Asaad AA (2014) Assessment of activated carbon prepared from corncob by chemical activation with phosphoric acid. Water Resour Ind 20:66–75. https://doi.org/10.1016/j.wri.2014.10.001
El-Barghouthi MI, El-Sheikh AH, Al-Degs YS, Walker GM (2007) Adsorption behavior of anionic reactive dyes on H-type activated carbon. Compet Adsorpt Desorption Stud 42:2195–2220. https://doi.org/10.1080/01496390701444030
Elkady MF, Ibrahim AM, Abd El-Latif MM (2011) Assessment of the adsorption kinetics, equilibrium and thermodynamic for the potential removal of reactive red dye using eggshell biocomposite beads. Desalination 278:412–423. https://doi.org/10.1016/j.desal.2011.05.063
Fadillah G, Saleh A, Wahyuningsih S (2019) Enhanced electrochemical degradation of 4-nitrophenol molecules using novel Ti/TiO2-NiO electrodes. J Mol Liq 289:108–111. https://doi.org/10.1016/j.molliq.2019.111108
Fytianos K, Voudrias E, Kokkalis E (2000) Sorption–desorption behavior of 2,4-dichlorophenol by marine sediments. Chemosphere 40:3–6. https://doi.org/10.1016/S0045-6535(99)00214-3
Galán J, Rodríguez A, Gómez JM, Allen SJ, Walker GM (2013) Reactive dye adsorption onto a novel mesoporous carbon. Chem Eng J 2019:62–68. https://doi.org/10.1016/j.cej.2012.12.073
Gao X, Liu K, Omosebi A, Landon J (2015) Removal in an inverted capacitive deionization cell using amine modified microporous carbon cathode. Environ Sci Technol 49:10920–10926. https://doi.org/10.1021/acs.est.5b02320
Gerçel Ö, Özcan A, Özcan AS, Gerçel HF (2007) Preparation of activated carbon from a renewable bio-plant of Euphorbia rigida by H2SO4 activation and its adsorption behavior in aqueous solutions. Appl Surf Sci 253:4843–4852. https://doi.org/10.1016/j.apsusc.2006.10.053
Grabsi M, Zabat N, Khellaf N, Ismail F (2019) Synthesis of an environmental nano-polyoxometalate (α2P2W17CoO61)8− as catalyst for dyes degradation: a comparative study oxidation of indigoid and azo dyes. Environ Nanotechnol Monit Manag 12:00269. https://doi.org/10.1016/j.enmm.2019.100269
Gratuito MKB, Panyathannaporn T, Chumnanklang RA, Sirinuntawittaya N, Dutta A (2008) Production of activated carbon from coconut shell: optimization using response surface methodology. Bioresour Technol 99:4847–4895. https://doi.org/10.1016/j.biortech.2007.09.042
Guendouz S, Khellaf N, Zerdaoui M, Ouchefoun M (2013) Biosorption of synthetic dyes (Direct Red 89 and Reactive Green 12) as an ecological refining step in textile effluent treatment. Environ Sci Pollut Res 20:3822–3829. https://doi.org/10.1007/s11356-012-1314-1
Gupta VK, Ali I, Saleh TA, Nayak A, Agarwal S (2012) Chemical treatment technologies for waste-water recycling: an overview. RSC Adv 2:6380–6388. https://doi.org/10.1039/C2RA20340E
Han R, Zhang L, Song C, Zhang M, Zhu H, Zhang L (2010) Characterization of modified wheat straw, kinetic and equilibrium study about copper ion and methylene blue adsorption in batch mode. Carbohydr Polym 79:1140–1149. https://doi.org/10.1016/j.carbpol.2009.10.054
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34:451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Ho YS, Ofomaja AE (2006) Biosorption thermodynamics of cadmium on coconut copra meal as biosorbent. Biochem Eng J 30:117–123. https://doi.org/10.1016/j.bej.2006.02.012
