Abstract
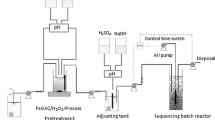
It is unusual to observe completely treated textile wastewater in Ethiopia. It is impossible to get better quality of treated effluent with a single treatment stage. Therefore, in this study the removal of COD and color was carried out on a wastewater which was obtained from acrylic fiber processing textile industry using a single-stage Fenton oxidation, single-stage sequencing batch reactor (SBR) and also with the integration of SBR with Fenton oxidation. Optimum amount of process factors was used for both Fenton oxidation and SBR treatment stages. The combination of SBR and Fenton oxidation was revealed better removal efficiency than single SBR-stage treatment. The effluent obtained from SBR at steady-state conditions indicated a maximum COD and color removal of 74.1% and 64.6%, respectively. The effluent obtained from Fenton followed by SBR (Fenton + SBR) at steady-state conditions was indicated a maximum COD and color removal efficiency of 86.3% and 84%, respectively. The effluent obtained from SBR followed by Fenton (SBR + Fenton) for three Fenton oxidation experimental runs indicated a maximum COD and color removal of 80.2% and 73.6%, respectively. Among the three wastewater treatment schemes, chemical treatment before biological stage (Fenton + SBR) was the best treatment option and also showed better quality of effluent.








Similar content being viewed by others
References
Paździor K, Bilińska L, Ledakowicz S (2019) A review of the existing and emerging technologies in the combination of AOPs and biological processes in industrial textile wastewater treatment. Chem Eng J. https://doi.org/10.1016/j.cej.2018.12.057
Oller I, Malato S, Sánchez-Pérez JA (2011) Combination of Advanced Oxidation Processes and biological treatments for wastewater decontamination-A review. Sci Total Environ 409:4141–4166. https://doi.org/10.1016/j.scitotenv.2010.08.061
Aslam MM, Baig MA, Hassan I et al (2004) Textile wastewater characterization and reduction of its rCOD & BOD by oxidation. Electron J Environ Agric Food Chem 3:804–811
Bisschops I, Spanjers H (2003) literature review on textile wastewater characterization. Environ Technol. https://doi.org/10.1080/09593330309385684
Aye T, Mehrvar M, Anderson WA (2004) Effects of photocatalysis on the biodegradability of cibacron brilliant yellow 3G-P (reactive yellow 2). J Environ Sci Heal Part A Toxic Hazard Substain Environ Eng 39:113–126. https://doi.org/10.1081/ESE-120027372
Renou S, Givaudan JG, Poulain S et al (2008) Landfill leachate treatment: review and opportunity. J Hazard Mater 150:468–493. https://doi.org/10.1016/j.jhazmat.2007.09.077
Shi B, Li G, Wang D et al (2007) Removal of direct dyes by coagulation: the performance of preformed polymeric aluminum species. J Hazard Mater 143:567–574. https://doi.org/10.1016/j.jhazmat.2006.09.076
Blanco J, Torrades F, De M, García-montaño J (2012) Fenton and biological-Fenton coupled processes for textile wastewater treatment and reuse. Desalination 286:394–399. https://doi.org/10.1016/j.desal.2011.11.055
Méndez-Arriaga F, Esplugas S, Giménez J (2008) Photocatalytic degradation of non-steroidal anti-inflammatory drugs with TiO2 and simulated solar irradiation. Water Res 42:585–594. https://doi.org/10.1016/j.watres.2007.08.002
Lucas MS (2006) Decolorization of the azo dye Reactive Black 5 by Fenton and photo-Fenton oxidation. Dye Pigm 71:236–244. https://doi.org/10.1016/j.dyepig.2005.07.007
Fongsatitkul P, Elefsiniotis P, Yamasmit A, Yamasmit N (2004) Use of sequencing batch reactors and Fenton’s reagent to treat a wastewater from a textile industry. Biochem Eng J 21:213–220. https://doi.org/10.1016/j.bej.2004.06.009
Nidheesh PV, Gandhimathi R (2013) Degradation of dyes from aqueous solution by Fenton processes: a review. Env Sci Pollut Res 20:2099–2132. https://doi.org/10.1007/s11356-012-1385-z
Rodrigues CSD, Madeira LM, Boaventura RAR (2009) Treatment of textile effluent by chemical (Fenton’ s Reagent) and biological (sequencing batch reactor) oxidation. J Hazard Mater 172:1551–1559. https://doi.org/10.1016/j.jhazmat.2009.08.027
