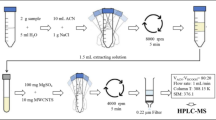
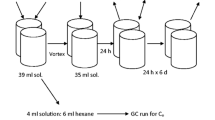
Abstract
Soil contaminated by organic pollutants such as dichlorodiphenyltrichloroethane (DDT) is an environmental concern due to the strong sorption of organochlorine pesticide onto the soil matrix and persistence in the environment. The remediation of contaminated soils with organochlorine pesticide using nanotechnology is an innovative technology for speeding up this process. This work presents a study of adsorption of DDT onto the zeolite surface. Experiments were conducted using batch adsorption procedures at different DDT concentrations, from 5 to 50 mg/L, and the amount of the zeolite used was 0.1, 0.5, 0.8, and 1.2 g. Results show that the zeolite has a moderate adsorption capacity for the DDT, and the highest adsorption capacity obtained from this study was about 30%. However, the percentage of adsorption can be increased significantly with the increase in the amount of the zeolite in samples. Also, adsorption kinetics and adsorption isotherms were applied. Five different kinetic models, i.e., pseudo-first-order kinetic model, the pseudo-second-order kinetic model, intraparticle diffusion model, Elovich kinetic model, and Bangham kinetic model were used to fit the kinetic data. The result shows that the pseudo-second-order model represented the best fits to the experiments. The adsorption isotherms were determined using three different models as well, i.e., Freundlich, Langmuir, and Temkin. The best-fitted adsorption isotherm models were found to be in both Langmuir and Freundlich. Moreover, results show that the effectiveness of treatment process is highly affected by pH. Increasing the pH has a negative effect on the adsorption process, and best uptake of DDT was noted in acidic media at pH 3.













Similar content being viewed by others
References
Sharma BK (2001) Soil noise polution, 2nd edn. Krishna Prakashan, Meerut
Mirsal IA (2004) Soil pollution: origin, monitoring & remediation. Springer, German
Misra DSG (2009) Soil pollution. APH Publishing Corporation, Delhi
Simeonov L, Sargsyan V (2008) Soil chemical pollution, risk assessment, remediation and security. In: Proceedings of the nato advanced research workshop on soil chemical pollution, risk assessment, remediation and security, Sofia, Bulgaria, 23–26 May 2007. Springer, Berlin
Houlding S (1994) Subsurface soil contamination assessment. In: Houlding SW (ed), 3d geoscience modeling. Springer, Berlin, pp 203–222. https://doi.org/10.1007/978-3-642-79012-6_12
FAO (2007) Assessing soil contamination: a reference manual. Daya Publishing House, Delhi
Pingali PL, Roger PA (1995) Impact of pesticides on farmer health and the rice environment. Kluwer Academic Publishers, Dordrecht
Garcia M (2004) Effects of pesticides on soil fauna: development of ecotoxicological test methods for tropical regions. Cuvillier
Sudharshan S, Naidu R, Mallavarapu M, Bolan N (2012) DDT remediation in contaminated soils: a review of recent studies. Biodegradation 23(6):851–863. https://doi.org/10.1007/s10532-012-9575-4
Zhang Y, Wong JWC, Zhao Z, Selvam A (2011) Microemulsion-enhanced remediation of soils contaminated with organochlorine pesticides. Environ Technol 32(16):1915–1922. https://doi.org/10.1080/09593330.2011.568009
Rios LE (2010) Removal of DDT from soil using combinations of surfactants. Thesis, University of Waterloo, Waterloo, Ontario, Canada