Hocini I, Khellaf N, Benabbas K, Djelal H, Amrane A (2020) Identification of the mechanism involved in the removal potential of textile pollutants by the aquatic plant Lemna gibba L. J Chem Health Risk 10(2):145–153
Hoseini MM, Safari GH, Kamani H (2014) Sonocatalytic degradation of tetracycline antibiotic in aqueous solution by sonocatalysis. Toxicol Environ Chem 95(10):1680–1689. https://doi.org/10.1080/02772248.2014.901328
Hu R, Xiao J, Chena G (2020) Micro-nano-engineered nitrogenous bone biochar developed with a ballmilling technique for high-efficiency removal of aquatic Cd(II), Cu(II) and Pb(II). J Hazard Mater 387:121980. https://doi.org/10.1016/j.jhazmat.2019.121980
Jain SN, Gogate PR (2017) Acid Blue 113 removal from aqueous solution using novel biosorbent based on NaOH treated and surfactant modified fallen leaves of Prunusdulcis. J Environ Chem Eng 5:3384–3394. https://doi.org/10.1016/j.jece.2017.06.047
Ju DJ, Byun IG, Park JJ, Lee CH, Ahn GH, Park TJ (2008) Biosorption of a reactive dye (Rhodamine-B) from an aqueous solution using dried biomass of activated sludge. Biores Technol 99:7971–7975. https://doi.org/10.1016/j.biortech.2008.03.061
Karoui S, Ben Arfi R, Mougin K, Ghorbal A, Assadi A, Amrane A (2019) Synthesis of novel biocomposite powder for simultaneous removal of hazardous ciprofloxacin and methylene blue: central composite design, kinetic and isotherm studies using Brouers-Sotolongo family models. J Haz Mat 387:121675. https://doi.org/10.1016/j.jhazmat.2019.121675
Kowanga KD, Gatebe E, Mauti GO, Mauti EM (2016) Kinetic sorption isotherms, pseudo-first-order model and pseudo-second-order model studies of Cu(II) and Pb(II) using defatted Moringa oleifera seed powder. J Phytopharmacol 5:71–78
Lansdown MV, Bazos I, Caria MC, Troia A, Wieringa JJ (2017) New distribution and taxonomic information on Callitriche (Plantaginaceae) in the Mediterranean region. Phytotaxa 1:91–104. https://doi.org/10.11646/phytotaxa.313.1.6
Lee CL, H’ng PS, Paridah MT, Chin KL, Rashid U, Maminski M, Khoo PS (2018) Production of bioadsorbent from phosphoric acid pretreated palm kernel shell and coconut shell by two-stage continuous physical activation via N2 and air. R Soc Open Sci 5(12):180775. https://doi.org/10.1098/rsos.180775
Li C, Xia H, Zhang L, Shu J, Shengzhou Z, Peng J, Cheng S (2016) Kinetics and isotherms studies for Congo red adsorption on mesoporous Eupatorium adenophorum-based activated carbon via microwave-induced H3PO4 activation. J Mol Liq 224:737–744. https://doi.org/10.1016/j.molliq.2016.10.048
Lillo-Ródenas MA, Cazorla-Amorós D, Linares-Solano A (2003) Understanding chemical reactions between carbons and NaOH and KOH: an insight into the chemical activation mechanism. Carbon 41:267–275. https://doi.org/10.1016/S0008-6223(02)00279-8
Lin L, Zhai S, Xiao Z, Song Y, An Q, Song X (2013) Dye adsorption of mesoporous activated carbons produced from NaOH-pretreated rice husks. Biores Technol 136:437–443. https://doi.org/10.1016/j.biortech.2013.03.048
Mahardiani L, Saputro S, Baskoro F, Zinki NM, Taufiq M (2019) Facile synthesis of carboxylated activated carbon using green approach for water treatment. Mater Sci Eng 578:012003. https://doi.org/10.1088/1757-899X/578/1/012003
Mashkoor F, Nasar A, Inamuddin A, Asiri M (2018) Exploring the reusability of synthetically contaminated wastewater containing crystal violet dye using Tectona grandis sawdust as a very low-cost adsorbent. Sci Rep 8:8314. https://doi.org/10.1038/s41598-018-26655-3