Rodrigues CSD, Madeira LM, Boaventura RAR (2014) Synthetic textile dyeing wastewater treatment by integration of advanced oxidation and biological processes—Performance analysis with costs reduction. J Environ Chem Eng 2:1027–1039
Hayat H, Mahmood Q, Pervez A et al (2015) Comparative decolorization of dyes in textile wastewater using biological and chemical treatment. Sep Purif Technol 154:149–153. https://doi.org/10.1016/j.seppur.2015.09.025
Singh M, Srivastava RK (2011) Sequencing batch reactor technology for biological wastewater treatment: a review. Asia-pac J Chem Eng 3:199–203
Sirianuntapiboon S, Sadahiro O, Salee P (2007) Some properties of a granular activated carbon-sequencing batch reactor (GAC-SBR) system for treatment of textile wastewater containing direct dyes. J Environ Manag 85:162–170. https://doi.org/10.1016/j.jenvman.2006.09.001
Sirianuntapiboon S, Chairattanawan K, Jungphungsukpanich S (2006) Some properties of a sequencing batch reactor system for removal of vat dyes. Bioresour Technol 97:1243–1252. https://doi.org/10.1016/j.biortech.2005.02.052
Abu-ghunmi LN, Jamrah AI (2006) Biological treatment of textile wastewater using sequencing batch reactor technology. Environ Model Assess 11:333–343. https://doi.org/10.1007/s10666-005-9025-3
Fu LY, Wen XH, Yi Qian QLL (2001) Treatment of dyeing wastewater in two SBR systems. Process Biochem 36:1111–1118. https://doi.org/10.1016/S0032-9592(01)00143-1
Lourenco ND, Novais JM, Pinheiro HM (2000) Reactive textile dye colour removal in a sequencing batch reactor. Water Sci Technol 42:321–328
Khouni I, Marrot B, Ben R (2012) Treatment of reconstituted textile wastewater containing a reactive dye in an aerobic sequencing batch reactor using a novel bacterial consortium. Sep Purif Technol 87:110–119. https://doi.org/10.1016/j.seppur.2011.11.030
Mohan D, Pittman CU (2007) Arsenic removal from water/wastewater using adsorbents: a critical review. J Colloid Interface Sci 142:1–53. https://doi.org/10.1016/j.jhazmat.2007.01.006
Yang C, Kheireddine M, Mohd W, Wan A (2007) Review of modifications of activated carbon for enhancing contaminant uptakes from aqueous solutions. Sep Purif Technol 52:403–415. https://doi.org/10.1016/j.seppur.2006.06.009
Hai FI, Yamamoto K, Fukushi K (2007) Hybrid treatment systems for dye wastewater. Crit Rev Environ Sci Technol 37:315–377. https://doi.org/10.1080/10643380601174723
Mantzavinos D, Psillakis E (2004) Enhancement of biodegradability of industrial wastewaters by chemical oxidation pre-treatment. J Chem Technol Biotechnol 79:431–454. https://doi.org/10.1002/jctb.1020
Al-Kdasi A, Idris A, Saed K, Guan CT (2004) Treatment of textile wastewater by advanced oxidation processes: a review. Glob Nest J 6:222–230. https://doi.org/10.30955/gnj.000288
Lodha B, Chaudhari S (2007) Optimization of Fenton-biological treatment scheme for the treatment of aqueous dye solutions. J Hazard Mater 148:459–466. https://doi.org/10.1016/j.jhazmat.2007.02.061
Tantak NP, Chaudhari S (2006) Degradation of azo dyes by sequential Fenton’s oxidation and aerobic biological treatment. J Hazard Mater 136:698–705. https://doi.org/10.1016/j.jhazmat.2005.12.049
Guieysse B, Norvill ZN (2014) Sequential chemical-biological processes for the treatment of industrial wastewaters: review of recent progresses and critical assessment. J Hazard Mater 267:142–152. https://doi.org/10.1016/j.jhazmat.2013.12.016
Nair AT, Makwana AR, Ahammed MM (2014) The use of response surface methodology for modelling and analysis of water and wastewater treatment processes: a review. Water Sci Technol 69(3):464–478. https://doi.org/10.2166/wst.2013.733
Kapoor SSS, Sharma SRAC (2017) Effect of Fenton process on treatment of simulated textile wastewater: optimization using response surface methodology. Int J Environ Sci Technol. https://doi.org/10.1007/s13762-017-1253-y