Wang X-P, Sheng J-J, Gong P, Xue Y-G, Yao T-D, Jones KC (2012) Persistent organic pollutants in the tibetan surface soil: spatial distribution, air-soil exchange and implications for global cycling. Environ Pollut 170:145–151. https://doi.org/10.1016/j.envpol.2012.06.012
Tremolada P, Comolli R, Parolini M, Moia F, Binelli A (2011) One-year cycle of DDT concentrations in high-altitude soils. Water Air Soil Pollut 217(1–4):407–419. https://doi.org/10.1007/s11270-010-0596-5
Dai R-L, Zhang G-Y, Gu X-Z, Wang M (2008) Sorption of 1,1,1-trichloro-2,2-bis(p-chlorophenyl) ethane (DDT) by clays and organoclays. Environ Geochem Health 30(5):479–488. https://doi.org/10.1007/s10653-007-9130-0
Calow P (2004) Euro chlor risk assessment for the marine environment osparcom region: north sea. Kluwer academic, dordrecht
Rodriguez-Morales AJ, Benítez JA, Arria M (2007) Malaria mortality in Venezuela: focus on deaths due to plasmodium vivax in children. J Trop Pediatr 54(2):94–101
McGinn AP (2002) Malaria, mosquitoes, and DDT: the toxic war against a global disease. WORLD•WATCH. Worldwatch Institute, Washington
Gautam SK, Suresh S (2007) Studies on dechlorination of DDT (1,1,1-trichloro-2,2-bis(4-chlorophenyl)ethane) using magnesium/palladium bimetallic system. J Hazard Mater 139(1):146–153. https://doi.org/10.1016/j.jhazmat.2006.06.017
Pushpavanam S (2002) Introduction to Chemical Engineering. PHI Learning, Delhi
Hajjar MJ (2012) The persisted organic pesticides pollutant (POPs) in the Middle East Arab Countries. Int J Agron Plant Prod 3(1):11–18
Ewing AE (2003) Water quality and public health monitoring of surface waters in the Kura-Araks river basin of Armenia, Azerbaijan and Georgia. Water Resources Program, University of New Mexico, Albuquerque
Howard PH (1991) Handbook of environmental fate and exposure data: for organic chemicals, volume III pesticides. Taylor & Francis, London
Zhongming G, Xuejun W, Bengang L, Jun C, Fuliu X, Yanhong C, Wenju Z, Weiran S, Baoping Q, Ren S, Zuo T (2003) The residues distribution of DDT and its metabolites in soils from Tianjin region, China. Acta Sci Circumst 23(4):447–451
Gong Z, Wang X, Li B, Cao J, Xu F, Cui Y, Zhang W, Shen W, Qin B, Sun Y, Tao S (2003) The residues distribution of DDT and its metabolites in soils from Tianjin region, China. Acta Sci Circumst 23(4):447–451
Taha OME, Taha MR (2015) Soil-water characteristic curves and hydraulic conductivity of nanomaterial-soil-bentonite mixtures. Arab J Geosci 9(1):12. https://doi.org/10.1007/s12517-015-2038-6
Taha M, Taha O (2012) Influence of nano-material on the expansive and shrinkage soil behavior. J Nanopart Res 14(10):1–13. https://doi.org/10.1007/s11051-012-1190-0
Varanasi P, Fullana A, Sidhu S (2007) Remediation of PCB contaminated soils using iron nano-particles. Chemosphere 66(6):1031–1038. https://doi.org/10.1016/j.chemosphere.2006.07.036
Reyes CR, Appasamy D, Roberts C (2011) An integrated remediation system using synthetic and natural zeolites for treatment of wastewater and contaminated sediments. Dynamics 78:125–134
Shanableh A, Kharabsheh A (1996) Stabilization of Cd, Ni and Pb in soil using natural zeolite. J Hazard Mater 45(2–3):207–217. https://doi.org/10.1016/0304-3894(95)00093-3
Gholikandi GB, Baneshi MM, Dehghanifard E, Salehi S, Yari AR (2010) Natural zeolites application as sustainable adsorbent for heavy metals removal from drinking water. Iran J Toxicol 3(3):302–310