Mateus GAP, Formentini-Schmitt DM, Nishi L, Fagundes-Klen MR, Gomes RG, Bergamasco R (2017) Coagulation/flocculation with Moringa oleifera and membrane filtration for dairy wastewater treatment. Water Air Soil Pollut 228:228–342. https://doi.org/10.1007/s11270-017-3509-z
Mirzaei A, Yerushalmi L, Chen Z, Haghighat F (2018) Photocatalytic degradation of sulfamethoxazole by hierarchical magnetic ZnO@g-C3N4: RSM optimization, kinetic study, reaction pathway and toxicity evaluation. J Hazard Mater 359:516–526. https://doi.org/10.1016/j.jhazmat.2018.07.077
Moldovan A, Neag E, Băbălău-Fuss V, Cadar O, Micle V, Roman C (2018) Optimized removal of methylene blue from aqueous solution using a commercial natural activated plant-based carbon and Taguchi experimental design. Environ Lett. https://doi.org/10.1080/00032719.2017.1418879
Namanea A, Mekarziab A, Benrachedib K, Belhaneche N, Hellala A (2005) Determination of the adsorption capacity of activated carbon made from coffee grounds by chemical activation with ZnCl2 and H3PO4. J Hazard Mater B 119:189–194. https://doi.org/10.1016/j.jhazmat.2004.12.006
Nigam P, Armour G, Banat IM, Singh D (2000) Physical removal of textile dyes from effluents and solid-state fermentation of dye-adsorbed agricultural residues. Biores Technol 72:219–226. https://doi.org/10.1016/S0960-8524(99)00123-6
Oussalah A, Boukerroui A (2020) Removal of cationic dye using alginate–organobentonite composite beads. Euro-Mediterr J Environ Integr 5:55. https://doi.org/10.1007/s41207-020-00199-3
Ponce J, Andrade JGS, Santos LND, Bulla MK, Barros BCB, Favaro SL, Hioka N, Caetano W, Batistela VR (2021) Alkali pretreated sugarcane bagasse, rice husk and corn husk wastes as lignocellulosic biosorbents for dyes. Carbohydr Polym Technol Appl. https://doi.org/10.1016/j.carpta.2021.100061
Puziy AM, Poddubnaya OI, Martinez-Alonso A, Suarez-Garcia F (2002) Synthetic carbons activated with phosphoric acid. I. Surface chemistry and ion binding properties. Carbon 40:1493–1505. https://doi.org/10.1016/S0008-6223(01)00317-7
Robach F, Hajnsek I, Eglin I, Trémolières M (1995) Phosphorus sources for aquatic macrophytes in running waters: water or sediment. Acta Bot Gall 142:719–731. https://doi.org/10.1080/12538078.1995.10515296
Sánchez Orozco R, Martínez-Juan M, García-Sánchez J, Ureña-Núñez F (2018) Removal of methylene blue from aqueous solution using Typha stems and leaves. Bio Resour 13:1696–1710. https://doi.org/10.15376/biores.13.1.1696-1710
Sewu DD, Boakye P, Woo SH (2017) Highly efficient adsorption of cationic dye by biochar produced with Korean cabbage waste. Biores Technol 224:206–213. https://doi.org/10.1016/j.biortech.2016.11.009
Shaban M, Abukhadra MR, Hamd A, Amin RR, Abdel Khalek A (2017) Photocatalytic removal of Congo red dye using MCM-48/Ni2O3 composite synthesized based on silica gel extracted from rice husk ash; fabrication and application. J Environ Manag 204:189–199. https://doi.org/10.1016/j.jenvman.2017.08.048
Shanmugam S, Karthik K, Veerabagu U, Hari A, Swaminathan K, Al-Kheraif AA, Whangchai K (2021) Bi-model cationic dye adsorption by native and surface-modified Trichoderma asperellum BPL MBT1 biomass: from fermentation waste to value-added biosorbent. Chemosphere. https://doi.org/10.1016/j.chemosphere.2021.130311