Solomon D, Kiflie Z, Van HS (2019) Kinetic investigation and optimization of a sequencing batch reactor for the treatment of textile wastewater. Nanotechnol Environ Eng 4:15. https://doi.org/10.1007/s41204-019-0062-6
APHA (2005) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association (APHA), Washington, DC, USA
Moghaddam SS, Moghaddam MRA, Arami M (2010) Coagulation/flocculation process for dye removal using sludge from water treatment plant: optimization through response surface methodology. J Hazard Mater 175:651–657. https://doi.org/10.1016/j.jhazmat.2009.10.058
Zhu X, Tian J, Liu R, Chen L (2011) Optimization of Fenton and electro-Fenton oxidation of biologically treated coking wastewater using response surface methodology. Sep Purif Technol 81:444–450. https://doi.org/10.1016/j.seppur.2011.08.023
Xu H, Qi S, Li Y, Zhao Y (2013) Heterogeneous Fenton-like discoloration of Rhodamine B using natural schorl as catalyst: optimization by response surface methodology. Env Sci Pollut Res 20:5764–5772. https://doi.org/10.1007/s11356-013-1578-0
Fathinia M, Khataee AR, Zarei M, Aber S (2010) A: chemical Comparative photocatalytic degradation of two dyes on immobilized TiO2 nanoparticles: effect of dye molecular structure and response surface approach. J Mol Catal A Chem 333:73–84. https://doi.org/10.1016/j.molcata.2010.09.018
Rosales E, Sanromán MA, Pazos M (2012) Application of central composite face-centered design and response surface methodology for the optimization of electro-Fenton decolorization of Azure B dye. Env Sci Pollut Res 19:1738–1746. https://doi.org/10.1007/s11356-011-0668-0
Mason R, Gunst RHJ (2003) Statistical design and analysis of experiments with applications to engineering and science, 2nd edn. Wiley, New York
Saldana-Robels A, Gerra-Sanchez R, Maldonado-Rubio MP-HJ (2014) Optimization of the operating parameters using RSM for the Fenton oxidation process and adsorption on vegetal carbon of MO solutions. J Ind Eng Chem 20:848–857. https://doi.org/10.1016/j.jiec.2013.06.015
Arslan-alaton I, Tureli G, Olmez-hanci T (2009) Treatment of azo dye production wastewaters using Photo-Fenton-like advanced oxidation processes: optimization by response surface methodology. J Photochem Photobiol, A 202:142–153. https://doi.org/10.1016/j.jphotochem.2008.11.019
Mansoorian HJ, Bazrafshan E, Yari AAM (2014) Removal of Azo dyes from aqueous solution using Fenton and modified Fenton processes. Heal Scope 3:e15507
Wang CT, Chou WL, Chung MH, Kuo YM (2010) COD removal from real dyeing wastewater by electro-Fenton technology using an activated carbon fiber cathode. Desalination 253:129–134. https://doi.org/10.1016/j.desal.2009.11.020
Zhang H, Heung JC, Huang CP (2005) Optimization of Fenton process for the treatment of landfill leachate. J Hazard Mater 125:166–174. https://doi.org/10.1016/j.jhazmat.2005.05.025
Kaptan D (2004) Color and COD removal from wastewater containing Reactive Black 5 using Fenton’ s oxidation process S u. Chemosphere 54:435–441. https://doi.org/10.1016/j.chemosphere.2003.08.010
Ay F, Catalkaya EC, Kargi F (2009) A statistical experiment design approach for advanced oxidation of Direct Red azo-dye by photo-Fenton treatment. J Hazard Mater 162:230–236. https://doi.org/10.1016/j.jhazmat.2008.05.027
Mandal T, Maity S, Dasgupta D, Datta S (2010) Advanced oxidation process and biotreatment: their roles in combined industrial wastewater treatment. Desalination 250:87–94. https://doi.org/10.1016/j.desal.2009.04.012
Acknowledgements
First, we would like to thank NORAD project of Hawaasa University for the financial support. Finally, the authors would like to acknowledge laboratory workers of Addis Ababa Institute of Technology, Addis Ababa university for providing the necessary laboratory facility.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Solomon, D., Kiflie, Z. & Van Hulle, S. Integration of sequencing batch reactor and homo-catalytic advanced oxidation processes for the treatment of textile wastewater. Nanotechnol. Environ. Eng. 5, 7 (2020). https://doi.org/10.1007/s41204-020-0070-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41204-020-0070-6