Tahir H, Uddin F (2005) Estimation of Pb from metal and electroplating industrial waste by zeolite-3a. J Appl Sci Environ Manag 9(2):49–55. http://dx.doi.org/10.4314/jasem.v9i2.17290
Shi W-Y, Shao H-B, Li H, Shao M-A, Du S (2009) Progress in the remediation of hazardous heavy metal-polluted soils by natural zeolite. J Hazard Mater 170(1):1–6. https://doi.org/10.1016/j.jhazmat.2009.04.097
Burlakovs J, Klavins M, Karklina A (2012) Remediation of soil contamination with heavy metals by using zeolite and humic acid additives. Latv J Chem. https://doi.org/10.2478/v10161-012-0019-6
Sayed SA (1996) Exchange of Zn2+, V2+, Cd2+, and Hg2+ using zeolite a and dinonylnaphthalenesulfonate. Zeolites 17(3):261–264. https://doi.org/10.1016/0144-2449(96)00040-1
Samarghandi MR, Al-Musawi TJ, Mohseni-Bandpi A, Zarrabi M (2015) Adsorption of cephalexin from aqueous solution using natural zeolite and zeolite coated with manganese oxide nanoparticles. J Mol Liq 211(Supplement C):431–441. https://doi.org/10.1016/j.molliq.2015.06.067
Mohseni-Bandpi A, Al-Musawi TJ, Ghahramani E, Zarrabi M, Mohebi S, Vahed SA (2016) Improvement of zeolite adsorption capacity for cephalexin by coating with magnetic Fe3o4 nanoparticles. J Mol Liq 218(Supplement C):615–624. https://doi.org/10.1016/j.molliq.2016.02.092
Qiu H, Lv L, Pan B-C, Zhang Q-J, Zhang W-M, Zhang Q-X (2009) Critical review in adsorption kinetic models. J Zhejiang Univ Sci A 10(5):716–724. https://doi.org/10.1631/jzus.A0820524
Tan IAW, Ahmad AL, Hameed BH (2009) Adsorption isotherms, kinetics, thermodynamics and desorption studies of 2,4,6-trichlorophenol on oil palm empty fruit bunch-based activated carbon. J Hazard Mater 164(2–3):473–482. https://doi.org/10.1016/j.jhazmat.2008.08.025
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34(5):451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Kalhori EM, Al-Musawi TJ, Ghahramani E, Kazemian H, Zarrabi M (2017) Enhancement of the adsorption capacity of the light-weight expanded clay aggregate surface for the metronidazole antibiotic by coating with mgo nanoparticles: studies on the kinetic, isotherm, and effects of environmental parameters. Chemosphere 175(Supplement C):8–20. https://doi.org/10.1016/j.chemosphere.2017.02.043
Shahmohammadi-Kalalagh S, Babazadeh H, Nazemi AH, Manshouri M (2011) Isotherm and kinetic studies on adsorption of Pb, Zn and Cu by kaolinite. Casp J Environ Sci 9(2):243–255
Ho YS, McKay G (1998) Sorption of dye from aqueous solution by peat. Chem Eng J 70(2):115–124. https://doi.org/10.1016/S0923-0467(98)00076-1
Tütem E, Apak R, Ünal ÇF (1998) Adsorptive removal of chlorophenols from water by bituminous shale. Water Res 32(8):2315–2324. https://doi.org/10.1016/S0043-1354(97)00476-4
Samadi MT, Rahman AR, Zarrabi M, Shahabi E, Sameei F (2009) Adsorption of chromium (VI) from aqueous solution by sugar beet bagasse-based activated charcoal. Environ Technol 30(10):1023–1029. https://doi.org/10.1080/09593330903045107
Wu F-C, Tseng R-L, Juang R-S (2001) Kinetic modeling of liquid-phase adsorption of reactive dyes and metal ions on chitosan. Water Res 35(3):613–618. https://doi.org/10.1016/S0043-1354(00)00307-9
Yang X, Al-Duri B (2005) Kinetic modeling of liquid-phase adsorption of reactive dyes on activated carbon. J Colloid Interface Sci 287(1):25–34. https://doi.org/10.1016/j.jcis.2005.01.093