Sharma S, Jadhav SK, Tiwari KL (2018) Diversity of fungal endophytes in Typha latifolia (L.) and their lead biosorption activity. Euro-Mediterr J Environ Integr 3:4. https://doi.org/10.1007/s41207-017-0041-x
Shen J, Huang G, An C, Xin X, Huang C, Rosendahl S (2018) Removal of tetrabromobisphenol A by adsorption on pinecone-derived activated charcoals: synchrotron FTIR, kinetics and surface functionality analyses. Biores Technol 247:812–820. https://doi.org/10.1016/j.biortech.2017.09.177
Siipola V, Tamminen T, Källi A, Lahti R, Romar H, Rasa K, Keskinen R, Hyväluoma J, Hannula M, Wikberg H (2018) Effects of biomass type, carbonization process, and activation method on the properties of bio-based activated carbons. Bio Resour 13:5976–6002. https://doi.org/10.15376/biores.13.3.5976-6002
Sivaraj R, Namasivayam C, Kadirvelu K (2001) Orange peel as an adsorbent in the removal of acid violet 17 (acid dye) from aqueous solutions. Waste Mang 21:105–110. https://doi.org/10.1016/S0956-053X(00)00076-3
Son Y, Hong J, Lim H, Kim T (2005) A study of heterobifunctional reactive dyes on nylon fibers: dyeing properties, dye moiety analysis and wash fastness. Dye Pigments 66:231–239. https://doi.org/10.1016/j.dyepig.2004.10.004
Stephenson RJ, Sheldon JB (1996) Coagulation and precipitation of mechanical pulping effluent—I. Remov Carbon Colour Turbid Water Res 30:781–792. https://doi.org/10.1016/0043-1354(95)00213-8
Sun D, Zhang Z, Wang M, Wu Y (2013) Adsorption of reactive dyes on activated carbon developed from Enteromorpha prolifera. Am J Analyt Chem 4:17–26. https://doi.org/10.4236/ajac.2013.47A003
Surech S (2016) Removal of Basic Violet 14 from aqueous solution using sulphuric acid activated materials. Springerplus 5:633. https://doi.org/10.1186/s40064-016-2294-2
Tabara A, Amane CY, Eguchi MS (2012) Adsorption mechanism for xanthene dyes to cellulose granules acid red. Biosci Biotechnol Biochem 76:870–874. https://doi.org/10.1271/bbb.110634
Tan IAW, Ahmad AL, Hameed BH (2008) Adsorption of basic dye on high-surface area activated carbon prepared from coconut husk: equilibrium, kinetic and thermodynamic studies. J Hazard Mater 154:337–346. https://doi.org/10.1016/j.jhazmat.2007.10.031
Tareq S, Hin Y, Yap T, Saleh TA, Abdullah AHAH, Rashid U, Izham SM (2018) Synthesis of bimetallic gold-pallidum loaded on carbon as efficient catalysts for the oxidation of benzyl alcohol into benzaldehyde. J Mol Liq 271:885–891. https://doi.org/10.1016/j.molliq.2018.09.037
Thinakaran N, Baskaralingam P, Pulikesi M, Panneerselvam P, Sivanesan S (2008) Removal of Acid Violet 17 from aqueous solutions by adsorption onto activated carbon prepared from sunflower seed hull. J Hazard Mater 151:316–322. https://doi.org/10.1016/j.jhazmat.2007.05.076
Tian S, Xu S, Liua J, He C, Xiong Y, Feng P (2019) Highly efficient removal of both cationic and anionic dyes from wastewater with a water-stable and eco-friendly Fe-MOF via host-guest encapsulation. J Clean Prod 239:117767. https://doi.org/10.1016/j.jclepro.2019.117767
Tran HN, Chao H (2018) Adsorption and desorption of potentially toxic metals on modified biosorbents through new green grafting process. Environ Sci Pollut Res 25:12808–12820. https://doi.org/10.1007/s11356-018-1295-9
Tran HN, Wang Y, You S, Chao H (2017) Insights into the mechanism of cationic dye adsorption on activated charcoal: the importance of π–π interactions. Process Saf Environ Prot 107:168–180. https://doi.org/10.1080/00986445.2017.1336090
Tseng RL (2007) Physical and chemical properties and adsorption type of activated carbon prepared from plum kernels by NaOH activation. J Hazard Mater 147:1020–1027. https://doi.org/10.1016/j.jhazmat.2007.01.140
Vasanth KK, Porkodi K (2007) Mass transfer, kinetics and equilibrium studies for the biosorption of methylene blue using Paspalum notatum. J Hazard Mater 146:214–226. https://doi.org/10.1016/j.jhazmat.2006.12.010
Vlyssides AG, Loizidou M, Karlis PK, Zorpas AA, Papaioannou DJ (1999) Electrochemical oxidation of a textile dye wastewater using a Pt/Ti electrode. J Hazard Mater 70:41–52. https://doi.org/10.1016/S0304-3894(99)00130-2
Wang S, Zhu ZH (2007) Effects of acidic treatment of activated carbons on dye adsorption. Dye Pigment 75:306–314. https://doi.org/10.1016/j.dyepig.2006.06.005
Wang L, Hou D, Li X, Tsang DCW, Jin F (2020) Green remediation of Cd and Hg contaminated soil using humic acid modified montmorillonite: immobilization performance under accelerated ageing conditions. J Hazard Mater 387:122005. https://doi.org/10.1016/j.jhazmat.2019.122005
Watt EDK, Evgenii LK, Shimada H, Patrick LJ (2007) The mechanism of rate-limiting motions in enzyme function. Proc Natl Acad Sci USA 104:11981–11986. https://doi.org/10.1073/pnas.0702551104
Weber JWJ, Morriss JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng Div Am Soc Civil Eng 89:31–60
Wood KA, Stillman RA, Clarke RT, Daunt F, O’Hare MT (2012) Understanding plant community responses to combinations of biotic and abiotic factors in different phases of the plant growth cycle. PLoS ONE 7:49824. https://doi.org/10.1371/journal.pone.0049824
Xiao J, Hu R, Chena G (2020) Micro-nano-engineered nitrogenous bone biochar developed with a ballmilling technique for high-efficiency removal of aquatic Cd(II), Cu(II) and Pb(II). J Hazard Mater 387:121980. https://doi.org/10.1016/j.jhazmat.2019.121980
Yagub MT, Sen TKS, Afroze S, Ang HM (2014) Dye and its removal from aqueous solution by adsorption: a review. Adv Colloid Interface Sci 209:172–184. https://doi.org/10.1016/j.cis.2014.04.002
Zhao L, Zhang Q, Li X, Ye J, Chena J (2020) Adsorption of Cu(II) by phosphogypsum modified with sodium dodecyl benzene sulfonate. J Hazard Mater 387:121–808
Acknowledgements
The authors wish to express their gratitude to the laboratory staff of Souk Ahras (Algeria) for their technical assistance with the spectroscopic analytical technique (FTIR), and to the laboratory (LOMOP). The authors thank the Ministry of Higher Education and Scientific Research, Algeria for financial support through the PRFU project (N°: A16N01UN230120180001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declares that there is no competing interest in this work.
Additional information
Communicated by Responsible Editor: Aymen Amin Assadi.
Rights and permissions
About this article
Cite this article
Benabbas, K., Zabat, N. & Hocini, I. Study of the chemical pretreatment of a nonconventional low-cost biosorbent (Callitriche obtusangula) for removing an anionic dye from aqueous solution. Euro-Mediterr J Environ Integr 6, 54 (2021). https://doi.org/10.1007/s41207-021-00265-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41207-021-00265-4