Weber WJ, Morris JC (1962) Advances in water pollution research: removal of biologically resistant pollutants from waste waters by adsorption. In: Proceedings of international conference on water pollution symposium. Pergamon Press, Oxford, pp 231–266
Alkan M, Demirbaş Ö, Doğan M (2007) Adsorption kinetics and thermodynamics of an anionic dye onto sepiolite. Microporous Mesoporous Mater 101(3):388–396. https://doi.org/10.1016/j.micromeso.2006.12.007
Zeldowitsch J (1934) Über Den Mechanismus Der Katalytischen Oxydation Von Co an Mno2. Acta Physicochem URSS
Yakout SM, Elsherif E (2010) Batch kinetics, isotherm and thermodynamic studies of adsorption of strontium from aqueous solutions onto low cost rice-straw based carbons. Carbon Sci Technol 3(1):144–153
Yousef RI, El-Eswed B, Al-Muhtaseb AAH (2011) Adsorption characteristics of natural zeolites as solid adsorbents for phenol removal from aqueous solutions: kinetics, mechanism, and thermodynamics studies. Chem Eng J 171(3):1143–1149. https://doi.org/10.1016/j.cej.2011.05.012
Bhattacharyya R, Ray SK (2013) Kinetic and equilibrium modeling for adsorption of textile dyes in aqueous solutions by carboxymethyl cellulose/poly(acrylamide-Co-hydroxyethyl methacrylate) semi-interpenetrating network hydrogel. Polym Eng Sci 53(11):2439–2453. https://doi.org/10.1002/pen.23501
Temkin MJ, Pyzhev V (1940) Kinetics of ammonia synthesis on promoted iron catalysts. Acta Physiochim 12:217–222
Mall ID, Srivastava VC, Agarwal NK (2006) Removal of orange-G and methyl violet dyes by adsorption onto bagasse fly ash—kinetic study and equilibrium isotherm analyses. Dyes Pigment 69(3):210–223. https://doi.org/10.1016/j.dyepig.2005.03.013
Bhatnagar A (2007) Removal of bromophenols from water using industrial wastes as low cost adsorbents. J Hazard Mater 139(1):93–102. https://doi.org/10.1016/j.jhazmat.2006.06.139
Sepehr MN, Yetilmezsoy K, Marofi S, Zarrabi M, Ghaffari HR, Fingas M, Foroughi M (2014) Synthesis of nanosheet layered double hydroxides at lower pH: optimization of hardness and sulfate removal from drinking water samples. J Taiwan Inst Chem Eng 45(5):2786–2800. https://doi.org/10.1016/j.jtice.2014.07.013
Húmpola PD, Odetti HS, Fertitta AE, Vicente JL (2013) Thermodynamic analysis of adsorption models of phenol in liquid phase on different activated carbons. J Chil Chem Soc 58(1):1541–1544. https://doi.org/10.4067/S0717-97072013000100009
Pan B, Xing B (2008) Adsorption mechanisms of organic chemicals on carbon nanotubes. Environ Sci Technol 42(24):9005–9013. https://doi.org/10.1021/es801777n
Soori MM, Ghahramani E, Kazemian H, Al-Musawi TJ, Zarrabi M (2016) Intercalation of tetracycline in nano sheet layered double hydroxide: an insight into UV/Vis spectra analysis. J Taiwan Inst Chem Eng 63(Supplement C):271–285. https://doi.org/10.1016/j.jtice.2016.03.015
Poursaberi T, Konozb E, Sarrafi AHM, Hassanisadi M, Hajifathli F (2012) Application of nanoscale zero-valent iron in the remediation of DDT from contaminated water. Chem Sci Trans 1(3):658–668. https://doi.org/10.7598/cst2012.246
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmed, S.M., Taha, M.R. & Taha, O.M.E. Kinetics and isotherms of dichlorodiphenyltrichloroethane (DDT) adsorption using soil–zeolite mixture. Nanotechnol. Environ. Eng. 3, 4 (2018). https://doi.org/10.1007/s41204-017-0033-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41204-017-0033-8